 Back
BackThermochemistry: Energy, Heat, and Enthalpy in Chemical Systems
Study Guide - Smart Notes

Thermochemistry
Introduction to Thermochemistry
Thermochemistry is the study of energy changes, particularly heat, that accompany chemical reactions and physical transformations. It is a fundamental topic in general chemistry, providing insight into how energy is transferred and conserved in chemical processes.
Energy and Its Forms
Definitions and Types of Energy
Energy: The capacity to do work or produce heat.
Kinetic Energy: Energy of motion, given by where m is mass and u is velocity.
Potential Energy: Energy due to position, condition, or composition.
Work (w): Related to force and distance, .
Heat (q): Transfer of thermal energy due to temperature difference.
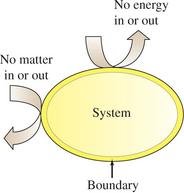
Systems and Surroundings
Types of Systems
Chemists define a system as the part of the universe under study, while everything else is the surroundings. Systems are classified based on their ability to exchange energy and matter:
Open System: Exchanges both matter and energy with surroundings.
Closed System: Exchanges energy but not matter.
Isolated System: Exchanges neither energy nor matter.

Heat and Calorimetry
Heat Capacity and Specific Heat
Heat capacity is the amount of heat required to change a system's temperature by one degree. Specific heat capacity refers to the heat required to raise the temperature of one gram of a substance by one degree Celsius.
Heat Capacity (C_p):
Molar Heat Capacity (C_{p,m}):
Specific Heat Capacity (c_p):

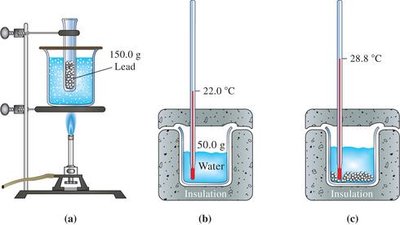
Experimental Determination of Specific Heat
Calorimetry is used to measure heat changes in chemical reactions or physical processes. The specific heat of a substance can be determined by measuring the temperature change when it is mixed with water of known temperature and heat capacity.

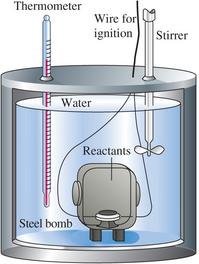
Calorimeters
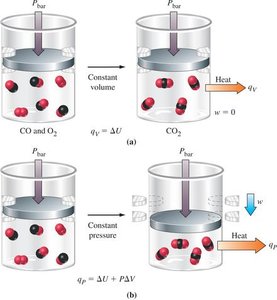
Bomb Calorimeter: Measures heat at constant volume (q_v = ΔU).
Coffee-Cup Calorimeter: Measures heat at constant pressure (q_p = ΔH).


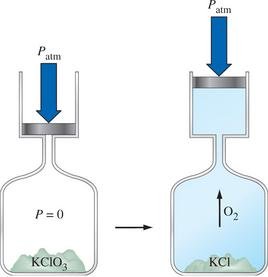
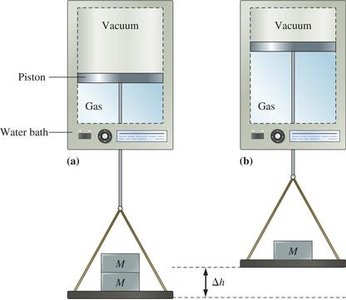
Work and Pressure-Volume Work
Pressure-Volume Work
When a gas expands or contracts against an external pressure, it does work on the surroundings or has work done on it. The work done is given by:

The First Law of Thermodynamics
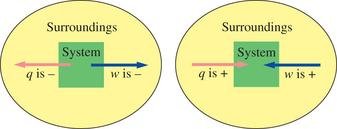
Statement and Mathematical Formulation
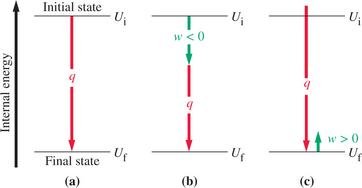
The first law states that energy cannot be created or destroyed, only transferred or converted. The change in internal energy (ΔU) of a system is the sum of heat (q) and work (w) exchanged with the surroundings:


