 Back
BackThermodynamics, Entropy, and Equilibrium: Step-by-Step General Chemistry Guidance
Study Guide - Smart Notes

Q1. Above what temperature does the following reaction become nonspontaneous?
FeO(s) + CO(g) → CO2(g) + Fe(s) ΔH = -11.0 kJ; ΔS = -17.4 J/K
Background
Topic: Thermodynamics – Spontaneity of Reactions
This question tests your understanding of how enthalpy (ΔH) and entropy (ΔS) affect the spontaneity of a reaction, using the Gibbs free energy equation.
Key formula:
= Gibbs free energy change
= enthalpy change (in kJ)
= entropy change (in J/K)
= temperature (in K)
Step-by-Step Guidance
Recall that a reaction is nonspontaneous when .
Set up the equation: and solve for the temperature where (the threshold between spontaneous and nonspontaneous).
Convert to J if needed, so units match with .
Rearrange the equation to solve for : .
Try solving on your own before revealing the answer!
Q2. Which of the following processes have a ΔS < 0?
Background
Topic: Entropy Changes in Physical Processes
This question tests your ability to recognize when entropy decreases (ΔS < 0) in phase changes or temperature changes.
Key Terms:
Entropy (): A measure of disorder or randomness in a system.
ΔS < 0: Indicates a decrease in entropy (system becomes more ordered).
Step-by-Step Guidance
Identify which processes involve a transition to a more ordered state (solidification, condensation, cooling).
Recall that freezing, condensation, and deposition (gas to solid) all decrease entropy.
Consider the effect of lowering temperature on a gas: less molecular motion, more order.
Try solving on your own before revealing the answer!
Q3. Consider the figure which shows ΔG° for a chemical process plotted against absolute temperature. What can you conclude?
Background
Topic: Gibbs Free Energy and Temperature Dependence
This question tests your ability to interpret a plot of ΔG° versus temperature and relate it to the signs of ΔH° and ΔS°.
Key formula:
Step-by-Step Guidance
Observe the slope of the line: If ΔG° increases with T, then is positive, so is negative.
Check the y-intercept: At , .
Determine the sign of from the y-intercept (positive or negative).
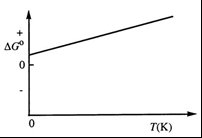
Try solving on your own before revealing the answer!
Q4. ΔG° for the reaction A(aq) + B(aq) ⇄ C(aq) is -4.95 kJ/mol at 25°C. If [A] = 0.30 M, [B] = 0.40 M, and [C] = 12.6 M, what will occur?
Background
Topic: Reaction Direction and Gibbs Free Energy
This question tests your ability to use ΔG° and concentrations to predict the direction of a reaction.
Key formula:
= reaction quotient, calculated from concentrations
= gas constant ()
= temperature in Kelvin
Step-by-Step Guidance
Calculate using the given concentrations.
Plug , , , and into the formula to find .
Determine whether is positive or negative to predict the direction of the reaction.
Try solving on your own before revealing the answer!
Q5. Of the following, which has the largest entropy?
Background
Topic: Entropy and States of Matter
This question tests your understanding of how entropy varies between different phases and substances.
Key Terms:
Entropy (): Higher in gases than liquids, higher in liquids than solids.
Step-by-Step Guidance
Recall the order: .
Compare the substances and their phases to determine which is most disordered.
Try solving on your own before revealing the answer!
Q6. Which of the following does not have a ΔG° value of zero?
Background
Topic: Standard Free Energy of Formation
This question tests your knowledge of which substances have a standard free energy of formation () of zero.
Key Terms:
Standard state: Most stable form of an element at 1 atm and 25°C.
for elements in their standard state.
Step-by-Step Guidance
Identify which substances are elements in their standard state.
Recall that only these have .
Try solving on your own before revealing the answer!
Q7. Which of the following statements is true?
Background
Topic: Laws of Thermodynamics and Entropy
This question tests your understanding of the second law of thermodynamics, entropy, and standard states.
Key Terms:
Second law: Entropy of the universe increases for spontaneous processes.
Standard entropy (): Absolute values can be measured.
Step-by-Step Guidance
Review each statement and compare it to thermodynamic principles.
Identify which statement accurately reflects the laws or properties discussed.
Try solving on your own before revealing the answer!
Q8. A container is filled with Ne gas. Which change increases the entropy of the system?
Background
Topic: Entropy and Molecular Motion
This question tests your understanding of how changes in volume, temperature, number of particles, and type of gas affect entropy.
Key Terms:
Entropy increases with more particles, higher temperature, larger volume, and heavier atoms.
Step-by-Step Guidance
Consider how each change affects the disorder or randomness of the system.
Recall that increasing any of these factors generally increases entropy.
Try solving on your own before revealing the answer!
Q9. Balance the following reactions:
Zn(s) + ReO4-(aq) → Re(s) + Zn2+(aq) in acidic solution CN-(aq) + Fe3+(aq) → CNO-(aq) + Fe2+(aq) in basic solution
Background
Topic: Redox Reactions and Balancing Equations
This question tests your ability to balance redox reactions in acidic and basic solutions.
