Back
BackStoichiometry, Empirical and Molecular Formulas, and Reactions in Aqueous Solution
Study Guide - Smart Notes

Stoichiometry and Chemical Reactions
Limiting Reactants, Theoretical Yield, Actual Yield, and Percent Yield
Understanding the quantitative relationships in chemical reactions is essential for predicting product formation and optimizing laboratory procedures.
Limiting Reactant: The reactant that is completely consumed in a reaction, thus determining the maximum amount of product formed.
Theoretical Yield: The maximum amount of product that can be produced from a given amount of reactants, calculated using stoichiometry and considering the limiting reactant.
Actual Yield: The amount of product actually obtained from a reaction, often less than the theoretical yield due to experimental losses.
Percent Yield: A measure of reaction efficiency, calculated as:
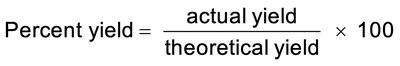
Stoichiometric Calculations
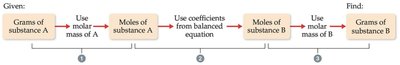
Stoichiometry allows chemists to relate quantities of reactants and products using balanced chemical equations.
Coefficients in balanced equations represent the number of moles (macro scale) or molecules (atomic scale).
To convert between grams and moles, use molar mass.
To relate reactants to products, use stoichiometric coefficients.

Example calculation:
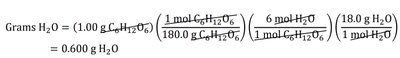
Limiting Reactant Determination
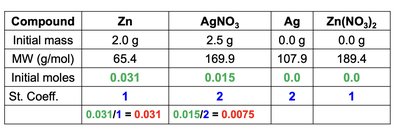
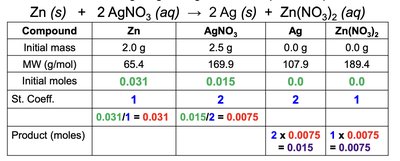
To identify the limiting reactant:
Convert masses of reactants to moles.
Divide moles by stoichiometric coefficients.
The smallest value indicates the limiting reactant.
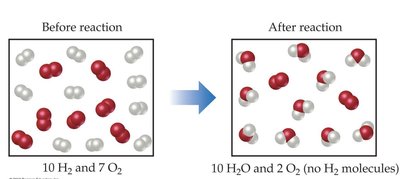
Example: Calculating product mass and percent yield.
Empirical and Molecular Formulas
Determining Empirical Formulas
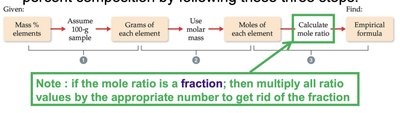
The empirical formula represents the simplest whole-number ratio of atoms in a compound. It can be determined from percent composition data.
Assume a 100 g sample for ease of calculation.
Convert mass of each element to moles using molar mass.
Divide each mole value by the smallest number of moles to obtain ratios.
If ratios are not whole numbers, multiply by an appropriate factor to eliminate fractions.

Example: Vitamin C empirical formula calculation.
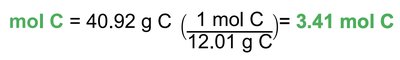



For nitrogen oxide:

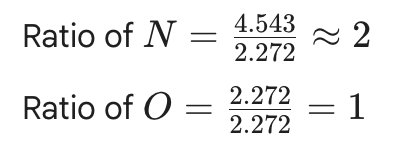
Determining Molecular Formulas
The molecular formula gives the actual number of atoms in a molecule. It is determined by comparing the empirical formula mass to the molar mass of the compound.
Calculate the empirical formula mass.
Divide the molar mass by the empirical formula mass to find the whole-number multiple.
Multiply the empirical formula by this multiple to obtain the molecular formula.

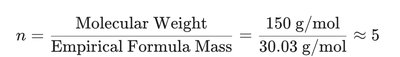
Combustion Analysis
Combustion analysis is a method used to determine empirical formulas, especially for compounds containing C, H, and O.
A sample is combusted in excess oxygen, producing CO2 and H2O.
CO2 and H2O are absorbed and measured to determine the original amounts of C and H.
O is determined by difference.
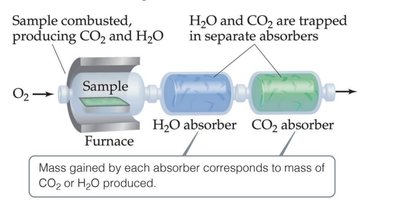
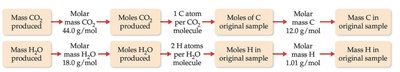
Example: Isopropyl alcohol combustion analysis.
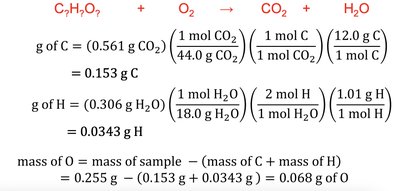


