 Back
BackStoichiometry, Chemical Equations, and Quantitative Analysis in General Chemistry
Study Guide - Smart Notes

Chemical Equations and Stoichiometry
Writing and Balancing Chemical Equations
Chemical equations provide a concise description of chemical reactions, showing the reactants and products involved. Each equation must be balanced to reflect the conservation of mass, meaning the number of each type of atom is the same on both sides of the equation. The numbers in front of chemical formulas are called stoichiometric coefficients, indicating the relative number of molecules or moles involved.
Key Point 1: Atoms are neither created nor destroyed in a chemical reaction; the equation must be balanced.
Key Point 2: Stoichiometric coefficients represent the relative amounts of reactants and products.
Key Point 3: State symbols indicate the physical state: (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous solution.
Example: The reaction of hydrogen and oxygen to form water:
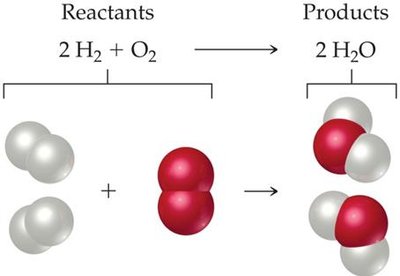
Additional info: Balancing equations involves adjusting coefficients, never subscripts, and often starts with the molecule containing the most atoms.
Types of Chemical Reactions
Chemical reactions can be classified into several types based on their patterns:
Combination: Two or more substances combine to form one product. Example:
Decomposition: One substance breaks down into two or more products. Example:
Combustion: Hydrocarbons react with oxygen to produce carbon dioxide and water. Example:
The Mole and Avogadro's Number
Definition and Use of the Mole
The mole is a fundamental unit in chemistry for counting particles. One mole contains entities (Avogadro's number), whether atoms, molecules, or ions. This allows chemists to relate mass, number of particles, and chemical reactions quantitatively.
Key Point 1: 1 mole of 12C atoms = atoms = 12 g of 12C.
Key Point 2: Molar mass (MM) is the mass in grams of 1 mole of a substance, numerically equal to its atomic weight in amu but with units of g/mol.
Example: What is the mass of 1.25 mol of Fe? Use Fe's molar mass:
Percent Composition and Empirical Formulas
Percent Composition from Chemical Formulas
The percent composition of a compound describes the mass percentage of each element present. It is calculated by comparing the mass of each element to the total mass of the compound.
Formula:
Example: Calculate the % mass of each element in dimethylsulfoxide (C2H6SO).
Empirical Formula Determination
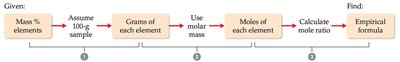
The empirical formula represents the simplest whole-number ratio of elements in a compound. It is often determined from percent composition data.
Key Point 1: Assume a 100 g sample so percent values become gram values.
Key Point 2: Convert grams to moles using atomic masses.
Key Point 3: Divide each mole value by the smallest to get the ratio.
Key Point 4: If ratios are not whole numbers, multiply by an integer to obtain whole numbers.
Example: Find the empirical formula for a compound with 61.31% C, 5.14% H, 10.21% N, and 23.33% O.
Additional info: The molecular formula is always a whole-number multiple of the empirical formula.
Combustion Analysis
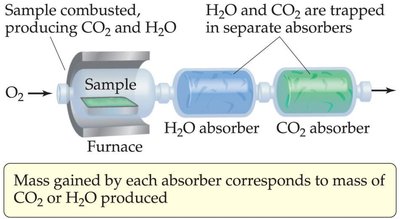
Empirical formulas are often determined by combustion analysis, where a sample containing C, H, and O is combusted in excess oxygen, producing CO2 and H2O. The masses of these products are measured to deduce the original composition.
Key Point 1: Mass of carbon is found from the mass of CO2 produced.
Key Point 2: Mass of hydrogen is found from the mass of H2O produced.
Key Point 3: Mass of oxygen is found by subtracting the masses of C and H from the total sample mass.
Stoichiometry: Quantitative Relationships in Reactions
Stoichiometric Calculations
Balanced chemical equations provide molar ratios for quantitative calculations. Stoichiometry allows chemists to predict the amounts of products and reactants involved in a reaction.
Key Point 1: Use molar ratios from the balanced equation to relate reactants and products.
Key Point 2: Convert masses to moles using molar mass, then use stoichiometric coefficients to find moles of other substances.
Example: How many grams of O2 can be produced from 4.5 g of KClO3? Use the equation:

Limiting Reactant and Yield
The limiting reactant is the reactant that is completely consumed first, thus determining the maximum amount of product formed. The theoretical yield is the maximum possible product, while the actual yield is what is obtained experimentally. Percent yield is calculated as:
Formula:
Example: If a reaction theoretically produces 8.52 g of product but only 6.27 g is obtained, percent yield is
Additional info: The limiting reactant is identified by comparing the amount of product each reactant can produce; the smallest amount determines the limiting reactant.
