 Back
BackUnit 8: Acids and Bases – General Chemistry Study Notes
Study Guide - Smart Notes

Unit 8: Acids and Bases
Overview
This unit explores the chemistry of acids and bases, focusing on their equilibrium behavior, calculations involving pH and pOH, titration analysis, buffer systems, and the molecular structure underlying acid and base strength. The concepts are foundational for understanding chemical equilibria and solution chemistry in general chemistry.

8.1 Introduction to Acids and Bases
pH, pOH, and Water Autoionization
pH is a measure of the hydronium ion concentration in aqueous solution:
pOH is a measure of the hydroxide ion concentration:
Water autoionizes:
The equilibrium constant for water at 25°C is
At 25°C, ; in pure water,
The value of is temperature dependent; at higher temperatures, $K_w$ increases and neutral pH decreases.
Example: In pure water at 0°C, M, so and .
8.2 pH and pOH of Strong Acids and Bases
Strong Acid and Base Calculations
Strong acids (e.g., HCl, HBr, HNO3) ionize completely in water: initial acid concentration.
Strong bases (e.g., NaOH, Ba(OH)2) dissociate completely: equals the number of hydroxide ions per formula unit times the base concentration.
Mixing strong acids and bases results in neutralization; the pH depends on the excess reagent.
Example: Mixing 50 mL of 0.50 M HCl with 50 mL of 0.30 M NaOH leaves excess acid; calculate the new concentration and pH accordingly.
8.3 Weak Acid and Base Equilibria
Equilibrium and Calculations for Weak Acids and Bases
Weak acids only partially ionize:
Acid dissociation constant: ;
Weak bases only partially ionize:
Base dissociation constant: ;
Percent ionization: for acids
Example: For 0.15 M acetic acid (), .
8.4 Acid-Base Reactions and Buffers
Types of Acid-Base Reactions
Strong acid + strong base: ; pH determined by excess reagent.
Weak acid + strong base: ; buffer forms if weak acid is in excess; use Henderson-Hasselbalch equation.
Weak base + strong acid: ; buffer forms if weak base is in excess.
Weak acid + weak base: ; equilibrium established.
Henderson-Hasselbalch Equation:
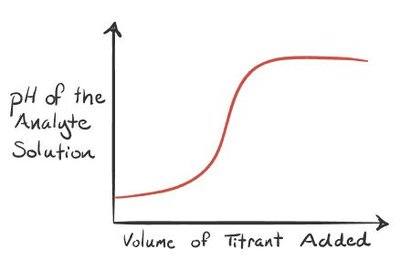
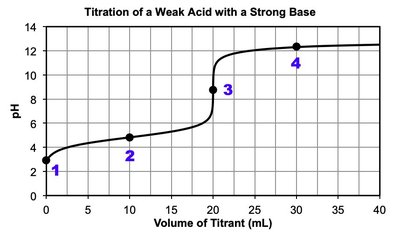
8.5 Acid-Base Titrations
Titration Curves and Analysis
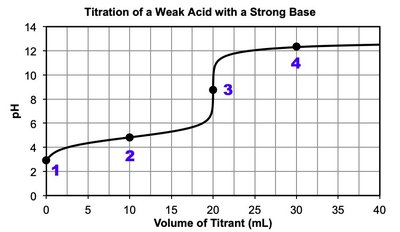
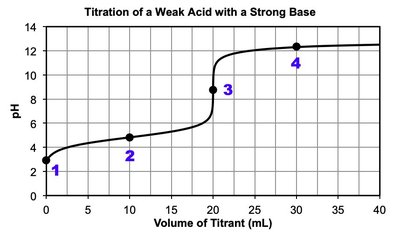
Titration: Laboratory technique to determine the concentration of an unknown acid or base by reacting it with a known titrant.
Equivalence point: Moles of titrant equal moles of analyte; for strong acid/base titrations, pH is neutral at equivalence.
Half-equivalence point: For weak acids/bases, and .
Polyprotic acids: Multiple equivalence points; each corresponds to the loss of a proton.
8.6 Molecular Structure of Acids and Bases
Structure-Strength Relationships
Acid and base strength can be inferred from molecular structure.
