 Back
BackThermodynamics: Entropy, Free Energy, and Equilibrium – Guided Study Notes
Study Guide - Smart Notes

Q1. What does freezing water (liquid to solid) do to the entropy and enthalpy of the system, the entropy of the surroundings, and the entropy of the universe? Is the process spontaneous? Why?
Background
Topic: Thermodynamics – Entropy, Enthalpy, and Spontaneity
This question tests your understanding of how phase changes affect entropy (), enthalpy (), and spontaneity, as well as the relationship between the system, surroundings, and universe.
Key Terms and Formulas:
Entropy (): A measure of disorder or randomness in a system.
Enthalpy (): The heat content of a system at constant pressure.
Spontaneity: A process is spontaneous if it occurs without outside intervention, often determined by the sign of (Gibbs Free Energy).
Gibbs Free Energy:
Step-by-Step Guidance
Consider what happens to the entropy of the system when water freezes (liquid to solid). Does the system become more ordered or disordered?
Think about the enthalpy change () for freezing. Is heat released or absorbed by the system?
Analyze the entropy of the surroundings. If the system releases heat, what happens to the surroundings' entropy?
Combine the changes in entropy for the system and surroundings to determine the entropy change of the universe ().
Use the relationship between and spontaneity to discuss whether freezing is spontaneous under typical conditions, and what extra information (if any) you might need.
Try solving on your own before revealing the answer!
Q2. What is Gibbs Free Energy and why is it important?
Background
Topic: Gibbs Free Energy
This question is about understanding the definition and significance of Gibbs Free Energy in predicting the spontaneity of chemical processes.
Key Terms and Formulas:
Gibbs Free Energy (): A thermodynamic quantity that combines enthalpy and entropy to predict spontaneity.
Formula:
Step-by-Step Guidance
Recall the definition of Gibbs Free Energy and what each term in the formula represents.
Explain how the sign of determines whether a process is spontaneous (), nonspontaneous (), or at equilibrium ().
Discuss why Gibbs Free Energy is useful for chemists and how it relates to real-world chemical reactions.
Try solving on your own before revealing the answer!
Q3. What is the 3rd law of thermodynamics? Order the following from least to most entropy: H2O (l), H2O (s), H2O2 (l), C (s, diamond), D2O (l)
Background
Topic: Third Law of Thermodynamics and Entropy Trends
This question tests your understanding of the third law of thermodynamics and how molecular structure and phase affect entropy.
Key Terms and Formulas:
Third Law of Thermodynamics: The entropy of a perfect crystal at absolute zero is zero.
Entropy Trends: Entropy increases with increasing molecular complexity, disorder, and phase (solid < liquid < gas).
Step-by-Step Guidance
Recall the third law of thermodynamics and its implications for entropy values at 0 K.
Compare the phases of the substances (solid vs. liquid) and predict which has higher entropy.
Consider molecular complexity (number of atoms, mass) when comparing entropy values for similar phases.
Arrange the substances in order from least to most entropy based on your analysis.
Try solving on your own before revealing the answer!
Q4. Use the standard free energies of formation to determine ΔGrxn for the reaction: CH4(g) + 0.5 O2(g) → CO2(g) + 2 H2O(g) + O3(g) at 25°C.
Background
Topic: Calculating Standard Free Energy Change
This question tests your ability to use standard free energies of formation () to calculate the standard free energy change for a reaction.
Key Terms and Formulas:
Standard Free Energy of Formation (): The free energy change when 1 mole of a compound forms from its elements in their standard states.
Formula:

Step-by-Step Guidance
Write out the balanced chemical equation and identify the number of moles of each reactant and product.
List the values for each substance from the provided table.
Multiply each value by the number of moles for that substance in the reaction.
Set up the equation for using the formula above, but do not calculate the final value yet.
Try solving on your own before revealing the answer!
Q5. Find ΔGrxn for the reaction: 3 C(s) + 4 H2(g) → C3H8(g) using the following reactions with known ΔGrxn values.
Background
Topic: Hess's Law for Gibbs Free Energy
This question tests your ability to use Hess's Law to combine reactions and their free energy changes to find the overall for a target reaction.
Key Terms and Formulas:
Hess's Law: The total free energy change for a reaction is the sum of the free energy changes for the steps into which the reaction can be divided.
Key Formula: (with appropriate manipulation of equations)
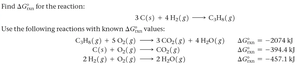
Step-by-Step Guidance
Write the target reaction and the given reactions with their values.
Determine how to manipulate (reverse or multiply) the given reactions so that, when added, they yield the target reaction.
Apply the same manipulations to the values (reverse sign if reaction is reversed, multiply if coefficients are changed).
Set up the sum of the adjusted values, but do not compute the final total yet.
Try solving on your own before revealing the answer!
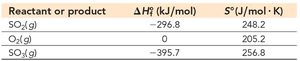
Q6. Calculate ΔGrxn at 25°C for SO2(g) + 0.5 O2(g) → SO3(g) and determine whether the reaction is spontaneous.
Background
Topic: Calculating ΔG from ΔH and ΔS
This question tests your ability to calculate the standard free energy change using enthalpy and entropy values, and to interpret spontaneity.
Key Terms and Formulas:
Standard Enthalpy Change (): The heat change at constant pressure for a reaction with all substances in their standard states.
Standard Entropy Change (): The entropy change for a reaction with all substances in their standard states.
Formula:
Step-by-Step Guidance
Write the balanced chemical equation and identify the number of moles of each reactant and product.
List the and values for each substance from the table.
Calculate and using the products minus reactants method.
Set up the equation with your calculated values, but do not solve for the final value yet.
Try solving on your own before revealing the answer!
