 Back
BackThermodynamics: Energy, Heat, Work, and Spontaneity in Chemical Systems
Study Guide - Smart Notes

Thermodynamics Overview
Introduction to Thermodynamics
Thermodynamics is the branch of chemistry that studies the transformation of energy, particularly how energy is transferred as heat and work in chemical systems. It provides the framework for understanding how and why chemical reactions occur, and how energy changes affect matter. 
Topic 4A: Work and Heat
System and Surroundings
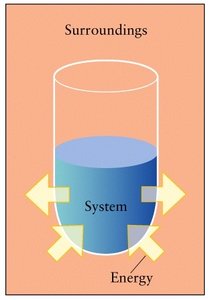
In thermodynamics, the universe is divided into the system (the part under study, such as a reaction mixture) and the surroundings (everything else). Energy can be transferred between the system and its surroundings as heat or work.
System: The portion of the universe chosen for study.
Surroundings: Everything outside the system.
Types of Systems
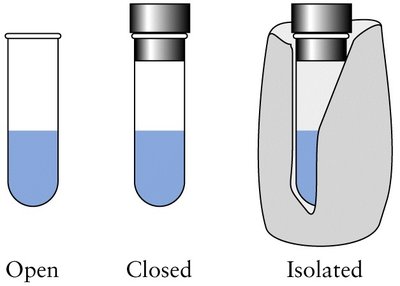
Systems are classified based on their ability to exchange energy and matter with their surroundings:
System | Energy Transfer | Matter Transfer |
|---|---|---|
Open | Yes | Yes |
Closed | Yes | No |
Isolated | No | No |
Energy: Kinetic and Potential
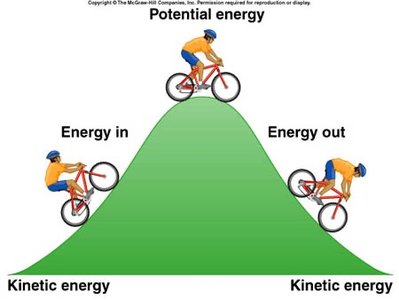
Kinetic Energy (KE): Energy of motion.
Potential Energy (PE): Energy due to position in a field of force. (gravitational)
Total Internal Energy (U):

Work and Heat
Work (w): Energy used to move an object.
Heat (q): Energy transferred due to temperature difference.
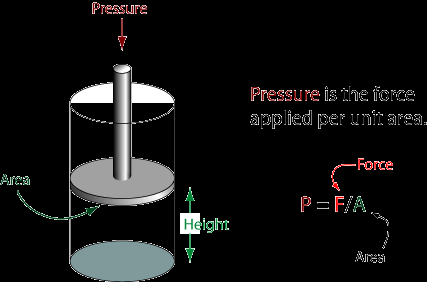
Pressure-Volume Work
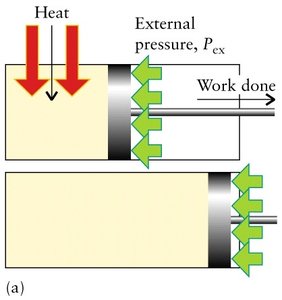
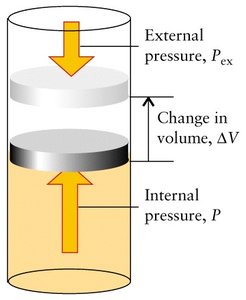
When a gas expands or contracts against an external pressure, work is done:
Expansion: , (system does work on surroundings)
Compression: , (surroundings do work on system)
Topic 4B: Internal Energy
First Law of Thermodynamics
The first law of thermodynamics states that energy cannot be created or destroyed, only transformed. The change in internal energy () of a system is given by:
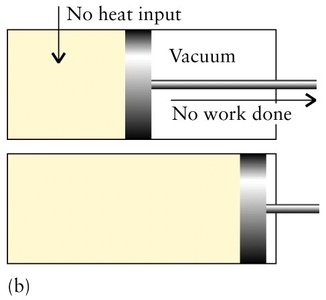
State and Path Functions
State functions: Depend only on the initial and final states (e.g., U, H, S, G).
