 Back
BackStrong Acid-Strong Base Titrations: Principles, Calculations, and Laboratory Setup
Study Guide - Smart Notes

Strong Acid-Strong Base Titrations
Introduction to Titrations
Titrations are a fundamental analytical technique in general chemistry used to determine the concentration of an unknown solution by reacting it with a solution of known concentration. In strong acid-strong base titrations, a strong acid reacts with a strong base, resulting in a neutralization reaction. This process is commonly used to analyze unknown solutions and can be extended to other types of titrations, such as redox titrations.
Titration: A method for determining the concentration of a solute in a solution by adding a reagent of known concentration until the reaction is complete.
Strong Acid: An acid that completely dissociates in water (e.g., HCl, HNO3).
Strong Base: A base that completely dissociates in water (e.g., NaOH, KOH).
Equivalence Point: The point at which stoichiometrically equivalent amounts of acid and base have reacted.
Indicator: A substance used to visually signal the completion of the reaction, often by a color change.
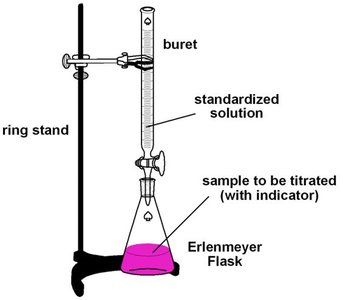
Laboratory Setup for Titration
The typical laboratory setup for a titration involves a buret, which dispenses the standardized solution, and an Erlenmeyer flask containing the sample to be titrated, often with an indicator added. The buret is mounted on a ring stand for stability and precision.
Buret: Used to accurately measure and deliver the titrant (standardized solution).
Erlenmeyer Flask: Contains the analyte (sample to be titrated) and indicator.
Ring Stand: Provides support for the buret.
Principles of Strong Acid-Strong Base Titrations
During a strong acid-strong base titration, the acid and base react according to the following neutralization equation:
Neutralization Reaction:
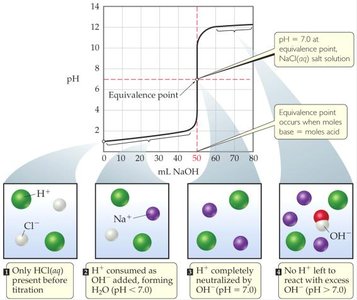
The titration curve shows how the pH changes as the titrant is added.
The equivalence point for a strong acid-strong base titration is typically at pH 7, since the resulting solution contains only water and a neutral salt.
Titration Curves and Equivalence Point
Titration curves graphically represent the change in pH as a function of the volume of titrant added. For strong acid-strong base titrations, the curve is characterized by a sharp rise in pH near the equivalence point.
Before titration: Only acid (e.g., HCl) is present.
During titration: Base (e.g., NaOH) is added, neutralizing the acid.
At equivalence point: All acid is neutralized; pH = 7.
After equivalence point: Excess base increases pH above 7.
Calculations in Strong Acid-Strong Base Titrations
Quantitative analysis in titrations involves calculating the volume of titrant required to reach the equivalence point and determining the pH at various stages of the titration.
Volume at Equivalence Point: Use stoichiometry to determine the volume of titrant needed.
Formula: , where and are the molarity and volume of the acid, and and are those of the base.
Example: To titrate 25.0 mL of 0.500 M HCl with 1.00 M NaOH, the required volume of NaOH is:
pH Calculations: Before, at, and after the equivalence point, pH is calculated based on the concentrations of acid and base remaining.
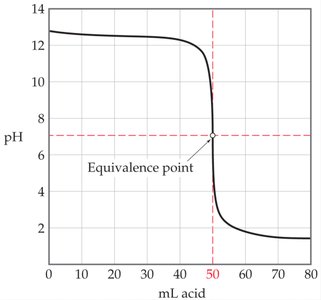
Titration Curve for Strong Base-Strong Acid Titration
When a strong base is titrated with a strong acid, the titration curve is similar but inverted, with a sharp drop in pH at the equivalence point.
Initial solution: Contains only base (e.g., KOH).
Acid is added: Neutralizes the base.
Equivalence point: pH drops to 7.
Excess acid: pH falls below 7.
Applications and Importance
Strong acid-strong base titrations are widely used in laboratories to determine unknown concentrations, analyze purity, and study reaction stoichiometry. The method is precise and forms the basis for many quantitative analyses in chemistry.
Used in quality control, environmental analysis, and research.
Can be extended to weak acid/base titrations and redox titrations.
Summary Table: Key Features of Strong Acid-Strong Base Titrations
Feature | Description |
|---|---|
Reactants | Strong acid and strong base |
Neutralization Reaction | |
Equivalence Point pH | 7 (neutral) |
Titration Curve | Sharp change in pH near equivalence point |
Calculation Formula |
