 Back
BackThermodynamics: Entropy, Gibbs Free Energy, and Spontaneity (Ch.19.3–19.6)
Study Guide - Smart Notes

Thermodynamics: Entropy, Gibbs Free Energy, and Spontaneity
Entropy at the Molecular Level
Entropy (S) is a measure of disorder or randomness in a system. At the molecular level, entropy is related to the number of possible arrangements (microstates) of molecules in a system. Statistical thermodynamics connects macroscopic properties to microscopic behaviors.
Microstate: A specific arrangement of the positions and energies of all molecules in a system.
Boltzmann Equation: The relationship between entropy and microstates is given by , where k is the Boltzmann constant and W is the number of microstates.
Entropy increases as the number of microstates increases.
Molecular Motions: Molecules exhibit translational, vibrational, and rotational motions, each contributing to the number of microstates and thus the entropy.
Translational: Movement from one place to another.
Vibrational: Periodic motion of atoms within a molecule.
Rotational: Rotation about an axis.
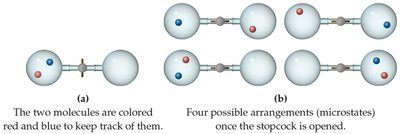

Factors Affecting Entropy
The number of microstates and thus entropy increases with:
Increasing volume (more possible positions)
Increasing temperature (greater distribution of molecular speeds)
Increasing number of atoms/molecules (more degrees of freedom)
Entropy increases with the freedom of motion: .
Processes that increase entropy include formation of gases from solids or liquids, formation of liquids or solutions from solids, and increase in the number of gas molecules during a reaction.
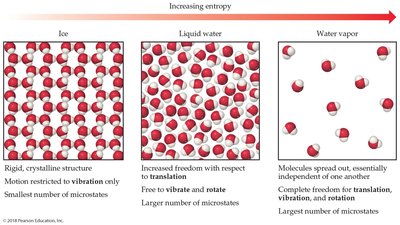
The Third Law of Thermodynamics
The third law states that the entropy of a pure crystalline substance at absolute zero (0 K) is zero. At this temperature, the substance has only one microstate.
At 0 K, .
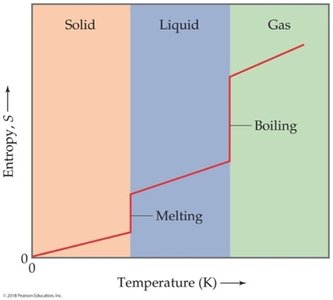
Standard Molar Entropy and Entropy Changes in Reactions
Standard molar entropy () is the entropy of one mole of a substance under standard conditions. Entropy changes in reactions can be calculated similarly to enthalpy changes:
Standard molar entropy is not zero for elements.
Gases have higher entropy than liquids, which have higher entropy than solids.
Entropy values generally increase with increasing molar mass and number of atoms in the formula.

Gibbs Free Energy ()
Gibbs free energy is a thermodynamic state function that predicts the spontaneity of a reaction. It combines enthalpy and entropy:
If , the reaction is spontaneous.
If , the reaction is non-spontaneous.
If , the system is at equilibrium.
Standard free energy changes can be calculated as:
for elements in their standard state is zero.
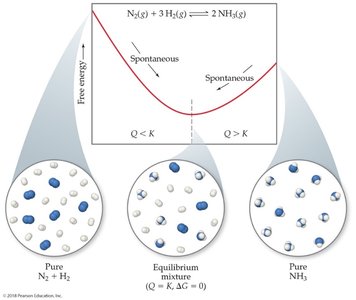
Temperature Dependence of Free Energy
The sign and magnitude of depend on both and , as well as temperature:
Spontaneity can change with temperature, especially for reactions where and have opposite signs.

Key Concepts and Examples
Spontaneous Process: Occurs without external intervention; .
Irreversible Process: Cannot return to original state without external work.
Reversible Process: Can return to original state; .
Entropy Change (): Positive when disorder increases (e.g., solid to gas).
Enthalpy Change (): Positive for endothermic, negative for exothermic processes.
Example: Boiling water () has and .
Example: Condensation () has and .
Sample Calculations and Practice
Calculate for vaporization of methanol using provided data:
Boiling point temperature can be found by setting :
Summary Table: Entropy and Spontaneity
Process | ΔS | ΔH | ΔG | Spontaneity |
|---|---|---|---|---|
Solid → Liquid | + | + | Depends on T | Spontaneous at high T |
Liquid → Gas | + | + | Depends on T | Spontaneous at high T |
Gas → Liquid | - | - | Depends on T | Spontaneous at low T |
Formation of gas from solid | + | Varies | Varies | Varies |
Practice Questions and Applications
Predict the sign of for sublimation, dissolution, and condensation processes.
Calculate standard entropy changes for reactions using tabulated values.
Determine spontaneity based on and temperature.
Additional info:
Entropy is a central concept in predicting the direction of chemical reactions.
Gibbs free energy links enthalpy and entropy to determine spontaneity.
Standard molar entropy and free energy values are essential for quantitative calculations in thermochemistry.
