 Back
BackSolutions: Properties, Formation, and Concentration in General Chemistry
Study Guide - Smart Notes

Solutions
Introduction to Solutions
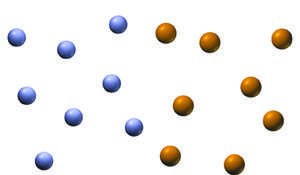
Solutions are homogeneous mixtures in which atoms, ions, or molecules of two or more substances are uniformly mixed. The majority component is called the solvent, and the minority component is the solute. For example, in seawater, water is the solvent and sodium chloride is the primary solute.
Homogeneous mixture: Uniform composition throughout.
Solvent: The component present in the largest amount.
Solute: The component present in a lesser amount.
Example: Ocean water, gasoline, and air are all solutions.

Why Solutions Form
Solutions form due to the natural tendency toward mixing and increased entropy (energy dispersal). Unless energetically unfavorable, substances tend to mix spontaneously, leading to uniform mixtures.
Spontaneous mixing: Driven by entropy, not always by a decrease in potential energy.
Example: When pure water and a sodium chloride solution are separated by a barrier, removing the barrier leads to spontaneous mixing and a uniform solution.
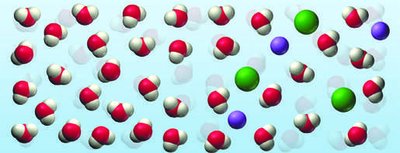
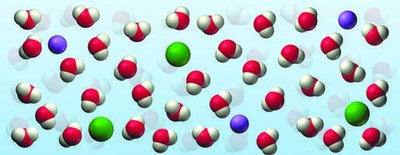
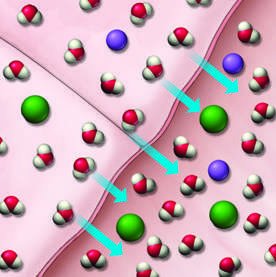
Osmosis and Biological Relevance
Seawater is a more concentrated solution than the fluids in body cells. When seawater passes through the digestive tract, it draws water out of body tissues due to osmosis, leading to dehydration.
Osmosis: Movement of water from a region of lower solute concentration to higher solute concentration through a semipermeable membrane.
Biological impact: Drinking seawater causes dehydration because it draws water out of cells.
Types of Solutions and Solubility
Types of Solutions
Solutions can be composed of various combinations of solids, liquids, and gases. In aqueous solutions, water is the solvent, and the solute can be a solid, liquid, or gas.
Examples: Salt water (solid in liquid), club soda (gas in liquid), vodka (liquid in liquid), brass (solid in solid).
Solution Phase | Solute Phase | Solvent Phase | Example |
|---|---|---|---|
Gaseous | Gas | Gas | Air (O2 and N2) |
Liquid | Gas | Liquid | Club soda (CO2 in water) |
Liquid | Liquid | Liquid | Vodka (ethanol and water) |
Liquid | Solid | Liquid | Seawater (salt in water) |
Solid | Solid | Solid | Brass (copper and zinc) |


Solubility and Intermolecular Forces
The solubility of a substance is the amount that will dissolve in a given amount of solvent. Solubility depends on the tendency toward mixing (entropy) and the types of intermolecular forces present.
Like dissolves like: Polar solvents dissolve polar or ionic solutes; nonpolar solvents dissolve nonpolar solutes.
Intermolecular forces: Dispersion, dipole-dipole, hydrogen bonding, ion-dipole.

Energetics of Solution Formation
Enthalpy Changes in Solution Formation
Solution formation involves three steps, each with an associated enthalpy change:
Separating solute particles: (endothermic)
Separating solvent particles: (endothermic)
Mixing solute and solvent: (exothermic)
The overall enthalpy change is:

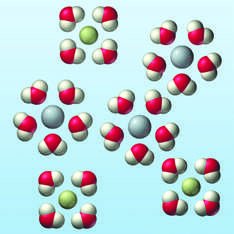
Heats of Hydration
For ionic compounds in water, the heat of hydration () is the enthalpy change when 1 mol of gaseous ions dissolves in water. It is always exothermic due to strong ion-dipole interactions.
Solution Equilibrium and Factors Affecting Solubility
Dynamic Equilibrium in Solutions
When a solute dissolves in a solvent, an equilibrium is established between dissolution and recrystallization. A saturated solution contains the maximum amount of dissolved solute at equilibrium. An unsaturated solution contains less than the equilibrium amount, and a supersaturated solution contains more (unstable).

Temperature and Solubility
The solubility of most solids in water increases with temperature, while the solubility of gases decreases with temperature. Pressure increases the solubility of gases in liquids (Henry's Law).
Henry's Law:


Expressing Solution Concentration
Common Concentration Units
Molarity (M):
Molality (m):
Percent by mass:
Parts per million (ppm):
Mole fraction (x):

Colligative Properties
Vapor Pressure Lowering
The addition of a nonvolatile solute to a solvent lowers the vapor pressure of the solution compared to the pure solvent. This is described by Raoult's Law:
The decrease in vapor pressure is proportional to the mole fraction of the solute:
Freezing Point Depression and Boiling Point Elevation
The presence of a solute lowers the freezing point and raises the boiling point of a solvent. These are colligative properties, depending only on the number of solute particles.
Freezing point depression:
Boiling point elevation:
Osmotic Pressure
Osmotic pressure is the pressure required to stop the flow of solvent through a semipermeable membrane from a dilute to a concentrated solution:
Colligative Properties of Electrolyte Solutions
For electrolytes, the van't Hoff factor (i) accounts for the number of particles produced per formula unit dissolved:
Colligative property equations for electrolytes:
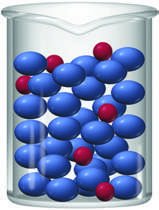
Colloids
Colloidal Dispersions
A colloid is a mixture where the dispersed particles are intermediate in size between those in solutions and suspensions (1 nm to 1000 nm). Colloids scatter light (Tyndall effect) and remain dispersed due to Brownian motion and electrostatic repulsions.
Examples: Fog, milk, whipped cream, soapy water.
Summary Table: Types of Solutions
Type | Solute | Solvent | Example |
|---|---|---|---|
Gas in gas | O2 | N2 | Air |
Gas in liquid | CO2 | H2O | Club soda |
Liquid in liquid | Ethanol | Water | Vodka |
Solid in liquid | NaCl | Water | Seawater |
Solid in solid | Zn | Cu | Brass |
Key Equations
Henry's Law:
Molarity:
Molality:
Percent by mass:
Raoult's Law:
Freezing point depression:
Boiling point elevation:
Osmotic pressure:
