 Back
BackThermodynamics and Chemical Equilibrium: Predicting Chemical Change
Study Guide - Smart Notes

Thermodynamics and Chemical Equilibrium
Introduction to Predicting Chemical Change
The study of chemical change involves understanding the factors that determine whether a reaction will occur and to what extent. Thermodynamics provides a quantitative framework for predicting the directionality and extent of chemical reactions, focusing on energetic and entropic factors.
Energetic and Entropic Factors in Chemical Reactions
Energetic Factors: Enthalpy (ΔH)
- Enthalpy (ΔH) is the heat content of a system. The change in enthalpy during a reaction, ΔHrxn, indicates whether a reaction is exothermic (releases heat, ΔH < 0) or endothermic (absorbs heat, ΔH > 0). - Exothermic reactions are generally product-favored, while endothermic reactions may require additional factors to be product-favored.
Entropic Factors: Entropy (ΔS)
- Entropy (ΔS) measures the disorder or randomness in a system. The change in entropy, ΔSrxn, reflects the increase or decrease in disorder during a reaction. - Positive ΔS means increased disorder, often favoring product formation, especially at higher temperatures.
Temperature (T)
- Temperature influences the relative importance of enthalpy and entropy in determining reaction directionality. At high temperatures, entropic effects dominate; at low temperatures, energetic effects are more significant. 
Classification of Chemical Processes
Chemical processes can be classified based on the signs of ΔHrxn and ΔSrxn:
ΔSrxn > 0, ΔHrxn < 0: Always product-favored
ΔSrxn < 0, ΔHrxn > 0: Always reactant-favored
ΔSrxn > 0, ΔHrxn > 0 or ΔSrxn < 0, ΔHrxn < 0: Directionality depends on temperature
Second Law of Thermodynamics and Gibbs Free Energy
Second Law of Thermodynamics
The second law states that for thermodynamically favored processes in an isolated system, the total entropy change must be positive:
Gibbs Free Energy (ΔG)
Gibbs free energy combines enthalpy and entropy to predict reaction spontaneity: - ΔG < 0: Reaction is product-favored (spontaneous) - ΔG > 0: Reaction is reactant-favored (non-spontaneous)
Example: Formation of Water
Reaction: 2 H2(g) + O2(g) → 2 H2O(g)  - ΔHrxn is negative (exothermic) - ΔSrxn is negative (decrease in disorder) - ΔGrxn is negative at low temperatures, favoring product formation
- ΔHrxn is negative (exothermic) - ΔSrxn is negative (decrease in disorder) - ΔGrxn is negative at low temperatures, favoring product formation
Quantitative Analysis of Reaction Directionality
Calculating ΔG, ΔH, and ΔS
Standard thermodynamic data are used to calculate these values:
ΔHrxn = Σ ΔHf,products - Σ ΔHf,reactants
ΔSrxn = Σ Sproducts - Σ Sreactants
ΔGrxn = Σ ΔGf,products - Σ ΔGf,reactants
Example Table: Standard Thermodynamic Data
Substance | So (J/(mol K)) | ΔHof (kJ/mol) | ΔGof (kJ/mol) |
|---|---|---|---|
CO2(g) | 213.7 | -393.5 | -394.6 |
H2(g) | 130.7 | 0 | 0 |
CH4(g) | 186.3 | -74.8 | -50.8 |
Chemical Equilibrium and the Equilibrium Constant
Definition of Chemical Equilibrium
At equilibrium, the concentrations of reactants and products remain constant over time. 
Equilibrium Constant (K)
The equilibrium constant quantifies the extent of a reaction: - K > 1: Product-favored equilibrium - K < 1: Reactant-favored equilibrium
Relationship Between ΔG and K
The equilibrium constant is related to Gibbs free energy:
Temperature Dependence of K
The value of K changes with temperature, reflecting the balance between enthalpy and entropy: 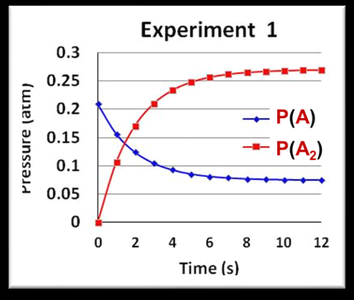
Summary Table: Directionality and Temperature
ΔHrxn | ΔSrxn | ΔGrxn | Directionality |
|---|---|---|---|
< 0 | > 0 | < 0 | Always product-favored |
> 0 | < 0 | > 0 | Always reactant-favored |
< 0 | < 0 | T-dependent | Product-favored at low T |
> 0 | > 0 | T-dependent | Product-favored at high T |
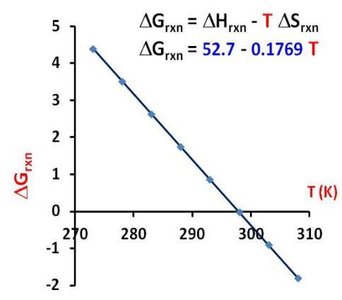
Practice Problem Example
Calculate ΔGrxn for N2O4(g) → 2 NO2(g) at 0°C (273 K)
ΔHrxn = 52.7 kJ
ΔSrxn = 176.9 J/K = 0.1769 kJ/K
ΔGrxn = 52.7 kJ - (273 K × 0.1769 kJ/K) = 52.7 kJ - 48.3 kJ = 4.4 kJ
Since ΔG > 0, the reaction is reactant-favored at 0°C.
Conclusion
Understanding the interplay between enthalpy, entropy, and temperature allows chemists to predict the directionality and extent of chemical reactions. The equilibrium constant provides a quantitative measure of reaction extent, and its relationship with Gibbs free energy is fundamental to chemical thermodynamics. Key Equations:
