 Back
BackSolutions and Their Properties (General Chemistry II, Ch. 13 Study Notes)
Study Guide - Smart Notes

Solutions and Their Properties
Introduction to Solutions
Solutions are homogeneous mixtures composed of two or more substances. The study of solutions is fundamental in chemistry, as it relates to solubility, concentration, and the physical properties of mixtures. This chapter explores the energetics, structure, and behavior of solutions, with a focus on aqueous systems.
Solubility and Intermolecular Forces
Predicting Solubility
Solubility is the ability of a solute to dissolve in a solvent to form a homogeneous mixture.
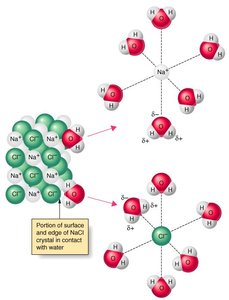
"Like dissolves like": Polar solutes dissolve in polar solvents, and nonpolar solutes dissolve in nonpolar solvents.
Key factors: Intermolecular forces (IMFs), molecular polarity, and the structure of solute and solvent molecules.
Types of solids: Covalent, ionic, atomic/network solids.
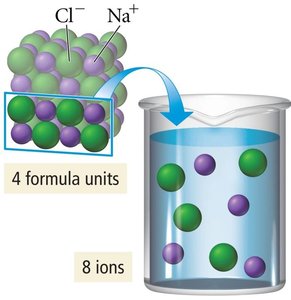
Example: Water (a polar solvent) dissolves ionic compounds like NaCl due to strong ion-dipole interactions.
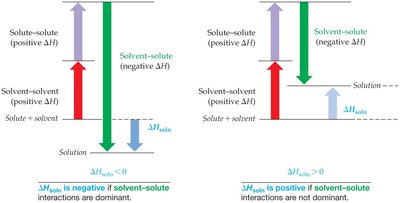
Energetics of Solution Formation
Enthalpy of solution (ΔHsolution): The overall energy change when a solution forms.
Steps in solution formation:
Breaking solute-solute IMFs (endothermic, ΔH > 0)
Breaking solvent-solvent IMFs (endothermic, ΔH > 0)
Forming solute-solvent IMFs (exothermic, ΔH < 0)
ΔHsolution = ΔHsolute-solute + ΔHsolvent-solvent + ΔHsolute-solvent
Solutions form when the energy released in forming solute-solvent interactions compensates for the energy required to separate solute and solvent particles.
Solvation and Entropy
Solvation: The process of surrounding solute particles with solvent molecules.
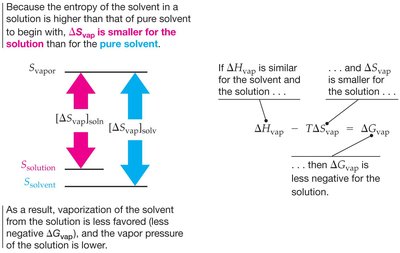
Entropy (ΔS): A measure of molecular randomness or disorder. Dissolution increases entropy (ΔS > 0).
Spontaneity of solution formation is determined by Gibbs Free Energy (ΔG):
A process is spontaneous if ΔG < 0.
Concentration Units
Common Concentration Units
Mass percent (%):
Mole fraction (χ):
Molarity (M):
Molality (m):
Example: Calculate the molality of a solution containing 1.00 g ethanol in 100.0 g water: m.
Types of Mixtures: Solutions, Colloids, and Suspensions
Classification of Mixtures
Solution: Homogeneous mixture, particle size 0.1–2 nm (e.g., saltwater).
Colloid: Heterogeneous mixture, particle size 2–1000 nm, exhibits the Tyndall effect (e.g., milk).
Suspension: Heterogeneous mixture, particle size >1 μm, particles settle out (e.g., muddy water).

