 Back
BackValence Bond Theory and Molecular Orbital Theory: Hybridization and Bonding in Molecules
Study Guide - Smart Notes

Valence Bond Theory
Introduction to Valence Bond Theory
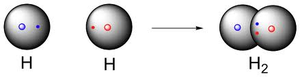
Valence Bond (VB) Theory provides a quantum mechanical explanation for how atoms bond to form molecules. Unlike the Lewis model, which only shows electron pairs, VB theory explains bonding as the overlap of half-filled atomic orbitals, resulting in covalent bonds.
Key Concept: Covalent bonds form when two half-filled atomic orbitals overlap, allowing electrons to be shared between atoms.
Example: The formation of H2 from two hydrogen atoms, each with one unpaired electron in a 1s orbital.
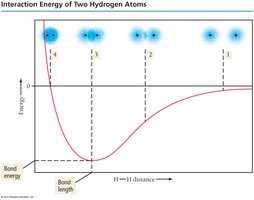
Energy Considerations in Bond Formation
As two hydrogen atoms approach each other, their atomic orbitals overlap, and the system's energy decreases until an optimal bond length is reached. This minimum energy corresponds to the bond energy of the H2 molecule.
Bond Energy: The energy required to break a bond between two atoms.
Bond Length: The distance at which the energy is minimized and the bond is most stable.
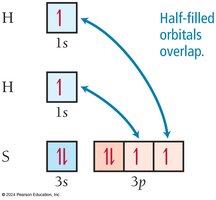
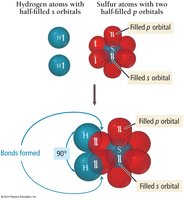
Application to More Complex Molecules: H2S Example
VB theory can be extended to molecules like H2S. Sulfur has two unpaired electrons in its 3p orbitals, which can each overlap with a hydrogen 1s orbital to form two S–H bonds.
Electron Configuration: H: 1s1; S: [Ne] 3s23p4
Bond Formation: Each hydrogen atom fills one of sulfur's unpaired electrons, forming two bonds.
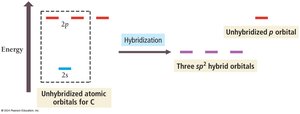
Orbital Hybridization
Need for Hybridization
Some molecules, such as methane (CH4), cannot be explained by simple orbital overlap. Carbon has only two unpaired electrons in its ground state, but forms four equivalent bonds in CH4. This discrepancy is resolved by the concept of hybridization.
Hybridization: The mixing of atomic orbitals to form new, equivalent hybrid orbitals suitable for bonding.
sp3 Hybridization: One 2s and three 2p orbitals combine to form four sp3 hybrid orbitals, each with one unpaired electron.
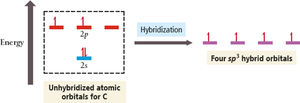
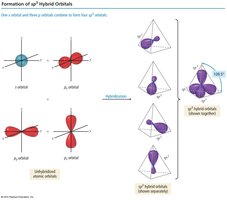
sp3 Hybridization in Methane
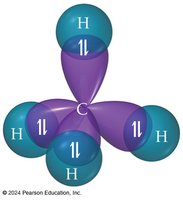
In methane, the four sp3 hybrid orbitals arrange themselves tetrahedrally, resulting in four equivalent C–H bonds with bond angles of 109.5°.
Geometry: Tetrahedral
Bond Angle: 109.5°
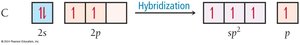
Hybridization with Fewer Bonding Domains: sp2 and sp
When an atom is bonded to fewer than four groups, it uses fewer hybrid orbitals. For example, in formaldehyde (CH2O), carbon forms three sigma bonds and one pi bond, using sp2 hybridization.
sp2 Hybridization: One s and two p orbitals combine to form three sp2 hybrid orbitals; one p orbital remains unhybridized for pi bonding.
sp Hybridization: One s and one p orbital combine to form two sp hybrid orbitals; two p orbitals remain unhybridized for pi bonding.
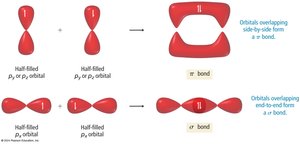
Sigma (σ) and Pi (π) Bonds
Types of Covalent Bonds
Covalent bonds are classified as sigma (σ) or pi (π) bonds based on the type of orbital overlap:
Sigma (σ) Bond: Formed by end-to-end overlap of orbitals along the axis connecting two nuclei. All single bonds are sigma bonds.
Pi (π) Bond: Formed by side-to-side overlap of parallel p orbitals, with electron density above and below the internuclear axis. Present in double and triple bonds.
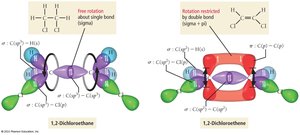
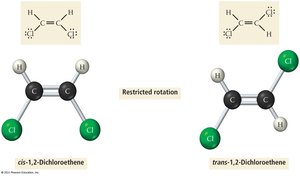
Double and Triple Bonds
Double bonds consist of one sigma and one pi bond, while triple bonds consist of one sigma and two pi bonds. Pi bonds restrict rotation around the bond axis, leading to geometric isomerism (cis/trans isomers).
Example: Ethylene (C2H4) has a double bond (one σ, one π); acetylene (C2H2) has a triple bond (one σ, two π).
Hybridization Schemes and Molecular Geometry
Summary Table: Electron Groups and Hybridization
The type of hybridization depends on the number of electron groups (bonding and lone pairs) around the central atom:
Electron Groups | Hybridization | Geometry | Bond Angles |
|---|---|---|---|
2 | sp | Linear | 180° |
3 | sp2 | Trigonal planar | 120° |
4 | sp3 | Tetrahedral | 109.5° |
5 | sp3d | Trigonal bipyramidal | 90°, 120° |
6 | sp3d2 | Octahedral | 90° |

Molecular Orbital Theory
Introduction to Molecular Orbital (MO) Theory
Molecular Orbital Theory describes bonding in terms of molecular orbitals that are delocalized over the entire molecule, rather than localized between two atoms. This approach explains properties such as magnetism and bond order more accurately than VB theory.
Bonding MO: Constructive combination of atomic orbitals, lower in energy, stabilizes the molecule.
Antibonding MO: Destructive combination, higher in energy, destabilizes the molecule.
Bond Order Calculation
Bond order indicates the number of chemical bonds between a pair of atoms. It is calculated as:
Bond Order Formula:
Example: For H2, bond order = 1; for He2, bond order = 0 (does not exist).
MO Diagrams for Second-Row Diatomics
For molecules like O2, MO theory predicts the presence of unpaired electrons, explaining its paramagnetic behavior (attraction to a magnetic field).
Paramagnetism: Caused by unpaired electrons in molecular orbitals.
Oxygen (O2): Has two unpaired electrons in π* antibonding orbitals, making it paramagnetic.
Summary
Valence Bond Theory explains bonding as the overlap of half-filled atomic orbitals.
Hybridization accounts for observed molecular geometries and equivalent bonds.
Sigma and pi bonds arise from different types of orbital overlap.
Molecular Orbital Theory provides a more complete picture, explaining bond order and magnetic properties.
