 Back
BackValence Bond Theory, Hybridization, and Molecular Orbital Theory: Study Notes
Study Guide - Smart Notes

Valence Bond Theory
Basic Principles of Valence Bond Theory
Valence Bond Theory explains the formation of covalent bonds through the overlap of atomic orbitals containing unpaired electrons. The number of bonds an atom can form is determined by its number of unpaired electrons. This theory is effective for simple diatomic molecules and explains the inertness of noble gases due to their lack of unpaired electrons.
Covalent Bond Formation: Requires unpaired electrons in atomic orbitals.
Bonding Capacity: Determined by the number of unpaired electrons.
Limitations: Works best for single bonds and simple molecules.
Orbital Overlap and Bond Formation
Covalent bonds are formed when atomic orbitals overlap in space, allowing electrons to be shared between atoms. The type of overlap determines the nature of the bond.
Sigma (σ) Bonds: Formed by head-on overlap of orbitals (s-s, s-p, or p-p).
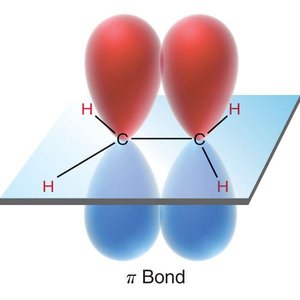
Pi (π) Bonds: Formed by side-to-side overlap of p orbitals, present in double and triple bonds.
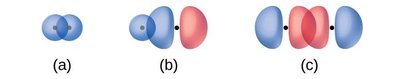
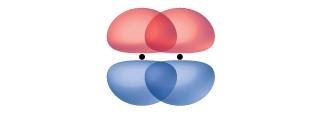
Sigma and Pi Bonds in Molecules
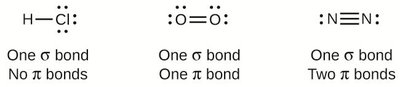
Sigma bonds are the first bonds formed between two atoms, while pi bonds are additional bonds found in double and triple bonds. The number of sigma and pi bonds in a molecule can be determined from its Lewis structure.
Single Bond: One σ bond, no π bonds.
Double Bond: One σ bond, one π bond.
Triple Bond: One σ bond, two π bonds.
Hybridization
Concept of Hybridization
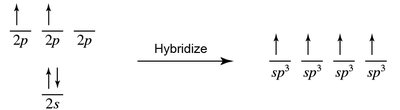
Hybridization is the mathematical combination of atomic s and p orbitals to form new, equivalent hybrid orbitals. This process, known as Linear Combination of Atomic Orbitals (LCAO), helps explain molecular geometry and bonding in molecules.
Hybrid Orbitals: Have equal energy and are oriented to maximize bond formation and minimize electron repulsion.
Types of Hybridization: sp, sp2, sp3, etc., depending on the number of electron domains.
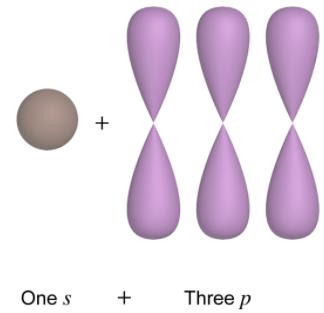
Determining Hybridization and Molecular Geometry
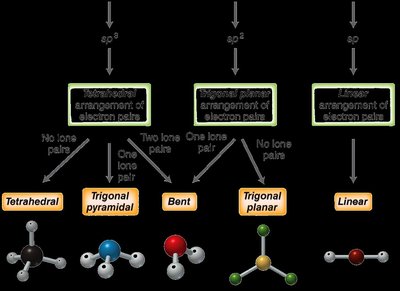
The type of hybridization can be quickly determined by counting the number of electron domains (regions of electron density) around the central atom:
2 electron domains: sp hybridization (linear geometry)
3 electron domains: sp2 hybridization (trigonal planar geometry)
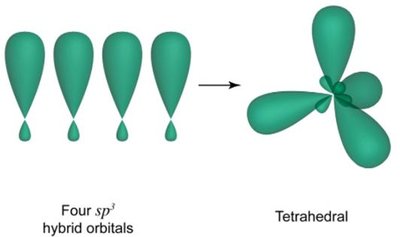
4 electron domains: sp3 hybridization (tetrahedral geometry)
Hybridization in Molecules
Hybridization explains the observed shapes of molecules and the equivalence of bonds. For example, in methane (CH4), carbon undergoes sp3 hybridization to form four equivalent tetrahedral bonds.
sp3 Hybridization: Four equivalent orbitals, tetrahedral geometry.
sp2 Hybridization: Three equivalent orbitals, trigonal planar geometry.
sp Hybridization: Two equivalent orbitals, linear geometry.