Key Steps:
Assign oxidation states to each element.
Write half-reactions for oxidation and reduction.
Balance atoms and charges using electrons, H+, and OH- as appropriate.
Step-by-Step Guidance
Write the oxidation and reduction half-reactions for each equation.
Balance atoms other than H and O.
Balance O using H2O, H using H+ (acidic) or OH- (basic).
Try solving on your own before revealing the answer!
Q10. Calculate ΔG° (kJ) for the reaction CO(g) → C(s) + 1/2 O2(g) given:
CO2(g) → C(s) + O2(g), ΔG°rxn = +394.4 kJ CO(g) + 1/2 O2(g) → CO2(g), ΔG°rxn = -257.2 kJ
Background
Topic: Thermodynamic Cycles and Hess's Law
This question tests your ability to use Hess's Law to combine reactions and calculate ΔG° for a new reaction.
Key Steps:
Reverse and/or multiply reactions as needed to match the target reaction.
Add the ΔG° values accordingly.
Step-by-Step Guidance
Write the target reaction and compare it to the given reactions.
Manipulate the given reactions (reverse, multiply) to construct the target reaction.
Add the ΔG° values, considering sign changes for reversed reactions.
Try solving on your own before revealing the answer!
Q11. a. Determine the standard molar entropy change of vaporization (ΔSvap) for CH3OH(l) → CH3OH(g) at its boiling point.
Background
Topic: Phase Changes and Entropy
This question tests your ability to calculate entropy changes for phase transitions using enthalpy and temperature.
Key formula:
= enthalpy change (in J/mol)
= temperature (in K)
Step-by-Step Guidance
Convert enthalpy of vaporization from kJ/mol to J/mol.
Plug values into using the boiling point.
Try solving on your own before revealing the answer!
Q11b. Determine the standard molar entropy change of fusion (ΔSfus) for CH3OH(s) → CH3OH(l) at its freezing point.
Background
Topic: Phase Changes and Entropy
This question tests your ability to calculate entropy changes for fusion using enthalpy and temperature.
Key formula:
Step-by-Step Guidance
Convert enthalpy of fusion from kJ/mol to J/mol.
Plug values into using the freezing point.
Try solving on your own before revealing the answer!
Q11c. Explain why one of the values is higher than the other.
Background
Topic: Entropy Changes in Phase Transitions
This question tests your understanding of why vaporization generally has a larger entropy change than fusion.
Key Concepts:
Vaporization involves a greater increase in disorder than fusion.
Step-by-Step Guidance
Compare the physical changes: solid to liquid vs. liquid to gas.
Discuss the difference in molecular freedom and randomness.
Try solving on your own before revealing the answer!
Q12a. Determine the boiling point (in Kelvin) of benzene (C6H6).
Background
Topic: Phase Equilibria and Thermodynamics
This question tests your ability to use thermodynamic data to determine boiling points.
Key formula:
(at boiling point, )
Step-by-Step Guidance
Set at the boiling point.
Rearrange to using values for benzene.
Try solving on your own before revealing the answer!
Q12b. Determine the vapor pressure of benzene at 25°C.
Background
Topic: Vapor Pressure and Thermodynamics
This question tests your ability to use thermodynamic relationships to calculate vapor pressure.
Key formula:
Step-by-Step Guidance
Use for vaporization at 25°C.
Plug values into the formula and solve for .
Try solving on your own before revealing the answer!
Q12c. If the pressure was lower than your answer for part b, what would be the sign of ΔG? Explain.
Background
Topic: Thermodynamics of Phase Changes
This question tests your understanding of how vapor pressure and ΔG relate.
Key Concepts:
If , is negative, so is positive.
Step-by-Step Guidance
Explain mathematically using the formula.
Discuss the physical meaning: vaporization is nonspontaneous if pressure is below equilibrium.
Try solving on your own before revealing the answer!
Q13a. Calculate Ksp for the reaction AgBr(s) → Ag+(aq) + Br-(aq) at 25°C.
Background
Topic: Solubility Product and Thermodynamics
This question tests your ability to relate ΔG° to Ksp using thermodynamic equations.
Key formula:
Step-by-Step Guidance
Find ΔG° for the dissolution reaction from the provided data.
Plug values into and solve for .
Try solving on your own before revealing the answer!
Q13b. Determine the concentration (M) of Ag+(aq) at 25°C.
Background
Topic: Solubility Equilibria
This question tests your ability to use Ksp to find ion concentrations.
Key formula:
Step-by-Step Guidance
Assume equal concentrations of Ag+ and Br- for a 1:1 salt.
Set and solve for .
Try solving on your own before revealing the answer!
Q13c. Determine the temperature (K) necessary to make the concentration of both ions 0.010 M.
Background
Topic: Solubility and Temperature Dependence
This question tests your ability to relate solubility, Ksp, and temperature using thermodynamic equations.
Key formula:
Step-by-Step Guidance
Set .
Plug values into the formula and solve for .
Try solving on your own before revealing the answer!
