 Back
BackThermal Energy Transfers and Thermal Equilibrium
Study Guide - Smart Notes

Ch. 5: Thermochemistry
Thermal Energy Transfers
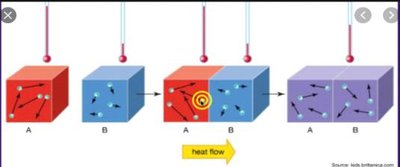
Thermal energy transfer is a fundamental concept in thermochemistry, describing how heat moves from one object to another. When two objects at different temperatures come into contact, thermal energy flows from the hotter object to the cooler one until both reach the same temperature, a state known as thermal equilibrium.
Direction of Heat Flow: Heat always flows from the object with higher temperature to the object with lower temperature.
Particle Motion: At the atomic and molecular level, particles in the hotter object move more rapidly. When in contact, energy is transferred through collisions, increasing the motion of particles in the cooler object.
Thermal Equilibrium: When both objects reach the same temperature, the net flow of thermal energy stops.
Quantitative Models: The amount of heat transferred can be calculated using the heat capacity and mass of the substances involved.
Mathematical Model of Thermal Equilibrium
To analyze thermal energy transfer quantitatively, we use mathematical models based on the principle of conservation of energy. The heat lost by the hotter object equals the heat gained by the cooler object.
Heat Released (q): The energy released by the hot object is given by:
Heat Absorbed (q): The energy absorbed by the cold object is:
Thermal Equilibrium Condition: At equilibrium, the heat released equals the heat absorbed (but with opposite signs):

Example: Calculating Final Temperature
Consider mixing 175 grams of hot aluminum (100°C) with 40.0 mL of ice-cold water (0.0°C) in an insulated cup. The heat capacity for aluminum is . To find the final temperature, set up the energy expressions for both substances and solve for the unknown temperature.
Step 1: Write the heat released by aluminum:
Step 2: Write the heat absorbed by water:
Step 3: Set and solve for .
Example Calculation: Suppose you mix 90.0 grams of hot water (90.0°C) with 10.0 grams of cold water (10.0°C), using . Let be the final temperature.
Energy released by hot water:
Energy absorbed by cold water:
Set and solve for .
Applications: This model is used in calorimetry experiments, material science, and engineering to predict temperature changes and energy transfer in physical systems.