Reactions in Aqueous Solution
Terminology and Types of Solutions
Many chemical reactions occur in aqueous solution, where water acts as the solvent.
Solution: Homogeneous mixture of two or more substances.
Aqueous Solution: Water is the solvent.
Saturated Solution: Maximum solute dissolved; no more will dissolve.
Electrolyte: Dissociates into ions in water.
Nonelectrolyte: Does not dissociate into ions.
Precipitation Reaction: Forms an insoluble product.
Acid: Increases H+ concentration.
Base: Increases OH- concentration.
Metathesis Reaction: Exchange of ions between reactants.
Solubility of Ionic Compounds
Solubility rules help predict whether an ionic compound will dissolve in water.
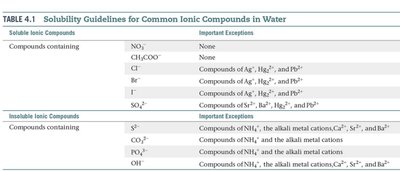
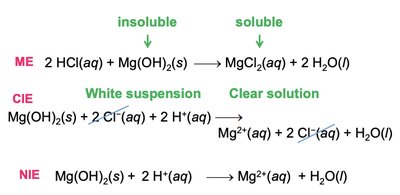
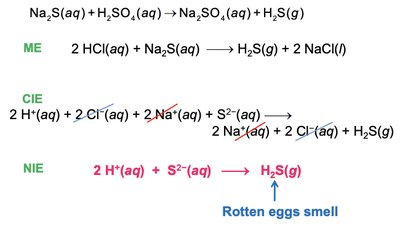
Precipitation and Metathesis Reactions
Precipitation reactions result in the formation of an insoluble product. Metathesis (exchange) reactions involve swapping ions between reactants.
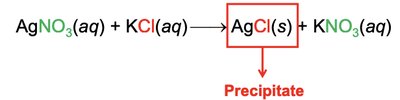
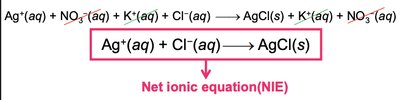
Writing Metathesis Reaction Equations
Three types of equations are used:
Molecular Equation (ME): Shows all reactants and products in their complete forms.
Complete Ionic Equation (CIE): Shows all strong electrolytes as ions.
Net Ionic Equation (NIE): Shows only the species that change during the reaction.

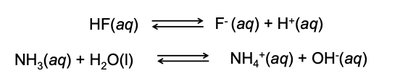
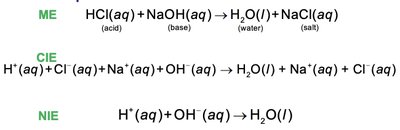
Acids and Bases
Acids and bases are classified by their ability to donate or accept protons.
Acids: Proton donors; increase H+ in solution.
Bases: Proton acceptors; increase OH- in solution.
Monoprotic Acids: Donate one proton (e.g., HCl, HNO3, CH3COOH).
Diprotic Acids: Donate two protons (e.g., H2SO4).
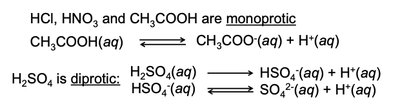

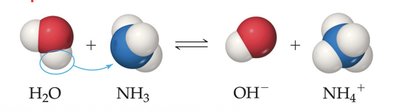
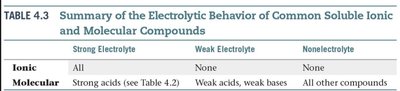
Strong and Weak Acids/Bases
Strong acids and bases dissociate completely in water, while weak acids and bases only partially dissociate.
Summary Table: Electrolytic Behavior
Electrolytic behavior depends on whether a compound is ionic or molecular, and whether it is a strong or weak acid/base.

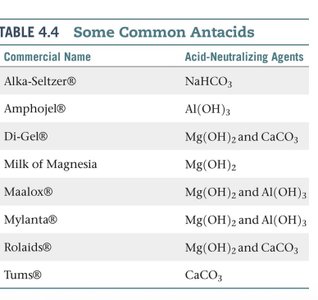
Application: Antacids
Neutralization reactions are used in antacids to relieve excess stomach acid.