 Back
BackUniversity Chemistry II Exam 1 – Step-by-Step Study Guidance
Study Guide - Smart Notes

Q1. Consider the following reaction: 2 C2H6(g) + 7 O2(g) → 4 CO2(g) + 6 H2O(g). The rate of oxygen consumption (Δ[O2]/Δt) has been measured as 9.34 M/s.
Background
Topic: Reaction Rates and Stoichiometry
This question tests your understanding of how to relate the rate of consumption or formation of one species in a reaction to the overall rate and to the rates of other species using stoichiometry.
Key Terms and Formulas:
Rate of reaction: The change in concentration of a reactant or product per unit time.
Stoichiometric coefficients: Numbers in front of compounds in a balanced equation, used to relate rates of different species.
Step-by-Step Guidance
Write the balanced equation and identify the stoichiometric coefficients for each species.
Recall that the rate of reaction is defined as the rate of change of concentration divided by the stoichiometric coefficient (with a negative sign for reactants).
Set up the relationship between the rate of oxygen consumption and the overall rate using the stoichiometric coefficient for O2.
Express the overall rate in terms of Δ[O2]/Δt and the coefficient from the balanced equation.
Try solving on your own before revealing the answer!
Q2. The experimentally observed rate law for a reaction is found to be: Rate = k [Ce4+]2[Mn2+]. Complete the following statements.
Background
Topic: Rate Laws and Reaction Order
This question tests your ability to interpret a rate law and determine the order of reaction with respect to each reactant and overall.
Key Terms and Formulas:
Order with respect to a reactant: The exponent of its concentration in the rate law.
Overall order: The sum of the exponents in the rate law.
Step-by-Step Guidance
Identify the exponent for [Ce4+] in the rate law.
Identify the exponent for [Mn2+] in the rate law.
Add the exponents to find the overall order of the reaction.
Try solving on your own before revealing the answer!
Q3. The rate constant for the first-order decomposition of N2O is 3.40 s-1. What is the half-life of the decomposition when [N2O]0 = 0.782 M?
Background
Topic: Kinetics – First-Order Reactions and Half-Life
This question tests your ability to use the half-life formula for a first-order reaction.
Key Terms and Formulas:
First-order half-life formula:
k = rate constant (s-1)
Step-by-Step Guidance
Identify the given rate constant, k = 3.40 s-1.
Recall the half-life formula for a first-order reaction.
Substitute the value of k into the formula to set up the calculation for t1/2.
Try solving on your own before revealing the answer!
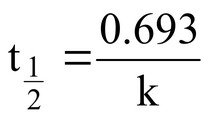
Q4. The second-order decomposition of HI has a rate constant of 1.80 x 10-3 M-1s-1. How much HI remains after 27.3 s if the initial concentration of HI is 4.78 M?
Background
Topic: Kinetics – Second-Order Reactions
This question tests your ability to use the integrated rate law for a second-order reaction to find the concentration at a given time.
Key Terms and Formulas:
Second-order integrated rate law:
k = rate constant (M-1s-1)
[A]0 = initial concentration
[A]t = concentration at time t
Step-by-Step Guidance
Write down the given values: k, [HI]0, and t.
Write the second-order integrated rate law for HI.
Substitute the given values into the equation to set up the calculation for [HI]t.
Rearrange the equation to solve for [HI]t (but do not calculate yet).
Try solving on your own before revealing the answer!
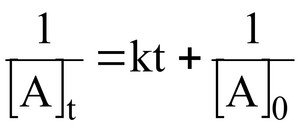
Q5. The reaction C2H5Br + OH- → C2H5OH + Br- in aqueous solution has an activation energy of 89.5 kJ/mol, and the rate constant for this reaction at 20°C is 4.76×10-5 M-1s-1. What is the value of the rate constant at 80°C?
Background
Topic: Arrhenius Equation and Temperature Dependence of Rate Constants
This question tests your ability to use the Arrhenius equation to relate rate constants at two different temperatures.
Key Terms and Formulas:
Arrhenius equation (two-point form):
k1, k2 = rate constants at T1 and T2
Ea = activation energy (J/mol)
R = gas constant (8.314 J/(mol·K))
Step-by-Step Guidance
Convert all temperatures to Kelvin.
Convert activation energy to J/mol if necessary.
Write the Arrhenius equation in the two-point form.
Substitute the known values (k1, T1, T2, Ea, R) into the equation.
