 Back
BackChapter 3 Chemistry
Study Guide - Smart Notes

Chemical Equations and Stoichiometry
Representing Chemical Reactions
Chemical reactions are represented by chemical equations, which show the reactants and products involved. The numbers in front of the formulas are called coefficients and indicate the relative number of molecules or moles of each substance.
Balanced chemical equations have equal numbers of atoms of each element on both sides of the arrow, reflecting the law of conservation of mass.
Coefficients change the amount of a substance, not its identity; subscripts define the identity and should never be changed when balancing equations.
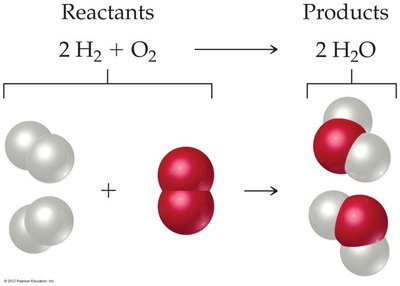
Balancing Chemical Equations
Balancing equations ensures that the number of atoms for each element is the same on both sides. This is achieved by adjusting coefficients, not subscripts.
Example:
Balance hydrogen and oxygen atoms by adjusting coefficients.
Types of Chemical Reactions
Combination Reactions
Two or more reactants combine to form a single product.
General form:
Example:
Decomposition Reactions
A single reactant breaks apart to form two or more products.
General form:
Example:
Single Replacement Reactions
One element replaces another in a compound.
General form:
Example:
Double Replacement Reactions
Two compounds exchange ions to form two new compounds.
General form:
Example:
Combustion Reactions
Combustion reactions are rapid and produce a flame, typically involving oxygen as a reactant and forming CO2 and H2O.
Example:
Formula Weights and Percentage Composition
Formula Weight (FW)
The formula weight is the sum of the atomic weights of all atoms in a chemical formula.
Example: FW of HNO3 = 1(1.01) + 1(14.01) + 3(15.99) = 62.99 amu
Percentage Composition
The percentage composition is the mass percentage of each element in a compound.
Formula:
Example: In C12H22O11, %C = 42.1%, %H = 6.5%, %O = 51.4%
The Mole and Avogadro’s Number
Definition of the Mole
The mole is the counting unit for atoms, ions, or molecules. One mole contains Avogadro’s number () of particles.
1 mol H2O = molecules H2O
1 mol NaCl = formula units NaCl
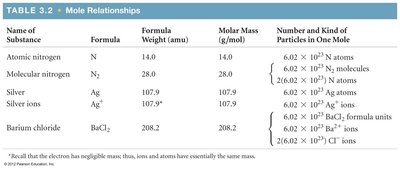
Mole Relationships Table
This table summarizes the relationship between formula weight, molar mass, and the number of particles in one mole for various substances.
Name of Substance | Formula | Formula Weight (amu) | Molar Mass (g/mol) | Number and Kind of Particles in One Mole |
|---|---|---|---|---|
Atomic nitrogen | N | 14.0 | 14.0 | 6.02 × 1023 N atoms |
Molecular nitrogen | N2 | 28.0 | 28.0 | 6.02 × 1023 N2 molecules |
Silver | Ag | 107.9 | 107.9 | 6.02 × 1023 Ag atoms |
Silver ions | Ag+ | 107.9 | 107.9 | 6.02 × 1023 Ag+ ions |
Barium chloride | BaCl2 | 208.2 | 208.2 | 6.02 × 1023 BaCl2 formula units, 6.02 × 1023 Ba2+ ions, 2 × 6.02 × 1023 Cl- ions |
Molar Mass and Conversions
Molar Mass
The molar mass (MM) is the mass in grams of one mole of a substance and is numerically equal to its formula weight in atomic mass units.
Example: MM of C6H12O6 = 180.12 g/mol
Converting Between Mass, Moles, and Particles
Conversions between grams, moles, and number of particles use the molar mass and Avogadro’s number.
To convert grams to moles:
To convert moles to particles:
Empirical and Molecular Formulas
Empirical Formula
The empirical formula gives the simplest whole-number ratio of atoms in a compound. It can be determined from percent composition.
Steps: Convert mass to moles, divide by the smallest number of moles, adjust to whole numbers if necessary.
Example: Sulfuric acid (H2SO4) has an empirical formula identical to its molecular formula.
Molecular Formula
The molecular formula is a whole-number multiple of the empirical formula, determined by the ratio of the molecular weight to the empirical formula weight.
Formula:
Example: Glucose (C6H12O6) has an empirical formula CH2O and a multiple of 6.
Quantitative Information from Balanced Equations
Stoichiometric Calculations
Balanced equations provide mole ratios for converting between reactants and products. These ratios are used in calculations involving mass, moles, and number of particles.
Example:
For every 2 moles of H2, 1 mole of O2 is consumed, and 2 moles of H2O are produced.
Limiting Reactants and Percent Yield
Limiting Reactant
The limiting reactant is the reactant that is completely consumed first, determining the maximum amount of product formed.
Any excess reactant will be left over after the reaction.
Example: In making sandwiches, if you have 14 slices of bread and 6 slices of cheese, you can make only 6 sandwiches; cheese is the limiting reactant.






Theoretical Yield and Percent Yield
The theoretical yield is the maximum amount of product that can be formed from the limiting reactant. The actual yield is the amount actually obtained, and percent yield is calculated as:
Formula:
Percent yield is always less than or equal to 100%.
Summary Table: Mole Relationships
Name of Substance | Formula | Formula Weight (amu) | Molar Mass (g/mol) | Number and Kind of Particles in One Mole |
|---|---|---|---|---|
Atomic nitrogen | N | 14.0 | 14.0 | 6.02 × 1023 N atoms |
Molecular nitrogen | N2 | 28.0 | 28.0 | 6.02 × 1023 N2 molecules |
Silver | Ag | 107.9 | 107.9 | 6.02 × 1023 Ag atoms |
Silver ions | Ag+ | 107.9 | 107.9 | 6.02 × 1023 Ag+ ions |
Barium chloride | BaCl2 | 208.2 | 208.2 | 6.02 × 1023 BaCl2 formula units, 6.02 × 1023 Ba2+ ions, 2 × 6.02 × 1023 Cl- ions |
