 Back
BackStoichiometry and Chemical Calculations: Step-by-Step Guidance
Study Guide - Smart Notes

Q1. What is the formula (molecular) weight of a compound? How do you calculate the formula weight of a compound such as acetic acid?
Background
Topic: Stoichiometry and Formula/Molecular Weight
This question tests your understanding of how to determine the formula (or molecular) weight of a chemical compound by summing the atomic masses of all atoms present in its formula.
Key Terms and Formulas:
Formula Weight (FW): The sum of the atomic masses of all atoms in a chemical formula, usually expressed in atomic mass units (amu) or grams per mole (g/mol).
Molecular Weight: For molecules, this is the same as formula weight, but specifically refers to covalently bonded compounds.
Atomic Mass: The mass of a single atom, typically found on the periodic table.
Key Formula:
Step-by-Step Guidance
Write the chemical formula for the compound. For acetic acid, the formula is or .
List the number of each type of atom in the formula. For acetic acid: C = 2, H = 4, O = 2.
Look up the atomic masses for each element (C, H, O) on the periodic table. For example: C = 12.01, H = 1.008, O = 16.00 (in amu or g/mol).
Multiply the number of each atom by its atomic mass, then sum the results to get the total formula weight.


Try solving on your own before revealing the answer!
Final Answer: The formula weight of acetic acid is 60.05 g/mol
Total = g/mol
This is the sum of the atomic masses for all atoms in acetic acid.
Q2. What is the percent composition of an element in a compound? Calculate the percent composition of carbon in glucose () to three significant figures.
Background
Topic: Percent Composition
This question tests your ability to calculate the percent by mass of a specific element in a compound, which is important for empirical formula and stoichiometry problems.
Key Terms and Formulas:
Percent Composition: The percentage by mass of each element in a compound.
Key Formula:
Step-by-Step Guidance
Write the chemical formula for glucose: .
Calculate the molar mass of glucose by summing the atomic masses of all atoms in the formula.
Calculate the total mass of carbon in one mole of glucose: g.
Divide the mass of carbon by the molar mass of glucose, then multiply by 100% to get the percent composition.

Try solving on your own before revealing the answer!
Final Answer: The percent composition of carbon in glucose is 40.0%
This means that 40.0% of the mass of glucose is due to carbon atoms.
Q3. How many moles of oxygen are in 0.312 mol of sodium nitrate ()?
Background
Topic: Mole Relationships in Compounds
This question tests your understanding of how to use chemical formulas to determine the number of moles of a specific atom in a given amount of compound.
Key Terms and Formulas:
Mole: A unit representing entities (Avogadro's number).
Mole Ratio: The ratio of the number of moles of a particular atom to the number of moles of the compound.
Step-by-Step Guidance
Write the formula for sodium nitrate: .
Identify the number of oxygen atoms per formula unit (there are 3 O atoms in ).
Set up a conversion factor: $1\mathrm{NaNO_3} moles of O atoms.
Multiply the given moles of by the ratio to find moles of O atoms.
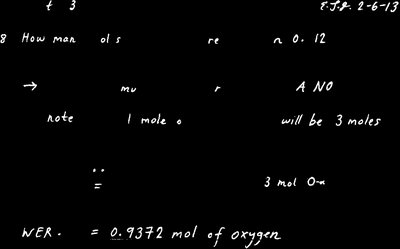
Try solving on your own before revealing the answer!
Final Answer: 0.936 mol of oxygen atoms
There are three times as many moles of oxygen as sodium nitrate.
Q4. How many molecules are in 5.832 g of methane ()? How many atoms of hydrogen are in this sample?
Background
Topic: Molecules and Atoms from Mass
This question tests your ability to convert grams to moles, then to molecules, and finally to atoms using Avogadro's number and the chemical formula.
Key Terms and Formulas:
Molar Mass: The mass of one mole of a substance (g/mol).
Avogadro's Number: entities/mol.
Key Formulas:
Step-by-Step Guidance
Calculate the molar mass of methane (): g/mol.
Convert 5.832 g of methane to moles using the molar mass.
Multiply the number of moles by Avogadro's number to get the number of molecules.
Multiply the number of molecules by 4 to get the number of hydrogen atoms.

Try solving on your own before revealing the answer!
Final Answer: molecules of methane, hydrogen atoms
First, convert grams to moles, then to molecules, and finally multiply by 4 for hydrogen atoms.