Rearrange to solve for k2 (but do not calculate yet).
Try solving on your own before revealing the answer!
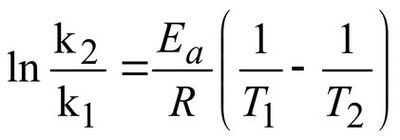
Q6. For the bromination of acetone, CH3COCH3 + Br2 → CH3COCH2Br + H+ + Br-, which takes place in acidic solution, the initial rate of the reaction was measured using different initial concentrations of acetone and bromine. The results of these experiments are summarized below:
Background
Topic: Determining Rate Laws from Experimental Data
This question tests your ability to determine the rate law and rate constant from initial rate data.
Key Terms and Formulas:
Rate law: Rate = k [acetone]^m [Br2]^n
Initial rate method: Compare experiments where only one reactant concentration changes to determine the order with respect to each reactant.
Step-by-Step Guidance
Compare runs where only [Br2] changes to determine the order with respect to Br2.
Compare runs where only [acetone] changes to determine the order with respect to acetone.
Write the rate law based on your findings.
Choose any run and substitute the values into the rate law to solve for the rate constant k (but do not calculate yet).
Try solving on your own before revealing the answer!
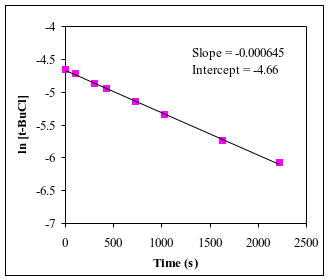
Q7. For the reaction (CH3)3CCl + H2O → (CH3)3COH + HCl, an experiment was performed to determine the order with respect to t-butyl chloride, (CH3)3CCl. The experimenter measured the concentration of t-butyl chloride [“t-BuCl” for short] at several different times during the course of the reaction. From the data obtained, the following plots were produced. Using the above graphs, determine the order of the reaction with respect to t-butyl chloride and find the numerical value of the rate constant (including units).
Background
Topic: Determining Reaction Order from Graphical Data
This question tests your ability to interpret kinetic data and determine reaction order and rate constant from plots.
Key Terms and Formulas:
Zero-order: [A] vs. t is linear
First-order: ln[A] vs. t is linear
Second-order: 1/[A] vs. t is linear
The slope of the linear plot gives the rate constant (or its negative).
Step-by-Step Guidance
Examine the three plots and identify which one is linear (straight line).
Match the linear plot to the corresponding reaction order.
Use the slope of the linear plot to determine the rate constant (be careful with sign and units).
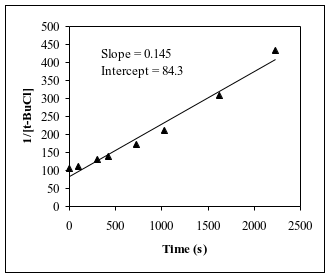
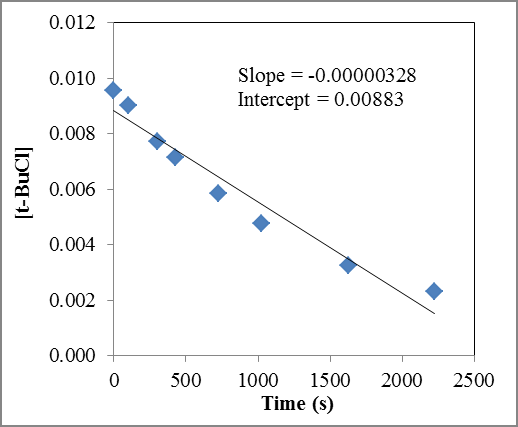
Try solving on your own before revealing the answer!
Q8. Write the Kp expression for the following reaction: P4O10(s) ⇌ P4(s) + 5 O2(g)
Background
Topic: Equilibrium Constants (Kp)
This question tests your ability to write the equilibrium constant expression for a reaction involving gases and solids.
Key Terms and Formulas:
Kp: Equilibrium constant in terms of partial pressures (only gases appear in the expression; solids and liquids are omitted).
Step-by-Step Guidance
Identify which species are gases and which are solids in the reaction.
Write the Kp expression using only the partial pressures of gaseous species, raised to the power of their coefficients.
Try solving on your own before revealing the answer!
