 Back
BackThermochemistry and the First Law of Thermodynamics: Study Notes
Study Guide - Smart Notes

Thermochemistry and the First Law of Thermodynamics
Introduction to Thermochemistry
Thermochemistry is a branch of chemistry that studies the heat energy involved in chemical reactions and physical changes. It is a subset of thermodynamics, which deals with the relationships between all forms of energy, including heat, work, and internal energy.
Thermodynamics: The study of energy transformations and the relationships between different forms of energy.
Thermochemistry: Focuses specifically on the heat absorbed or released during chemical reactions.
Example: The combustion of octane in air, which releases heat energy.

Energy, Work, and Heat
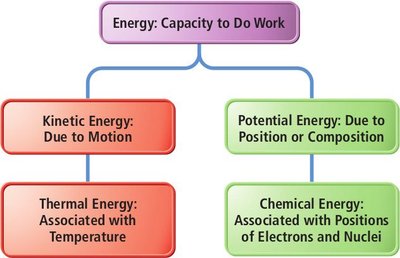
Energy is the capacity to do work. In chemistry, energy is categorized as kinetic (energy of motion) or potential (energy due to position or composition). Internal energy is the sum of all kinetic and potential energies of the particles in a system.
Kinetic Energy: Associated with the motion of particles (translation, rotation, vibration).
Potential Energy: Stored in molecular configurations, such as bond lengths and angles.
Internal Energy (E): The total energy (kinetic + potential) of a system; a state function.
Work (w): Energy transfer resulting from a force acting over a distance (macroscopic motion).
Heat (q): Energy transfer due to temperature difference (microscopic motion).
State Functions
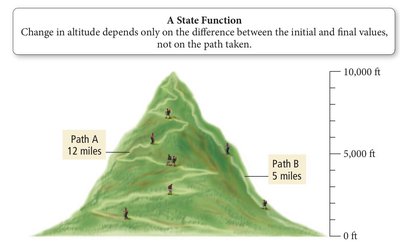
A state function is a property whose value depends only on the current state of the system, not on the path taken to reach that state. Internal energy is a state function.
Example: Altitude is a state function; the change in altitude depends only on the initial and final positions, not the path taken.
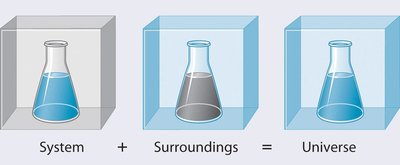
The System and Surroundings
In thermodynamics, the system is the part of the universe under study, while the surroundings are everything else. The total energy of the universe is the sum of the energy of the system and the surroundings.
Equation:
The First Law of Thermodynamics
The First Law of Thermodynamics states that energy cannot be created or destroyed, only transferred or converted from one form to another. The total energy of the universe remains constant.
Equation:
Change in Internal Energy:
Energy Transfer:
Sign Conventions: Energy transferred into the system is positive; energy transferred out is negative.

Calorimetry
Calorimetry is the experimental technique used to measure the heat exchanged in chemical or physical processes. The amount of heat is related to the temperature change of a substance.
Heat Capacity (C): The amount of heat required to change the temperature of a substance by 1 K.
Specific Heat Capacity (c): The amount of heat required to change the temperature of 1 gram of a substance by 1 K.
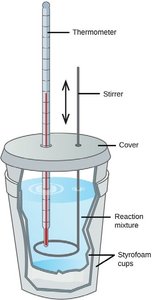
Constant Pressure Calorimetry: Measures heat at constant pressure, often using a coffee-cup calorimeter.
Constant Volume Calorimetry: Measures heat at constant volume, typically using a bomb calorimeter.
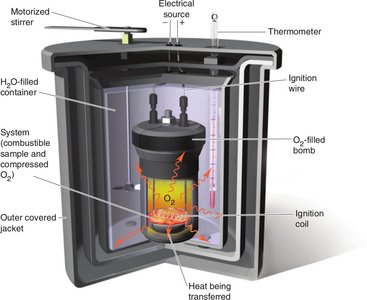
Enthalpy (H)
Enthalpy is a thermodynamic quantity defined as . The change in enthalpy () at constant pressure equals the heat absorbed or released by the system.
Equation:
Exothermic Process: Releases heat (); heat is a product.
Endothermic Process: Absorbs heat (); heat is a reactant.
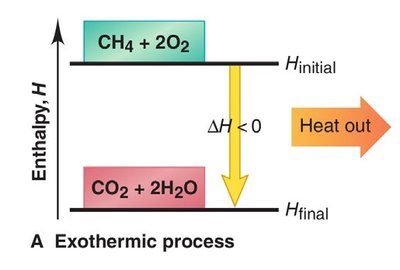
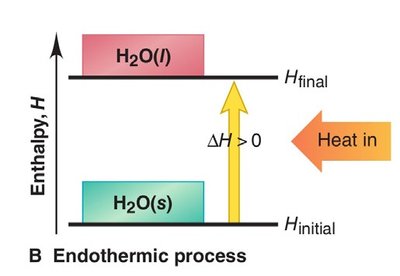
Thermochemical Equations and Stoichiometry
A thermochemical equation is a balanced chemical equation that includes the enthalpy change () for the reaction. The enthalpy change is proportional to the amount of substance reacted.
Example:
Stoichiometry: Use the balanced equation to convert between moles of reactant/product and heat absorbed or released.

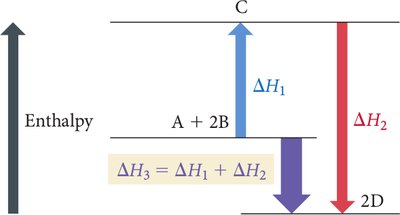
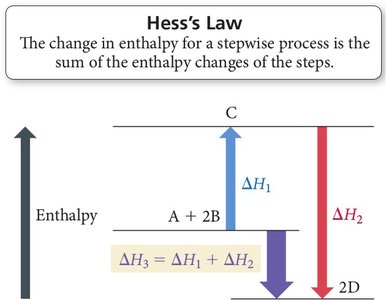
Hess’s Law
Hess’s Law states that the total enthalpy change for a reaction is the same, no matter how many steps the reaction is carried out in. This is because enthalpy is a state function.
Key Relationships:
If a reaction is multiplied by a factor, is multiplied by the same factor.
If a reaction is reversed, the sign of is reversed.
If reactions are added, their values are added.
Standard Enthalpy of Formation
The standard enthalpy of formation () is the enthalpy change for the formation of 1 mole of a compound from its elements in their standard states (most stable form at 1 atm and 298 K).
For an element in its standard state:
Equation for Standard Enthalpy of Reaction:
Summary Table: Sign Conventions for q, w, and ΔE
q | w | = | ΔE |
|---|---|---|---|
+ (heat absorbed) | + (work done on) | = | + (energy absorbed) |
+ (heat absorbed) | − (work done by) | = | Depends on the sizes of q and w |
− (heat released) | + (work done on) | = | Depends on the sizes of q and w |
− (heat released) | − (work done by) | = | − (energy released) |
Key Equations
Additional info: These notes cover the core concepts of thermochemistry, including energy transfer, calorimetry, enthalpy, Hess's Law, and standard enthalpy of formation, as relevant to a general chemistry curriculum.
