 Back
BackThermochemistry: Hess's Law and Standard Enthalpies of Formation
Study Guide - Smart Notes

Hess's Law
Definition and Application
Hess's Law states that if a chemical reaction is carried out in a series of steps, the overall enthalpy change (ΔH) for the reaction is equal to the sum of the enthalpy changes for the individual steps. This principle is a direct consequence of the fact that enthalpy is a state function, meaning its change depends only on the initial and final states, not on the path taken.
Key Point 1: The total enthalpy change for a reaction is independent of the reaction pathway.
Key Point 2: Hess's Law allows calculation of enthalpy changes for reactions that are difficult to measure directly by combining known enthalpy changes of related reactions.
Example: If you know the enthalpy changes for the formation and decomposition of compounds, you can sum or subtract these values to find the enthalpy change for a target reaction.
Standard Enthalpies of Formation (ΔH°f)
Definition and Importance
The standard enthalpy of formation, ΔH°f, is defined as the enthalpy change when one mole of a compound is formed from its constituent elements in their standard states under standard conditions (usually 298 K and 1 atm pressure).
Key Point 1: The standard enthalpy of formation for any element in its standard state is zero.
Key Point 2: ΔH°f values are used to calculate the enthalpy changes of reactions using the formula:
Example: The standard enthalpy of formation of water (H2O(l)) is -285.8 kJ/mol.
Calculating Enthalpy Changes Using Standard Enthalpies of Formation
Stepwise Calculation and Example
To calculate the standard enthalpy change for a reaction, use the standard enthalpies of formation for all reactants and products, applying the stoichiometric coefficients from the balanced equation.
Key Point 1: Multiply each ΔH°f value by its respective coefficient in the balanced equation.
Key Point 2: Subtract the sum for reactants from the sum for products.
Example: For the reaction 4 NH3(g) + 5 O2(g) → 4 NO(g) + 6 H2O(l):

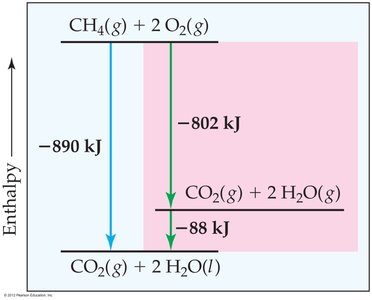
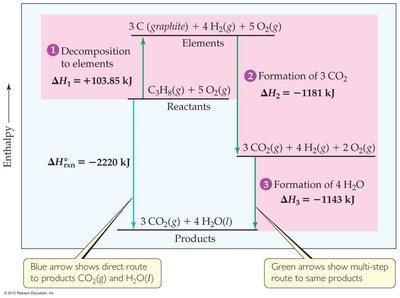
Combustion Reactions and Enthalpy Diagrams
Visualizing Enthalpy Changes
Enthalpy diagrams are useful for visualizing the energy changes during chemical reactions, such as combustion. They show the relative enthalpy levels of reactants, products, and any intermediate steps, illustrating how Hess's Law applies to multi-step reactions.
Key Point 1: The direct route and the sum of stepwise routes between the same reactants and products yield the same overall enthalpy change.
Key Point 2: These diagrams help in understanding the energy flow and the application of Hess's Law in practical scenarios.
Example: The combustion of propane (C3H8) can be analyzed by breaking it into formation and decomposition steps, then summing the enthalpy changes.
Worked Example: Calculating ΔH° for a Reaction
Using Tabulated Data
Given the following reaction:
CaCO3(s) → CaO(s) + CO2(g)
To find ΔH° for the reaction, use the standard enthalpies of formation:
ΔH°f [CaCO3(s)] = -1207.0 kJ/mol
ΔH°f [CaO(s)] = -635.5 kJ/mol
ΔH°f [CO2(g)] = -393.7 kJ/mol

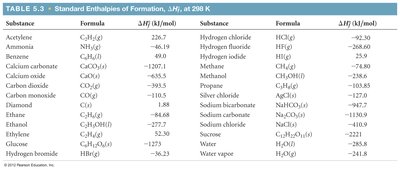
Summary Table: Standard Enthalpies of Formation
Reference Values for Calculations
The following table provides standard enthalpies of formation for commonly encountered substances, which are essential for solving thermochemistry problems:

Additional info: Standard enthalpy values are typically measured at 298 K (25°C) and 1 atm pressure. These values are critical for calculating reaction enthalpies in both laboratory and industrial settings.
