 Back
BackSolutions, Dissolution, Electrolytes, and Solubility: Study Notes for General Chemistry
Study Guide - Smart Notes

Solutions and Solution Formation
Definition and Properties of Solutions
A solution is a homogeneous mixture composed of two or more substances. The solvent is the component present in the greatest amount, while the solute is present in a lesser amount. Solutions can exist in any phase (solid, liquid, or gas), and the solute is distributed at the molecular level throughout the solvent.
Homogeneous mixtures: Uniform composition throughout.
Solvent defines physical state: The phase of the solution matches the solvent.
Permanent mixing: Solute does not settle out over time.
Concentration: Amount of solute per unit solvent, varies up to the solubility limit.
Miscibility and Intermolecular Forces
Miscibility refers to the ability of two liquids to mix and form a homogeneous solution. This process is governed by intermolecular forces (IMFs). The general rule is "like dissolves like": substances with similar polarity and IMFs will mix, while those with different IMFs will not.
Miscible: Liquids that mix to form a solution (e.g., ethanol and water).
Immiscible: Liquids that do not mix (e.g., oil and water).

Dissolution Reactions
Writing Dissolution Reactions
Dissolution reactions describe how a solute dissolves in a solvent, often water. For ionic compounds, the reaction shows the separation into ions; for covalent compounds, the molecule simply enters the aqueous phase.
Ionic compound example:
Covalent compound example:
Common Ions in Solution
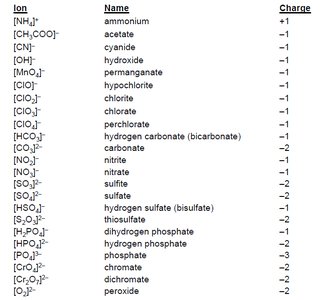
Many dissolution reactions involve common ions. Recognizing these ions and their charges is essential for predicting solubility and writing reactions.
Ion | Name | Charge |
|---|---|---|
NH4+ | ammonium | +1 |
CH3COO- | acetate | -1 |
NO3- | nitrate | -1 |
SO42- | sulfate | -2 |
PO43- | phosphate | -3 |
Solubility and Solubility Rules
Solubility Rules for Ionic Compounds
Solubility rules help predict whether an ionic compound will dissolve in water. These rules are based on the ions present in the compound.
Salts containing nitrate, chlorate, perchlorate, and acetate are soluble.
Salts containing alkali metal cations and ammonium ions are soluble.
Halides (Cl-, Br-, I-) are soluble except with Ag+, Pb2+, or Hg22+.
Sulfates are soluble except with Ca2+, Sr2+, Ba2+, Ag+, or Pb2+.
Carbonates, phosphates, and chromates are insoluble except with alkali metals or ammonium.
Hydroxides and oxides are insoluble except with alkali metals or ammonium; Ba(OH)2 and Sr(OH)2 are somewhat soluble.

