 Back
BackThermochemistry and Energy Changes in Chemical Processes
Study Guide - Smart Notes

Thermochemistry: Energy in Chemical Processes
Introduction to Thermochemistry
Thermochemistry is the study of energy changes, particularly heat, that occur during chemical reactions and physical changes of state. Understanding how energy is transferred between a system and its surroundings is fundamental in chemistry.
System: The part of the universe under study, typically the reactants and products in a chemical reaction.
Surroundings: Everything outside the system, including the container, air, and other external factors.
Energy Transfer: Energy can enter or leave the system as heat or work, but matter does not cross the system boundary in a closed system.
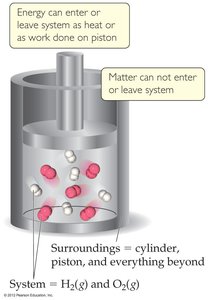
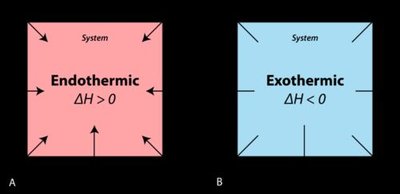
Endothermic and Exothermic Processes
Chemical reactions and physical changes can be classified based on whether they absorb or release heat.
Endothermic Process: Heat is absorbed by the system from the surroundings. The temperature of the surroundings decreases, and the process feels cold.
Exothermic Process: Heat is released by the system into the surroundings. The temperature of the surroundings increases, and the process feels hot.
Enthalpy Change (ΔH): Endothermic reactions have ΔH > 0, while exothermic reactions have ΔH < 0.

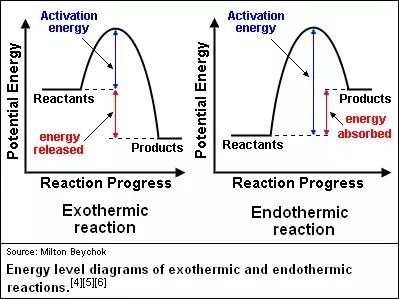
Energy Diagrams
Energy diagrams visually represent the energy changes during chemical reactions. They show the relative energies of reactants and products and the activation energy required for the reaction.
Exothermic Reaction: The products have lower energy than the reactants; energy is released.
Endothermic Reaction: The products have higher energy than the reactants; energy is absorbed.
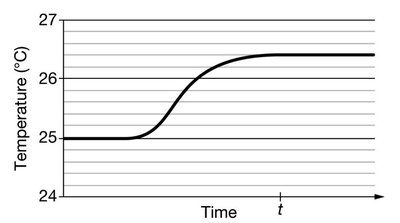
Heat Transfer and Thermal Equilibrium
Heat transfer occurs when two objects at different temperatures are brought into contact. Energy flows from the hotter object to the cooler one until thermal equilibrium is reached.
Thermal Equilibrium: The state at which two objects in contact reach the same temperature and no net heat flows between them.
Kinetic Energy: At equilibrium, the average kinetic energy of particles in both objects is equal.
Classification of Processes: Endothermic vs. Exothermic
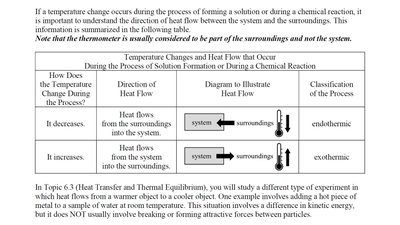
The following table summarizes how temperature changes and heat flow direction classify processes as endothermic or exothermic:
How Does the Temperature Change During the Process? | Direction of Heat Flow | Diagram to Illustrate Heat Flow | Classification of the Process |
|---|---|---|---|
It decreases. | Heat flows from the surroundings into the system. | system <-- surroundings | endothermic |
It increases. | Heat flows from the system into the surroundings. | system --> surroundings | exothermic |
Energy of Phase Changes
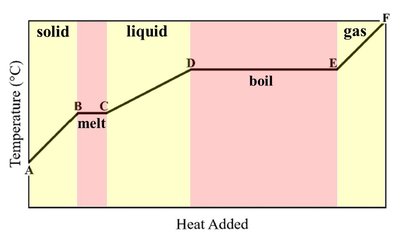
Phase changes such as melting, boiling, and condensation involve energy transfer. The direction of heat flow determines whether the process is endothermic or exothermic.
Melting (solid → liquid): Endothermic; energy is absorbed.
Boiling (liquid → gas): Endothermic; energy is absorbed.
Condensation (gas → liquid): Exothermic; energy is released.
Freezing (liquid → solid): Exothermic; energy is released.

Bond Energies and Chemical Reactions
Breaking chemical bonds requires energy (endothermic), while forming bonds releases energy (exothermic).
Bond Dissociation Energy: The energy required to break one mole of a specific bond in a molecule in the gas phase.
Example: Breaking an H–H bond requires 436 kJ/mol (endothermic), while forming an H–H bond releases 436 kJ/mol (exothermic).
Practice: Classifying Processes
Process | + or – |
|---|---|
K(s) → K(g) | + |
K(g) → K+(g) + e– | + |
Cl2(g) → 2 Cl(g) | + |
Cl(g) + e– → Cl–(g) | – |
K+(g) + Cl–(g) → KCl(s) | – |
Key Equations in Thermochemistry
Heat (q): Where m is mass, c is specific heat capacity, and ΔT is the temperature change.
First Law of Thermodynamics: Where ΔE is the change in internal energy, q is heat, and w is work.
Enthalpy Change (ΔH):
Summary Table: Endothermic vs. Exothermic
Process Type | Heat Flow | ΔH Sign | Temperature Change | Example |
|---|---|---|---|---|
Endothermic | Surroundings → System | + | Surroundings decrease | Melting ice, boiling water |
Exothermic | System → Surroundings | – | Surroundings increase | Combustion, freezing water |
Example Application
Heating Curve: When a solid is heated, its temperature rises until it reaches the melting point, where energy is used to break intermolecular forces (endothermic). The same principle applies to boiling and vaporization.
Calorimetry: Used to measure the amount of heat transferred in a chemical or physical process.
 Additional info: These notes cover AP Chemistry Unit 6 topics 6.1–6.5, which are directly relevant to general chemistry college courses, specifically thermochemistry, energy diagrams, heat transfer, calorimetry, and phase changes.
Additional info: These notes cover AP Chemistry Unit 6 topics 6.1–6.5, which are directly relevant to general chemistry college courses, specifically thermochemistry, energy diagrams, heat transfer, calorimetry, and phase changes.