Path functions: Depend on the process taken (e.g., q, w).
Topic 4C: Enthalpy
Definition and Measurement
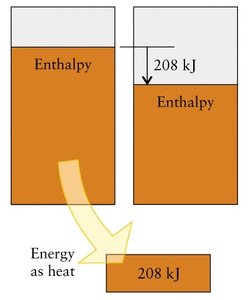
Enthalpy (H) is a state function defined as . The change in enthalpy () at constant pressure equals the heat transferred: .
Exothermic: (heat released)
Endothermic: (heat absorbed)
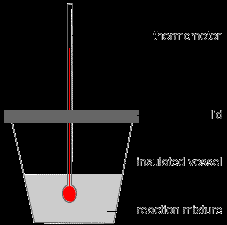
Topic 4D: Thermochemistry
Enthalpy of Reaction and Hess's Law
Enthalpy of reaction ():
Hess's Law: The enthalpy change for a reaction is the sum of the enthalpy changes for each step.
Standard Enthalpy of Formation
is the enthalpy change when 1 mole of a compound forms from its elements in their standard states.
For elements in their standard states, .
Topic 4E: Contributions to Enthalpy
Bond Enthalpy
Bond enthalpy: Energy required to break a chemical bond.
Bond breaking is endothermic; bond formation is exothermic.
Topic 4F: Entropy
Definition and Quantification
Entropy (S) is a measure of disorder or randomness. It is a state function and increases with temperature, volume, and phase changes (solid → liquid → gas).
(volume change)
(temperature change)
(phase change)
Second and Third Laws of Thermodynamics
Second Law: The entropy of an isolated system increases in any spontaneous process.
Third Law: The entropy of a perfect crystal at 0 K is zero.
Topic 4G: Molecular Interpretation of Entropy
Boltzmann Formula
Where is Boltzmann's constant and is the number of microstates.
Topic 4H: Absolute Entropies
Standard Molar Entropy
Standard molar entropy () is the entropy of 1 mole of a substance at 1 bar and 298 K.
Large, complex molecules have higher than small, simple ones.
Topic 4I: Global Changes in Entropy
Total Entropy Change
For a process at constant pressure:
A process is spontaneous if .
Topic 4J: Gibbs Free Energy
Definition and Spontaneity
Gibbs free energy (G) is a state function that predicts spontaneity at constant temperature and pressure.
If , the process is spontaneous.
If , the process is nonspontaneous.
If , the system is at equilibrium.
Temperature Dependence
Spontaneity depends on both and and the temperature.
At equilibrium:
Tables and Data
Specific and Molar Heat Capacities
Substance | Specific Heat Capacity (J/g·K) | Molar Heat Capacity (J/mol·K) |
|---|---|---|
Aluminum | 0.897 | 24.2 |
Graphite | 0.685 | 8.23 |
Iron | 0.449 | 25.1 |
Copper | 0.385 | 24.5 |
Gold | 0.129 | 25.6 |
Water (liquid) | 4.184 | 75.4 |
Water (ice) | 2.06 | 37.1 |
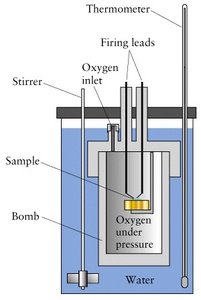
Varieties of Work
Type of Work | Equation | Units |
|---|---|---|
Expansion | Pa·m³ | |
Extension | N·m | |
Raising a weight | kg·m²·s⁻² | |
Electrical | V·C | |
Surface expansion | N·m⁻¹·m² |
Summary
Thermodynamics provides the essential principles for understanding energy changes in chemical reactions, the direction of spontaneous change, and the relationship between heat, work, and entropy. Mastery of these concepts is fundamental for further study in chemistry and related sciences.
