 Back
BackThermodynamics and Kinetics in Chemical Reactions: Study Notes
Study Guide - Smart Notes

Chemical Reactions
Reactants and Products
Chemical reactions involve the transformation of reactants into products. The process is governed by the rearrangement of atoms and the breaking and formation of chemical bonds.
Reactants: The starting substances in a chemical reaction.
Products: The substances formed as a result of the reaction.
Example: Combustion of methane: CH4 + 2 O2 → CO2 + 2 H2O
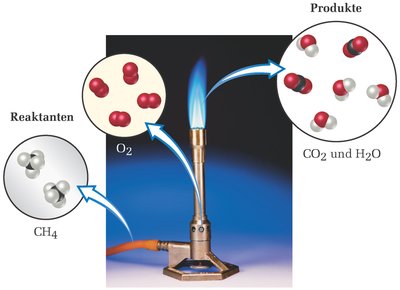

Thermodynamics vs. Kinetics
Fundamental Questions in Chemistry
Understanding chemical reactions requires addressing two key questions:
Thermodynamics: Does the reaction want to take place? This is determined by energy changes such as heat and work.
Kinetics: If the reaction takes place, how fast will it go? This is determined by the reaction rate and the pathway from reactants to products.
Thermodynamics studies the energy changes and feasibility of reactions, while kinetics studies the speed and mechanism of reactions.
Thermodynamics
Laws of Thermodynamics
The laws of thermodynamics govern the energy changes in chemical reactions:
0th Law: If two systems are in thermal equilibrium with a third, they are in equilibrium with each other.
1st Law: Energy cannot be created or destroyed; it can only be transferred or transformed. Internal energy changes according to the law of conservation of energy.
2nd Law: The entropy of a system increases in any natural thermodynamic process.
3rd Law: As temperature approaches absolute zero, the entropy of a system approaches a constant value.
Entropy (S)
Entropy is a measure of disorder or randomness in a system. It reflects the directionality of natural processes, with systems tending toward higher entropy.
Gibbs Free Energy and the Gibbs-Helmholtz Equation
The Gibbs free energy (G) determines whether a reaction is spontaneous. The Gibbs-Helmholtz equation relates enthalpy, entropy, and temperature:
ΔG: Change in Gibbs free energy
ΔH: Change in enthalpy
ΔS: Change in entropy
T: Temperature in Kelvin
The equation is:
Interpretation:
If : The process is exergonic and occurs spontaneously.
If : The process is endergonic and requires energy input.
If : The system is at equilibrium.
Kinetics
Activation Energy
Activation energy () is the minimum energy required for a reaction to occur. It represents the energy barrier that must be overcome for reactants to transform into products.
Reactions with high activation energy proceed slowly.
Reactions with low activation energy proceed quickly.

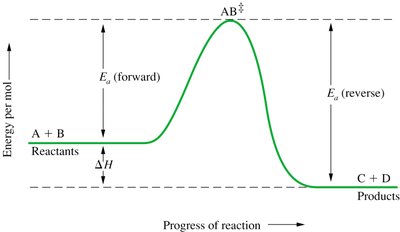
Reaction Pathways and Transition States
During a reaction, reactants must pass through a transition state (AB‡) before forming products. The energy profile of a reaction shows the activation energy and the enthalpy change ().
Effect of Orientation and Catalysts
The orientation of colliding particles affects the likelihood of reaction. Catalysts lower the activation energy, providing an alternative pathway and increasing the reaction rate without being consumed.


Catalysts: Example
Catalysts can dramatically increase the rate of reactions. For example, chlorine atoms catalyze the decomposition of ozone:
Without catalyst: O3 + O → 2 O2
With catalyst (Cl): Cl + O3 + O → ClO + O2 + O → Cl + O2 + O2

Thermodynamics vs. Kinetics: Real-World Examples
Hydrogen and Oxygen Reaction
Although the reaction 2 H2(g) + O2(g) → 2 H2O(l) is thermodynamically favored (large negative ), it does not occur spontaneously without a catalyst or energy input due to kinetic inhibition.
Diamond to Graphite Conversion
The conversion of diamond to graphite is thermodynamically favored (), but diamonds are metastable and do not convert rapidly due to kinetic barriers.
Metastable compounds are thermodynamically unstable but kinetically stable.
Summary Table: Thermodynamics vs. Kinetics
Aspect | Thermodynamics | Kinetics |
|---|---|---|
Focus | Energy changes, spontaneity | Reaction rate, mechanism |
Key Question | Does the reaction want to take place? | How fast will the reaction go? |
Key Quantity | ΔG (Gibbs free energy) | Activation energy (Ea) |
Example | Diamond to graphite conversion | Hydrogen and oxygen reaction |
