 Back
BackThermochemistry and Calorimetry: Step-by-Step Study Guidance
Study Guide - Smart Notes

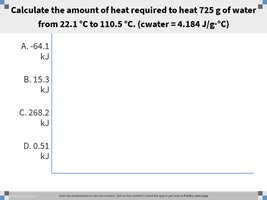
Q1. Calculate the amount of heat required to heat 725 g of water from 22.1 °C to 110.5 °C. (cwater = 4.184 J/g °C)
Background
Topic: Calculating Heat Transfer (q) in Calorimetry
This question tests your ability to use the specific heat formula to determine the amount of heat required to change the temperature of a given mass of water.
Key Terms and Formulas
Specific heat (): The amount of heat required to raise the temperature of 1 gram of a substance by 1 °C.
Heat (): The energy transferred due to temperature difference.
Formula:
= mass (in grams)
= specific heat (in J/g °C)
= change in temperature ()
Step-by-Step Guidance
Identify the known values: g, J/g °C, °C, °C.
Calculate the temperature change: .
Plug the values into the formula: .
Multiply the mass, specific heat, and temperature change together to find (in Joules).
Convert your answer from Joules to kilojoules by dividing by 1000.
Try solving on your own before revealing the answer!
Final Answer: 268.2 kJ
This is the amount of heat required to raise the temperature of the water as specified.
Q2. A 38.6 g piece of metal at 75.4 °C is dropped into 98.6 g of water originally at 29.5 °C. The final temperature of both the metal and water is 37.8 °C. What is the specific heat, c, of the metal? (cwater = 4.184 J/g °C)
Background
Topic: Calorimetry—Determining Specific Heat of a Metal
This question tests your understanding of heat transfer between substances and how to use calorimetry data to solve for an unknown specific heat.
Key Terms and Formulas
Heat lost by metal = Heat gained by water (assuming no heat loss to surroundings)
for both metal and water
Specific heat () is what you are solving for (for the metal)
Step-by-Step Guidance
Calculate for both the metal and the water.
Calculate using for water.
Set (because heat lost by metal = heat gained by water).
Write the equation for : .
Rearrange to solve for , but stop before plugging in the final numbers.

Try solving on your own before revealing the answer!
Final Answer: 2.4 J/g °C
By equating the heat lost by the metal to the heat gained by the water and solving for , you find the specific heat of the metal.
Q3. What is the change in internal energy for a system that releases 71 J of heat and does 3.1 × 10−2 kJ of work on its surroundings?
Background
Topic: First Law of Thermodynamics—Internal Energy Change
This question tests your understanding of how to calculate the change in internal energy () using heat () and work ().
Key Terms and Formulas
First Law of Thermodynamics:
= heat (Joules), negative if released by the system
= work (Joules), negative if done by the system on the surroundings
Convert all units to Joules before adding
Step-by-Step Guidance
Assign the correct sign to (released heat means $q$ is negative).
Convert the work value from kJ to J.
Assign the correct sign to (work done by the system is negative).
Plug the values into and simplify, but stop before the final calculation.

Try solving on your own before revealing the answer!
Final Answer: -102 J
Both and are negative, so the internal energy decreases by the sum of their magnitudes.
Q4. What mass of iron must be in a hot pack to provide -335 kJ of heat when the iron reacts with oxygen and is converted to iron(III) oxide according to the following thermochemical equation? 2 Fe(s) + 1.5 O2(g) → Fe2O3(s) ΔH° = -824.2 kJ
Background
Topic: Stoichiometry and Thermochemistry—Relating Heat to Mass
This question tests your ability to use a thermochemical equation to relate the amount of heat released to the mass of a reactant consumed.
Key Terms and Formulas
Thermochemical equation: relates moles of reactants/products to heat change ()
Molar mass of Fe: 55.85 g/mol
Proportionality: =
Step-by-Step Guidance
Set up a proportion relating the desired heat ( kJ) to the heat in the equation ( kJ for 2 mol Fe).
Solve for the number of moles of Fe needed to produce kJ.
Multiply the moles of Fe by the molar mass to get the mass of Fe required.
Stop before the final multiplication step.

Try solving on your own before revealing the answer!
Final Answer: 45.4 g
By setting up the proportion and solving for the mass, you find the amount of iron needed for the specified heat output.
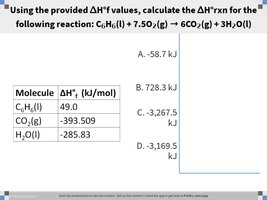
Q5. Using the provided ΔHf values, calculate the ΔHrxn for the following reaction: C6H6(l) + 7.5O2(g) → 6CO2(g) + 3H2O(l)
Background
Topic: Enthalpy of Reaction from Standard Enthalpies of Formation
This question tests your ability to use standard enthalpy of formation values to calculate the enthalpy change for a reaction.
Key Terms and Formulas
Standard enthalpy of formation (): Enthalpy change for forming 1 mol of a compound from its elements in their standard states.
Formula:
Step-by-Step Guidance
List the values for all reactants and products, including their coefficients.
Multiply each value by its coefficient in the balanced equation.
Add up the total for products and for reactants separately.
Subtract the sum for reactants from the sum for products, but stop before the final subtraction.
Try solving on your own before revealing the answer!
Final Answer: -3,267.5 kJ
By applying the formula and plugging in the values, you determine the enthalpy change for the reaction.
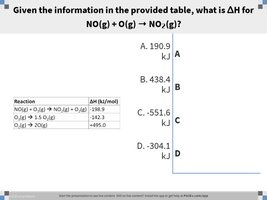
Q6. Given the information in the provided table, what is ΔH for NO(g) + O(g) → NO2(g)?
Background
Topic: Hess's Law—Combining Equations to Find Enthalpy Change
This question tests your ability to use Hess's Law and provided reactions to calculate the enthalpy change for a target reaction.
Key Terms and Formulas
Hess's Law: The enthalpy change for a reaction is the sum of the enthalpy changes for the steps that lead to the overall reaction.
Manipulate and combine the given equations to match the target equation.
Step-by-Step Guidance
Write out the target equation and the provided equations with their values.
Determine how to add, subtract, or reverse the given equations to obtain the target equation.
Adjust the values accordingly (reverse sign if reversing an equation, multiply if multiplying the equation).
Sum the adjusted values, but stop before the final addition.
Try solving on your own before revealing the answer!
Final Answer: -304.1 kJ
By manipulating the equations and summing the enthalpy changes, you find the enthalpy for the target reaction.
Q7. Calculate the bond energy of the O–F bond using the standard enthalpy of reaction and the bond energy data provided. OF2(g) + H2O(g) → O2(g) + 2HF(g) ΔH°= -318 kJ
Background
Topic: Bond Energies and Enthalpy of Reaction
This question tests your ability to use bond energies and the enthalpy of reaction to solve for an unknown bond energy.
Key Terms and Formulas
Bond energy: Energy required to break one mole of a bond in a molecule.
Formula:
Set up the equation for all bonds broken and formed in the reaction.
Step-by-Step Guidance
Write the Lewis structures for all reactants and products to identify all bonds broken and formed.
List the bond energies for all bonds except the O–F bond (which is unknown).
Set up the equation and plug in all known values.
Rearrange the equation to solve for the O–F bond energy, but stop before the final calculation.

Try solving on your own before revealing the answer!
Final Answer: 376 kJ
By setting up the bond energy equation and solving for the unknown, you find the O–F bond energy.
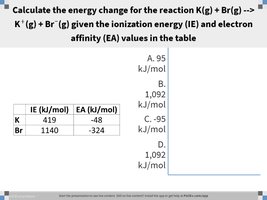
Q8. Calculate the energy change for the reaction K(g) + Br(g) → K+(g) + Br−(g) given the ionization energy (IE) and electron affinity (EA) values in the table.
Background
Topic: Ionization Energy, Electron Affinity, and Formation of Ions
This question tests your understanding of how to use ionization energy and electron affinity to calculate the energy change for forming ions from atoms.
Key Terms and Formulas
Ionization energy (IE): Energy required to remove an electron from a gaseous atom.
Electron affinity (EA): Energy change when an electron is added to a gaseous atom (usually negative).
Energy change for the reaction:
Step-by-Step Guidance
Identify the IE for K and the EA for Br from the table.
Plug the values into the formula (be careful with the sign of EA).
Add the values together, but stop before the final addition.
Try solving on your own before revealing the answer!
Final Answer: 95 kJ/mol
By adding the ionization energy and electron affinity, you find the energy change for the reaction.