Types of Solutions
Kind of Solution | Example |
|---|---|
Gas in gas | Air (O2, N2, Ar, etc.) |
Gas in liquid | Carbonated water (CO2 in water) |
Gas in solid | H2 in palladium metal |
Liquid in liquid | Gasoline (mixture of hydrocarbons) |
Liquid in solid | Dental amalgam (mercury in silver) |
Solid in liquid | Seawater (NaCl and other salts in water) |
Solid in solid | Metal alloys (e.g., sterling silver) |

Solubility Trends
Temperature and Pressure Effects
Solubility of solids generally increases with temperature.
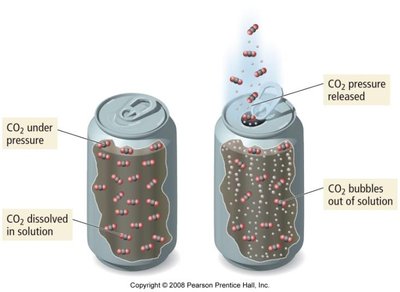
Solubility of gases decreases with increasing temperature but increases with pressure (Henry's Law).
Henry's Law:
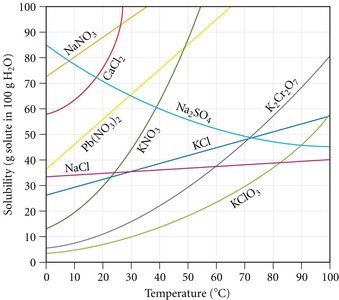
Solubility Curves
Solubility curves show how much solute dissolves in 100 g of water at various temperatures.
Used to determine if a solution is saturated, unsaturated, or supersaturated at a given temperature.
Colligative Properties
Definition and Types
Colligative properties depend on the number of solute particles, not their identity.
Key colligative properties:
Vapor pressure lowering
Boiling point elevation
Freezing point depression
Osmotic pressure
Vapor Pressure Lowering and Raoult's Law
Adding a non-volatile solute lowers the vapor pressure of the solvent.
Raoult's Law:
Boiling Point Elevation and Freezing Point Depression
Boiling point elevation:
Freezing point depression:
Where m is molality, i is the van't Hoff factor, and K_b, K_f are solvent-specific constants.
Example: Calculate the freezing point of a 0.89 m CaCl2 solution (i = 3): New freezing point =
Osmotic Pressure
Osmotic pressure () is the pressure required to stop osmosis.
Calculated by:
Where M is molarity, R is the gas constant, T is temperature in Kelvin, and i is the van't Hoff factor.
Van't Hoff Factor and Ion Pairing
The van't Hoff factor (i) is the number of particles a solute produces in solution.
For non-electrolytes, i = 1; for ionic compounds, i equals the number of ions formed.
Ion pairing can cause deviations from the ideal value of i.
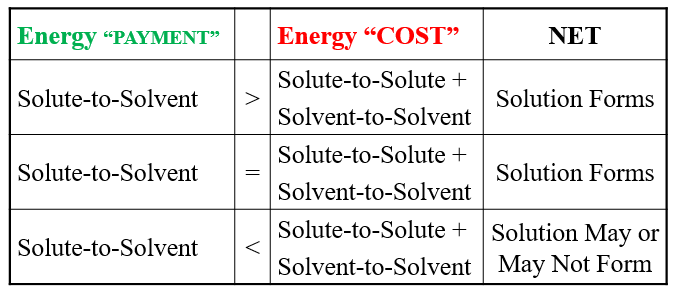
Summary Table: Energy Considerations in Solution Formation
Energy "PAYMENT" | Energy "COST" | NET |
|---|---|---|
Solute-to-Solvent > | Solute-to-Solute + Solvent-to-Solvent | Solution Forms |
Solute-to-Solvent = | Solute-to-Solute + Solvent-to-Solvent | Solution Forms |
Solute-to-Solvent < | Solute-to-Solute + Solvent-to-Solvent | Solution May or May Not Form |
Practice Problems and Applications
Calculate concentrations using different units (molarity, molality, mole fraction, mass percent).
Apply colligative property equations to determine boiling point elevation, freezing point depression, and osmotic pressure.
Interpret solubility curves and predict solution behavior under varying conditions.
Key Equations
Additional info: For cumulative problems, use logical assumptions (e.g., 100 g solution for mass %, 1 L for molarity, 1 kg solvent for molality) to simplify calculations.
