 Back
BackThermochemistry and Calorimetry: Chapter 7 Study Notes
Study Guide - Smart Notes

Thermodynamics and Internal Energy
Systems, Surroundings, and Energy Transfer
Thermodynamics studies energy changes in chemical reactions and physical processes. The system is the part under study, while the surroundings are everything else. The universe is the sum of system and surroundings.
Energy Conservation: The total energy lost by the system equals the energy gained by the surroundings, and vice versa.
Internal Energy Change: where q is heat and w is work.
Sign Conventions:
Heat absorbed by the system: (endothermic)
Heat released by the system: (exothermic)
Work done by the system:
Work done on the system:
First Law of Thermodynamics: Energy cannot be created or destroyed; it is conserved.
Calculating Internal Energy and Heat Capacity
Internal Energy Calculations
To calculate the change in internal energy:
Formula:
Example: If a gas absorbs 5.973 kJ of heat and does 0.559 kJ of work, kJ
Specific Heat and Molar Heat Capacity
Specific heat () is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C. Molar heat capacity is the heat required to raise 1 mole by 1°C.
Formula:
Example: To heat 5.00 L of antifreeze from 22.2°C to 100.0°C, use the density to find mass, then apply the formula.
Pressure-Volume Work
Work Done by Expanding Gases
When a gas changes volume against an external pressure, it does pressure-volume work.
Formula:
Conversion:
Example: Inflating a balloon from 2.0 L to 3.8 L at 0.00500 atm: atm·L
Calorimetry: Measuring Energy Changes
Constant Volume Calorimetry (Bomb Calorimeter)
Bomb calorimetry measures energy changes at constant volume. All energy from the reaction is converted to heat because .
Formula:
Energy Conservation:
Units: Usually reported in kJ/mol for a balanced reaction.
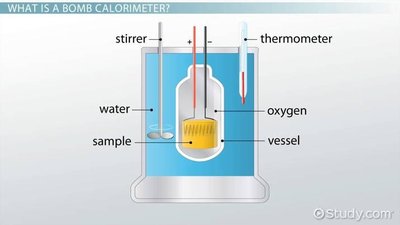
Example: Combustion of ethanol in a bomb calorimeter: If 25.0 g ethanol causes a temperature change of 12.4°C in a calorimeter with kJ/°C, kJ.
Constant Pressure Calorimetry (Coffee Cup Calorimeter)
Measures energy changes at constant pressure, typically for reactions in solution. The heat measured is the enthalpy change ().
Formula:
For negligible :
Example: Reaction of cadmium with HCl in a coffee-cup calorimeter: Use mass, specific heat, and temperature change to find .
Enthalpy and Chemical Reactions
Enthalpy () and Enthalpy Change ()
Enthalpy is a thermodynamic property representing the heat content of a system at constant pressure.
Formula:
Enthalpy Change:
State Function: Depends only on initial and final states, not the path.
Stoichiometry and Enthalpy
Enthalpy changes can be included in chemical equations and used for stoichiometric calculations.
Example: Combustion of propane: kJ/mol
Scaling: If the equation is multiplied by a factor, is multiplied by the same factor.
Reversing: If the reaction is reversed, the sign of changes.
Hess's Law
If a reaction can be expressed as the sum of steps, the overall is the sum of the $\Delta H$ values for each step.
Example: Formation of from and using given values for intermediate steps.
Bond Energies and Enthalpy
Bond Energy Calculations
Bond energies can be used to estimate for reactions.
Breaking bonds: Requires energy (endothermic)
Making bonds: Releases energy (exothermic)
Net : (energy to break bonds) (energy released in forming bonds)
Standard Enthalpy of Formation and Reaction
Standard Enthalpy of Formation ()
The energy required to form 1 mole of a compound from its elements in their standard states at 25°C.
Standard States:
Gases: pure gas at 1 atm
Liquids/Solids: pure substance at 1 atm and 25°C
Solutions: concentration of 1 M
Pure elements:
Standard Enthalpy of Reaction ()
Calculated from the enthalpy of formation values:
Formula:
Example: For , use given values to calculate .
Summary Table: Key Thermodynamic Quantities
Quantity | Definition | Formula |
|---|---|---|
Internal Energy () | Total energy of a system | |
Enthalpy () | Heat content at constant pressure | |
Work () | Energy transfer by volume change | |
Heat () | Energy transfer by temperature change | |
Standard Enthalpy of Formation () | Formation of 1 mole from elements | Varies by substance |
Standard Enthalpy of Reaction () | Reaction enthalpy from formation values |
