 Back
BackThermochemistry: Energy Changes in Chemical Reactions
Study Guide - Smart Notes

Thermochemistry
Section 9.1: Energy Changes in Chemical Reactions
Thermochemistry is the study of energy changes that occur during chemical reactions. Energy can be transferred in different ways, and understanding these transfers is essential for analyzing chemical processes.
Internal Energy (E): The total energy contained within a system, including both kinetic and potential energy.
Kinetic Energy: Energy due to motion of particles.
Potential Energy: Energy stored due to position or arrangement of particles.
System and Surroundings: The system is the part of the universe under study (e.g., reactants in a flask), while the surroundings include everything else (e.g., flask, solvent, room).

Example: In the methanol cannon demonstration, energy is transferred as both kinetic and potential energy within the system.
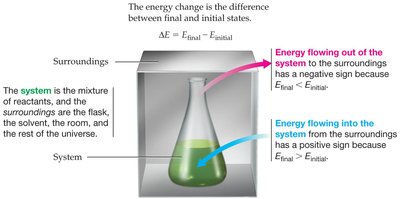
Key Point: The sign of ΔE depends on the direction of energy flow. Energy flowing out of the system is negative; energy flowing into the system is positive.
First Law of Thermodynamics
The First Law of Thermodynamics states that energy cannot be created or destroyed, only converted from one form to another. This principle is fundamental in chemistry.
System: The mixture of reactants.
Surroundings: The flask, solvent, room, and universe.
Energy Transfer: Energy is transferred as heat (q) or work (w).
Sign Conventions: Heat absorbed by the system (q > 0); heat released by the system (q < 0).
Energy Units:
1 calorie (cal) = 4.184 joules (J)
1 kcal = 1 Calorie (Cal)

Example: A typical glazed donut contains 250 Cal, which equals 104,600 J.
Section 9.3 and 9.4: Energy Transfer as Heat and Work
Energy transfer in chemical reactions occurs as heat or work. The total change in internal energy is the sum of heat and work:
Heat (q): Energy transferred due to temperature difference.
Work (w): Energy transferred when a force moves an object.
Example: If a system does 75 J of work on the surroundings and absorbs 25 J of heat, .
Classifying Energy Exchange
Energy exchange can be classified as heat or work, and the sign of the energy change is determined relative to the system.
Work, ΔE < 0: System loses energy by doing work.
Work, ΔE > 0: System gains energy by work done on it.
Heat, ΔE < 0: System loses energy as heat.
Heat, ΔE > 0: System gains energy as heat.


Example: Riding an elevator to the top of Willis Tower: you (the system) do work, ΔE < 0.

Example: An ice cube melts and cools your drink: the ice cube (system) absorbs heat, ΔE > 0.
Expansion Work
Expansion work occurs when a system changes volume against an external pressure. The equation for expansion work is:
P: External pressure
ΔV: Change in volume

Example: Combustion of methanol in a container with a movable piston results in an increase in volume, so work is done by the system (w < 0).

Calculating Work in Chemical Reactions
To calculate the energy transferred as work during a reaction:
1 L·atm = 101 J

Example: Combustion of 2 moles of methanol with a volume increase of 73.4 L against 1 atm:
Heat Transfer at Constant Pressure: Enthalpy Change (ΔH)
Enthalpy (H) is the heat content of a system at constant pressure. The change in enthalpy (ΔH) is used to quantify heat transfer in reactions.
Endothermic: System absorbs heat, ΔH > 0
Exothermic: System releases heat, ΔH < 0
Example: Combustion of propane:
Calculating the Amount of Heat Transfer
Given ΔH for a reaction, calculate the heat transferred for a given amount of reactant or product.
Use stoichiometry and molar mass to convert grams to moles.
Multiply moles by ΔH to find total heat transfer.

Example: 10.0 g of I2(g) turns into I2(s): (endothermic, heat absorbed).
Enthalpies of Chemical and Physical Changes
Enthalpy changes accompany both chemical and physical processes. The sign and magnitude of ΔH indicate whether heat is absorbed or released.


Example: Thermite reaction: (exothermic, heat released).
Calorimetry: Measuring Energy Changes
Calorimetry is the measurement of heat transfer in chemical reactions. There are two main types: bomb calorimetry (constant volume) and coffee cup calorimetry (constant pressure).
State Function: A property that depends only on the current state, not the path taken (e.g., internal energy, enthalpy).
Specific Heat Capacity (c): Energy required to raise the temperature of 1 g of a substance by 1°C.
Molar Heat Capacity (Cm): Energy required to raise the temperature of 1 mol of a substance by 1°C.
Example: Combustion and metabolism of glucose both release energy, demonstrating internal energy as a state function.
Heat Transfer Demonstrations
Heat transfer can be observed experimentally by heating metal rods and measuring their temperature changes.



Example: Four metal rods (Pb, Cu, Fe, Al) of equal mass are heated and placed in boiling water. Their specific heat capacities determine how much heat they transfer to the surroundings.
Calculating Heat Transfer
The amount of heat transferred is calculated using:
q: Heat (J)
m: Mass (g)
c: Specific heat capacity (J/g·°C)
ΔT: Temperature change (°C)

Example: A 55.0 g aluminum block absorbs 725 J of heat. Calculate the final temperature using .
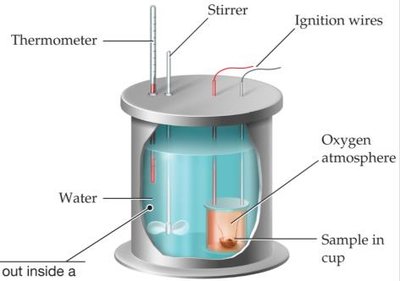
Bomb Calorimetry
Bomb calorimetry measures energy changes under constant volume. The energy change (ΔE) is measured, not enthalpy (ΔH).
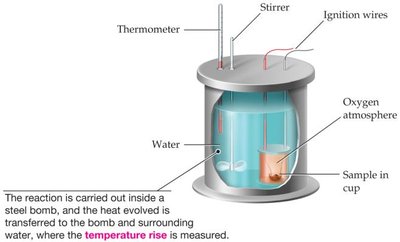

Example: Combustion of biodiesel in a bomb calorimeter: the temperature rise is used to calculate ΔE.
Coffee Cup Calorimetry
Coffee cup calorimetry measures enthalpy changes under constant pressure, typically for reactions in solution.


Example: Dissolving NH4NO3 in water and measuring the temperature change allows calculation of ΔH for the reaction.
Hess' Law
Hess' Law states that the overall enthalpy change for a reaction is equal to the sum of the enthalpy changes for individual steps.
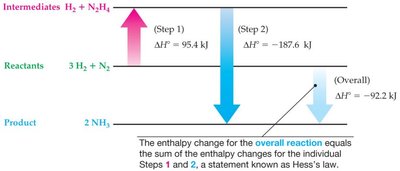
Example: The enthalpy change for the formation of NH3 from H2 and N2 can be calculated by summing the enthalpy changes of intermediate steps.
Summary Table: Types of Calorimetry
Type | Constant | Measured | Equation for qsys |
|---|---|---|---|
Bomb Calorimetry | Volume | ΔE | |
Coffee Cup Calorimetry | Pressure | ΔH |
Additional info: All equations are provided in LaTeX format for clarity and academic rigor.