Strong acids have very weak, stabilized conjugate bases (by electronegativity, resonance, inductive effects).
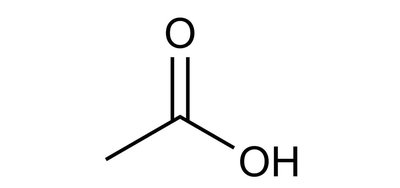
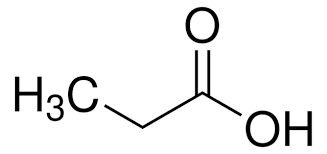
Carboxylic acids (–COOH) are common weak acids.
Strong bases (group I/II hydroxides) have very weak conjugate acids.
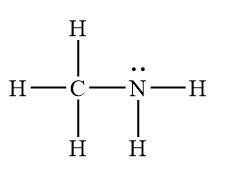
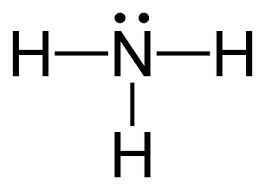
Nitrogenous bases (amines) are common weak bases.
Example: The stability of conjugate bases increases with resonance and inductive effects (e.g., ClO4− is more stable than ClO3−).
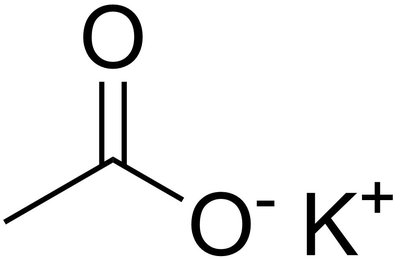
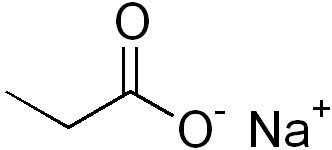
8.7 pH and pKa
Protonation State and Indicators
The ratio of acid to base forms in solution depends on the relationship between pH and pKa.
If pH < pKa, the acid form predominates; if pH > pKa, the base form predominates.
Acid-base indicators change color depending on their protonation state, which is pH-dependent.
Indicator | Color Change | pH Range | pKa |
|---|---|---|---|
Bromophenol blue | Yellow to blue | 3 - 4.5 | 4 |
Bromocresol green | Yellow to blue | 4 - 5 | 4.5 |
Methyl red | Red to yellow | 4.5 - 6 | 5 |
Bromothymol blue | Yellow to blue | 6 - 7.5 | 7 |
Phenol red | Yellow to red | 7 - 8 | 7.5 |
Phenolphthalein | Colorless to pink | 8 - 10 | 9 |
Alizarin yellow R | Yellow to red | 10 - 12 | 11 |
8.8 Properties of Buffers
Buffer Action and Preparation
A buffer contains significant concentrations of both a weak acid and its conjugate base (or weak base and conjugate acid).
Buffers resist changes in pH when small amounts of strong acid or base are added.
Preparation methods include mixing a weak acid with its salt, or partially neutralizing a weak acid/base with a strong base/acid.
8.9 Henderson-Hasselbalch Equation
Buffer Calculations
The Henderson-Hasselbalch equation relates buffer pH to the ratio of conjugate base to acid:
Adding small amounts of acid or base to a buffer does not significantly change the pH.
8.10 Buffer Capacity
Factors Affecting Buffer Capacity
Buffer capacity increases with the total concentration of buffer components, not their ratio.
If [acid] > [base], the buffer can neutralize more added base; if [base] > [acid], it can neutralize more added acid.
Buffer capacity is the amount of acid or base a buffer can neutralize before a significant pH change occurs.
8.11 pH and Solubility
Effect of pH on Salt Solubility
The solubility of salts containing weak acids or bases is affected by pH.
For basic salts (e.g., Mg(OH)2), increasing [H+] (lower pH) increases solubility by shifting equilibrium right.
For acidic salts (e.g., HNO2), increasing [OH−] (higher pH) increases solubility by removing H+.
Le Châtelier’s principle explains these shifts in equilibrium.
Example: Mg(OH)2 is more soluble in acidic solutions; HNO2 is more soluble in basic solutions.
