 Back
BackThe Periodic Table of the Elements: Classification and Structure
Study Guide - Smart Notes

Periodic Properties of the Elements
The Periodic Table: Structure and Classification
The periodic table is a systematic arrangement of the chemical elements, organized by increasing atomic number and recurring chemical properties. It is a foundational tool in general chemistry, allowing for the prediction of element behavior and relationships.
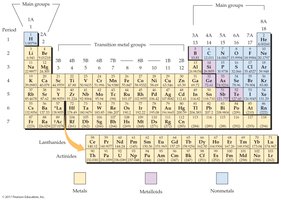
Periods: Horizontal rows on the periodic table, numbered from 1 to 7. Each period corresponds to the filling of a principal energy level (shell) of electrons.
Groups (Families): Vertical columns, numbered 1A to 8A (main groups) and 1B to 8B (transition metals). Elements in the same group have similar chemical properties due to similar valence electron configurations.
Main Group Elements: Elements in groups 1A–8A (except hydrogen and helium), which include the s- and p-block elements.
Transition Metals: Elements in the d-block (groups 3B–2B), characterized by partially filled d orbitals.
Lanthanides and Actinides: The two rows below the main table, representing the f-block elements. Lanthanides are elements 57–71, and actinides are elements 89–103.
Classification of Elements
Elements are broadly classified into three categories based on their physical and chemical properties:
Metals: Shown in yellow on the table. Metals are typically shiny, malleable, ductile, and good conductors of heat and electricity. Most elements are metals.
Metalloids: Shown in purple. Metalloids have properties intermediate between metals and nonmetals. They are often semiconductors and include elements such as Boron (B), Silicon (Si), and Arsenic (As).
Nonmetals: Shown in blue. Nonmetals are generally poor conductors of heat and electricity and are not malleable or ductile. Examples include Oxygen (O), Nitrogen (N), and Chlorine (Cl).
Key Features and Trends
Staircase Line: The zigzag line on the table separates metals (to the left and below) from nonmetals (to the right and above). Metalloids border this line.
Group Names:
Group 1A: Alkali metals
Group 2A: Alkaline earth metals
Group 7A: Halogens
Group 8A: Noble gases
Element Symbols: Each element is represented by a one- or two-letter symbol, with the first letter always capitalized.
Example: Classification of Silicon
Silicon (Si): Located along the staircase line, silicon is classified as a metalloid. It exhibits properties of both metals and nonmetals and is widely used in the electronics industry as a semiconductor.
Summary Table: Classification of Elements
Category | Location on Table | Key Properties | Examples |
|---|---|---|---|
Metals | Left and center (yellow) | Shiny, malleable, conductive | Fe, Cu, Na |
Metalloids | Along staircase (purple) | Intermediate properties, semiconductors | B, Si, As |
Nonmetals | Right (blue) | Dull, brittle, poor conductors | O, N, Cl |
Additional info: The periodic table is periodically updated as new elements are discovered and named. The arrangement reflects recurring (periodic) trends in element properties, such as atomic radius, ionization energy, and electronegativity, which are explored in detail in later chapters.