Energetics and Entropy of Solution Formation
Energy Changes in Solution Formation
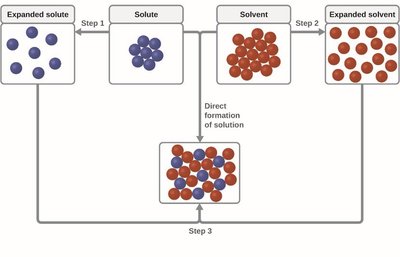
Solution formation involves breaking and forming intermolecular forces. The process can be exothermic or endothermic depending on the relative strengths of solute-solute, solvent-solvent, and solute-solvent interactions.
Exothermic: Solute-solvent interactions are stronger than the original interactions.
Endothermic: Solute-solvent interactions are weaker than solute-solute and solvent-solvent interactions.
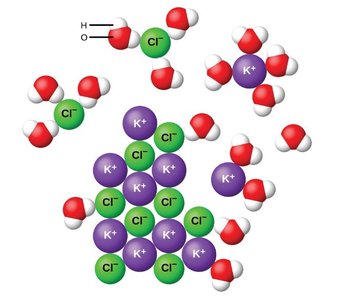
Ion-Dipole Forces in Aqueous Solutions
When ionic compounds dissolve in water, ion-dipole forces form between the ions and the polar water molecules. Water molecules orient themselves so that the partially negative oxygen faces cations and the partially positive hydrogens face anions, stabilizing the ions in solution.
Entropy and Spontaneity of Solution Formation
Entropy is a measure of the number of ways a system can be arranged. Solution formation increases entropy, which favors the process even if it is endothermic. The increase in disorder as solute particles disperse throughout the solvent is a key driving force for dissolution.
Ideal solution: No net energy change (), but entropy still increases.
Spontaneity: Solution formation can be spontaneous due to increased entropy.
Electrolytes and Electrical Conductivity
Types of Electrolytes
Electrolytes are substances that produce ions in solution and conduct electricity. They are classified as strong, weak, or nonelectrolytes based on the extent of ion formation.
Strong electrolytes: Completely dissociate into ions (e.g., soluble ionic compounds, strong acids, strong bases).
Weak electrolytes: Partially dissociate, producing some ions (e.g., weak acids, weak bases).
Nonelectrolytes: Do not produce ions; do not conduct electricity (e.g., most covalent compounds).
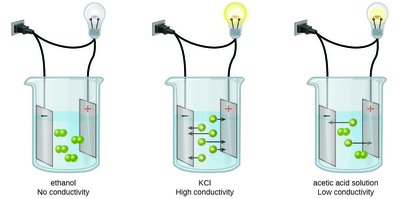
Examples of Electrolytes
Electrolyte solutions are important in biological and industrial contexts. For example, sports drinks contain electrolytes to help maintain ion balance in the body.

Solubility Vocabulary and Equilibrium
Key Terms in Solubility
Solubility describes how much solute can dissolve in a solvent at equilibrium. Several terms are used to describe the state of a solution:
Unsaturated: Contains less solute than the solubility limit.
Saturated: Contains solute at the solubility limit; additional solute will not dissolve.
Supersaturated: Contains more solute than the solubility limit; unstable and will precipitate excess solute.
Dynamic equilibrium: Rate of dissolution equals rate of precipitation.
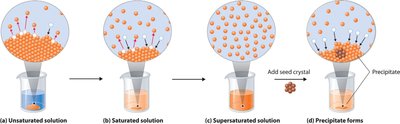
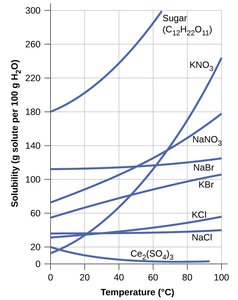
Solubility and Temperature
Effect of Temperature on Solubility
For most solid solutes, solubility increases with temperature. For gases, solubility decreases as temperature increases. This principle is used in kitchen science (e.g., making simple syrup) and laboratory techniques (e.g., recrystallization).



Solubility and Pressure: Henry's Law
Henry's Law for Gas Solubility
Henry's Law states that the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the liquid.
Equation:
kH: Henry's Law constant (units: M atm-1)
Pgas: Partial pressure of the gas
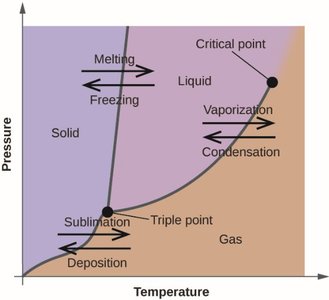
Phase Diagrams and Heating Curves
Phase Changes and Diagrams
Phase diagrams show the relationship between pressure, temperature, and the phases of a substance. Key features include the triple point (where all three phases coexist) and the critical point (beyond which the liquid and gas phases are indistinguishable).
Phase changes: Melting, freezing, vaporization, condensation, sublimation, deposition
Heating curves: Show temperature changes as heat is added, with plateaus at phase change temperatures
Summary Table: Solution Types and Properties
Type | Definition | Example |
|---|---|---|
Strong Electrolyte | Complete ionization in solution | KCl, NaOH |
Weak Electrolyte | Partial ionization in solution | Acetic acid, NH3 |
Nonelectrolyte | No ionization in solution | Glucose, ethanol |