Q9. Given the balanced reaction for the formation of ammonia below, (a) write the concentration equilibrium constant expression for this reaction. (b) Given that the equilibrium concentrations are [N2] = 0.373 M, [H2] = 0.114 M, and [NH3] = 0.305 M at 500 K, calculate the numerical value of the equilibrium constant, Kc. (c) At 298 K [note this is a different temperature than in part (B)], Kp = 5.4×105. Given this Kp value, calculate the value of Kc for the formation of ammonia at 298 K.
Background
Topic: Chemical Equilibrium – Kc and Kp
This question tests your ability to write equilibrium constant expressions and convert between Kp and Kc.
Key Terms and Formulas:
Kc: Equilibrium constant in terms of concentrations.
Kp: Equilibrium constant in terms of partial pressures.
Relationship between Kp and Kc:
Δn = moles of gaseous products – moles of gaseous reactants
R = 0.08206 L·atm·mol-1·K-1
Step-by-Step Guidance
Write the balanced equation and the Kc expression for the reaction.
Substitute the given equilibrium concentrations into the Kc expression to set up the calculation.
For part (c), use the Kp/Kc relationship and solve for Kc, substituting the appropriate values for R, T, and Δn.
Try solving on your own before revealing the answer!
Q10. The oxidation of thallium(I) by cerium(IV) in the presence of manganese(II) proceeds by the following mechanism...
Background
Topic: Reaction Mechanisms, Catalysts, and Intermediates
This question tests your ability to deduce the overall reaction, identify catalysts and intermediates, and determine molecularity from a proposed mechanism.
Key Terms and Formulas:
Catalyst: A species that appears in the mechanism, is consumed in one step, and regenerated in another.
Intermediate: A species produced in one step and consumed in another, not present in the overall reaction.
Molecularity: The number of reactant particles involved in an elementary step.
Step-by-Step Guidance
Add up the elementary steps to find the overall reaction (cancel species that appear on both sides).
Identify which species are catalysts and which are intermediates based on their appearance in the steps.
For Step 2, count the number of reactant particles to determine its molecularity.
Try solving on your own before revealing the answer!
Q11. (a) Explain how a catalyst works to increase the rate of a reaction. (b) Consider the generic rate law: Rate = k [A]^x [B]^y. Which rate law parameter is affected by the catalyst and how is it changed?
Background
Topic: Catalysis and Rate Laws
This question tests your understanding of how catalysts affect reaction rates and which parameters in the rate law are influenced.
Key Terms and Formulas:
Catalyst: Lowers activation energy, increases rate constant k.
Rate law: Rate = k [A]^x [B]^y
Step-by-Step Guidance
Explain the effect of a catalyst on the activation energy and reaction pathway.
Identify which parameter in the rate law is directly affected by the catalyst.
Describe how this parameter changes (increases or decreases) in the presence of a catalyst.
Try solving on your own before revealing the answer!
Q12. Consider the following reactions at 500 K. (a) Which of the above reactions has the greatest tendency towards product formation? (b) Which of the above reactions has the least tendency towards product formation?
Background
Topic: Equilibrium Constants and Product Tendency
This question tests your ability to interpret Kp values to determine which reactions favor products or reactants.
Key Terms and Formulas:
Kp: The larger the Kp, the greater the tendency toward product formation; the smaller the Kp, the greater the tendency toward reactant formation.
Step-by-Step Guidance
Compare the Kp values for all reactions listed.
Identify the reaction with the largest Kp (greatest product tendency).
Identify the reaction with the smallest Kp (least product tendency).
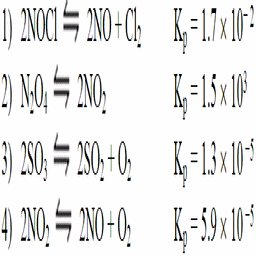
Try solving on your own before revealing the answer!
Q13. The equilibrium constant is given for one of the reactions below. Determine the value of the missing equilibrium constant.
Background
Topic: Manipulating Equilibrium Constants
This question tests your ability to relate equilibrium constants for reactions that are multiples or reversals of each other.
Key Terms and Formulas:
If a reaction is reversed, K becomes 1/K.
If a reaction is multiplied by n, K is raised to the nth power.
Step-by-Step Guidance
Compare the two reactions and determine how one is related to the other (reversal, multiplication, etc.).
Apply the appropriate mathematical operation to the given Kc to find the missing value.
Try solving on your own before revealing the answer!