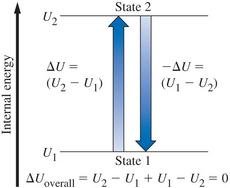
State Functions and Path Functions
Internal energy (U) is a state function, meaning its value depends only on the state of the system, not on how it got there. Heat and work are path functions, depending on the process taken.
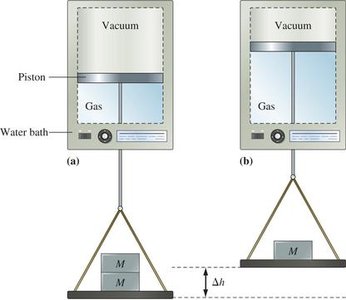
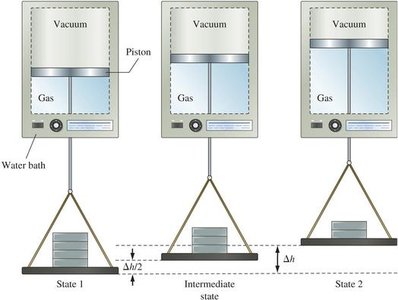
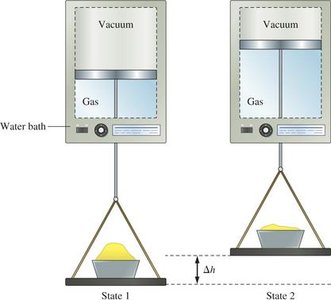
Enthalpy and Its Applications
Enthalpy (H)
Enthalpy is a state function defined as . At constant pressure, the heat exchanged is equal to the change in enthalpy:
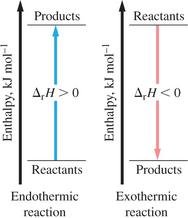
Enthalpy of Reaction and Phase Changes
Enthalpy of Reaction (ΔH_rxn): Heat change for a reaction at constant pressure.
Molar Enthalpy of Vaporization (ΔH_vap): Heat required to vaporize one mole of liquid.
Molar Enthalpy of Fusion (ΔH_fus): Heat required to melt one mole of solid.


Standard States and Standard Enthalpy Changes
The standard state of a substance is its pure form at 1 bar pressure and a specified temperature (usually 298.15 K). The standard enthalpy change (ΔH°) is the enthalpy change when all reactants and products are in their standard states.
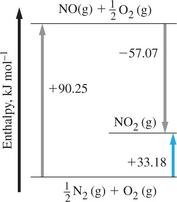
Hess's Law and Indirect Determination of Enthalpy
Hess's Law
Hess's Law states that the total enthalpy change for a reaction is the same, no matter how many steps the reaction is carried out in. This allows calculation of enthalpy changes for reactions that are difficult to measure directly.
If a process occurs in steps,

Standard Enthalpies of Formation
Definition and Use
The standard enthalpy of formation (ΔH°f) is the enthalpy change when one mole of a compound is formed from its elements in their standard states. For elements in their reference forms, ΔH°f = 0.
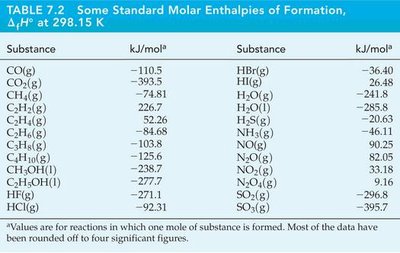
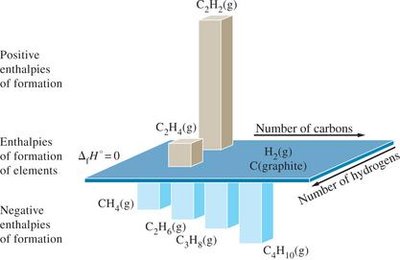
Summary Table: Standard Molar Enthalpies of Formation
Substance | ΔH°f (kJ/mol) | Substance | ΔH°f (kJ/mol) |
|---|---|---|---|
CO(g) | -110.5 | HBr(g) | -36.40 |
CO2(g) | -393.5 | HI(g) | 26.48 |
CH4(g) | -74.81 | H2O(g) | -241.8 |
C2H4(g) | 52.26 | H2O(l) | -285.8 |
C2H6(g) | -84.68 | NH3(g) | -45.94 |
C3H8(g) | -103.8 | NO(g) | 90.25 |
C4H10(g) | -125.6 | NO2(g) | 33.18 |
CH3OH(l) | -238.7 | SO2(g) | -296.8 |
HF(g) | -271.1 | SO3(g) | -395.7 |
HCl(g) | -92.31 |
Additional info: Table values are for reactions in which one mole of substance is formed. Most data are rounded to four significant figures.