Sigma and Pi Bonding in Multiple Bonds
Bonding in Double and Triple Bonds
In molecules with double or triple bonds, hybridized orbitals form sigma bonds, while unhybridized p orbitals form pi bonds. Pi bonds are generally weaker than sigma bonds due to less effective orbital overlap.
Double Bond: One σ bond (from hybridized orbitals), one π bond (from unhybridized p orbitals).
Triple Bond: One σ bond, two π bonds.
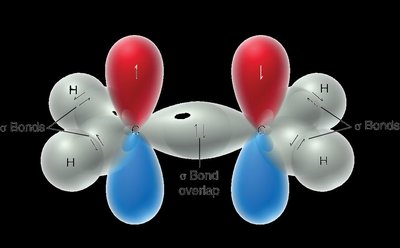
Molecular Orbital (MO) Theory
Fundamentals of MO Theory
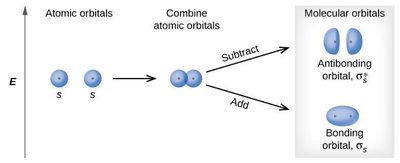
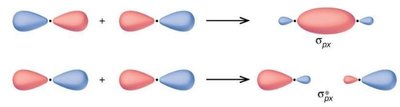
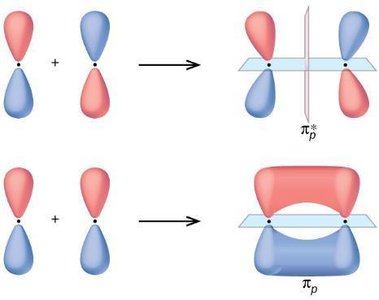
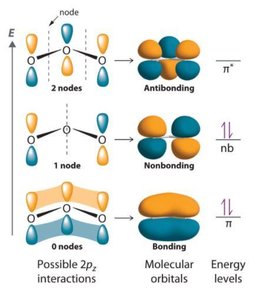
Molecular Orbital Theory describes electrons in molecules as occupying molecular orbitals that are delocalized over the entire molecule. These orbitals are formed by the constructive or destructive combination of atomic orbital wavefunctions.
Bonding Orbitals: Formed by constructive interference, lower in energy.
Antibonding Orbitals: Formed by destructive interference, higher in energy.
Non-bonding Orbitals: Associated with lone pairs.
Molecular Orbital Diagrams
MO diagrams show the relative energy levels of molecular orbitals formed from atomic orbitals. They are used to determine bond order, bond stability, bond length, and magnetic properties.
Bond Order:
Stability: Higher bond order means greater stability.
Bond Length: Higher bond order means shorter bond length.
Magnetism: Unpaired electrons in MO diagram indicate paramagnetism.
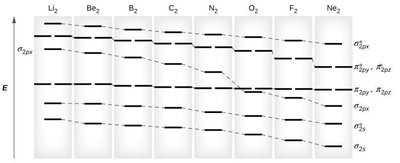
MO Diagrams for Heteronuclear Molecules and Resonance
When two different atoms form a molecule, the MO diagram is adjusted to reflect differences in electronegativity. MO theory also provides a framework for understanding resonance by showing delocalized electrons in molecular orbitals.
Heteronuclear Molecules: Less electronegative atom's orbitals are higher in energy.
Resonance: Delocalization of electrons is best described by MO theory.
Frontier Orbitals: HOMO and LUMO
The most chemically important molecular orbitals are the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO). Chemical reactions often involve electron transfer from the HOMO of one molecule to the LUMO of another.
HOMO: Highest energy orbital containing electrons.
LUMO: Lowest energy orbital that can accept electrons.
Reactivity: Determined by the energy gap between HOMO and LUMO.
