 Back
BackStrong Acid-Strong Base Titrations: Principles, Calculations, and Laboratory Setup
Study Guide - Smart Notes

Strong Acid-Strong Base Titrations
Introduction to Titrations
Titrations are a fundamental analytical technique in chemistry used to determine the concentration of an unknown solution by reacting it with a solution of known concentration. In general chemistry, titrations can involve acid-base reactions or redox reactions, but here we focus on strong acid-strong base titrations.
Titration: A process where a solution of known concentration (the titrant) is gradually added to a solution of unknown concentration until the reaction reaches completion.
Equivalence Point: The stage in titration where the amount of titrant added exactly reacts with the analyte in the sample.
Applications: Used to determine molar mass, pKa/pKb, and to identify unknown species by calculating characteristic quantities.
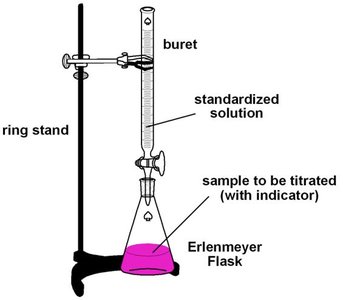
Laboratory Setup for Acid-Base Titrations
The typical laboratory setup for a titration involves a buret, ring stand, and an Erlenmeyer flask containing the sample and indicator. The titrant is added from the buret to the sample until the endpoint is reached.
Buret: Used to accurately dispense the titrant.
Erlenmeyer Flask: Contains the sample to be titrated, often with an indicator to signal the endpoint.
Indicator: A substance that changes color at or near the equivalence point.
Principles of Strong Acid-Strong Base Titrations
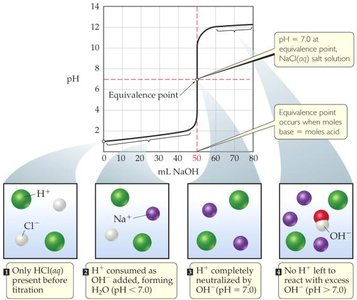
In a strong acid-strong base titration, both the acid and base dissociate completely in water. The titration curve shows how the pH changes as the titrant is added, with a sharp rise at the equivalence point.
Strong Acid: Completely dissociates in water (e.g., HCl, HNO3).
Strong Base: Completely dissociates in water (e.g., NaOH, KOH).
Equivalence Point: For strong acid-strong base titrations, the equivalence point occurs at pH = 7.
Calculating the Equivalence Point
The equivalence point is reached when the moles of acid equal the moles of base. The volume of titrant required can be calculated using the formula:
Formula: Where: = molarity of acid = volume of acid = molarity of base = volume of base
Example: To titrate 25.0 mL of 0.500 M HCl with 1.00 M NaOH, the required volume of NaOH is:
Titration Curves: Strong Acid vs. Strong Base
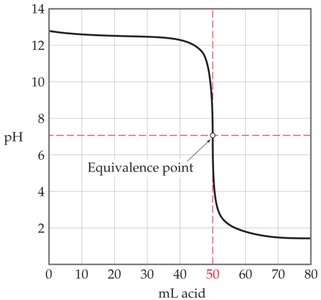
Titration curves graphically represent the change in pH as titrant is added. For strong acid-strong base titrations, the curve is characterized by a steep rise at the equivalence point.
Before Equivalence: Excess H+ or OH- determines pH.
At Equivalence: All acid and base are neutralized; pH = 7.
After Equivalence: Excess titrant determines pH.
Sample Calculations: pH at Different Points
Calculating pH at various stages of titration involves considering the amounts of acid and base present.
Before Equivalence: pH is determined by the excess strong acid or base.
At Equivalence: pH is neutral (pH = 7).
After Equivalence: pH is determined by the excess strong base or acid.
Example: Calculate pH when 24.9 mL and 25.1 mL of 0.100 M HNO3 are added to 25.0 mL of 0.100 M KOH. - For 24.9 mL: Slight excess of KOH, pH > 7. - For 25.1 mL: Slight excess of HNO3, pH < 7.
Summary Table: Key Features of Strong Acid-Strong Base Titrations
Stage | pH | Species Present |
|---|---|---|
Before Equivalence | < 7 (acidic) or > 7 (basic) | Excess H+ or OH- |
At Equivalence | 7 (neutral) | NaCl (salt), water |
After Equivalence | > 7 (basic) or < 7 (acidic) | Excess titrant (OH- or H+) |
Additional info:
Strong acid-strong base titrations are a core topic in general chemistry, relevant to Chapter 17 and acid-base equilibria.
Understanding titration curves is essential for interpreting laboratory data and for exam questions involving pH calculations.
