 Back
BackValence Bond Theory, Hybridization, and Molecular Geometry
Study Guide - Smart Notes

Valence Bond Theory and Molecular Geometry
Introduction to Valence Bond Theory (VBT)
Valence Bond Theory (VBT) is a fundamental model in chemistry that explains how atoms bond to form molecules by the overlap of atomic orbitals. It provides insight into the shapes and properties of molecules, complementing the Lewis structure approach and connecting with the Valence Shell Electron Pair Repulsion (VSEPR) theory.
Atomic Orbitals and Their Shapes
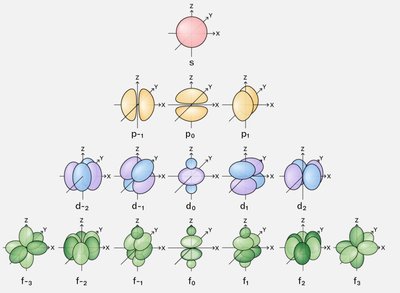
Atomic orbitals are regions in an atom where electrons are likely to be found. The main types are s, p, d, and f orbitals, each with distinct shapes and orientations:
s orbital: Spherical shape, centered around the nucleus.
p orbitals: Dumbbell-shaped, oriented along x, y, and z axes.
d and f orbitals: More complex shapes, important for transition metals and inner transition elements.
Overlap of Atomic Orbitals and Bond Formation
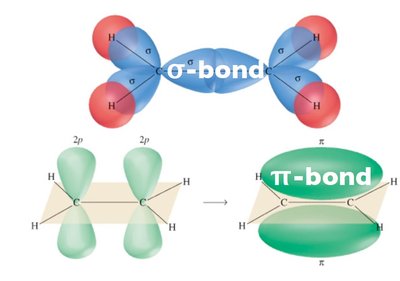
Chemical bonds form when atomic orbitals overlap. The type of overlap determines the bond type:
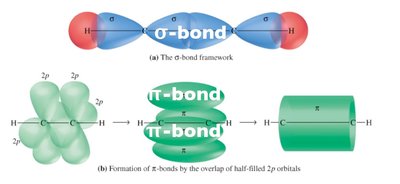
σ (sigma) bonds: Formed by head-on overlap (s-s, s-p, or p-p along the axis).
π (pi) bonds: Formed by sidewise (lateral) overlap of p orbitals.
Hybridization of Atomic Orbitals
Concept of Hybridization
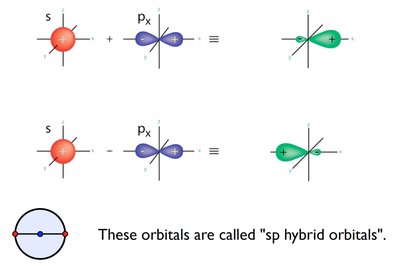
Hybridization is the process by which atomic orbitals mix to form new, equivalent hybrid orbitals suitable for the pairing of electrons to form chemical bonds in molecules. This concept explains the observed molecular geometries that cannot be described by simple atomic orbitals alone.
Only orbitals with at least one electron and that participate in bonding can hybridize.
Hybridization leads to orbitals with the same energy and shape, optimized for bonding.
Types of Hybridization
sp Hybridization: Mixing of one s and one p orbital, forming two linear hybrid orbitals (180° bond angle). Example: BeCl2.
sp2 Hybridization: Mixing of one s and two p orbitals, forming three trigonal planar hybrid orbitals (120° bond angle). Example: BF3.
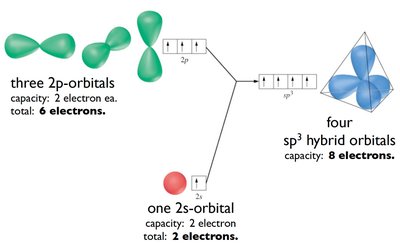
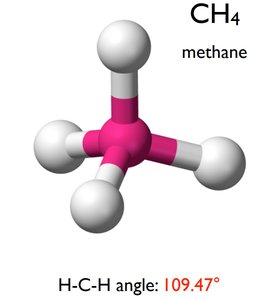
sp3 Hybridization: Mixing of one s and three p orbitals, forming four tetrahedral hybrid orbitals (109.5° bond angle). Example: CH4.
sp3d and sp3d2 Hybridization: Involves d orbitals, leading to trigonal bipyramidal and octahedral geometries, respectively.

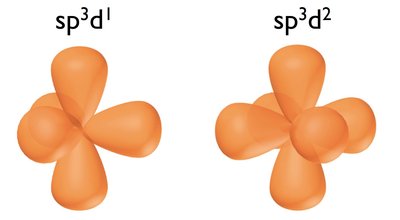
Examples and Molecular Geometry
BeCl2: Linear geometry, sp hybridization, 180° bond angle.
BF3: Trigonal planar geometry, sp2 hybridization, 120° bond angle.
CH4: Tetrahedral geometry, sp3 hybridization, 109.5° bond angle.
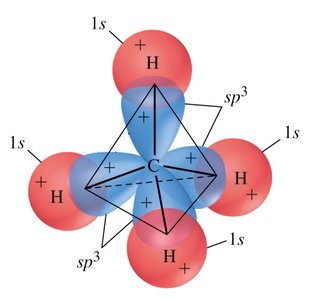
Hybridization and VSEPR Theory
Hybridization is closely linked to VSEPR theory, which predicts molecular shapes based on electron pair repulsion. The type of hybridization adopted by the central atom determines the geometry of the molecule.
Comparison: Lewis Structures vs. Valence Bond Theory
Lewis structures provide a simple way to represent molecules, showing how atoms are connected and where lone pairs reside. However, they do not explain the three-dimensional geometry or the nature of bonding orbitals. Valence Bond Theory, through hybridization, fills this gap by describing the spatial arrangement of bonds and the types of orbitals involved.
Geometry of Organic Molecules: Ethane, Ethylene, Acetylene, and Benzene
Ethane (C2H6): sp3 Hybridization
Each carbon atom forms four sp3 hybrid orbitals, resulting in a tetrahedral geometry with bond angles of 109.5°.
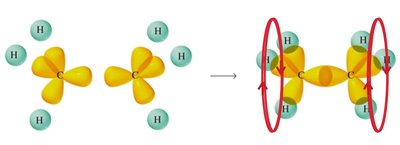
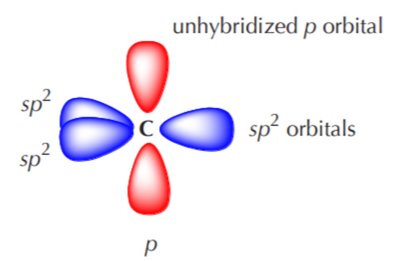
Ethylene (C2H4): sp2 Hybridization
Each carbon forms three sp2 hybrid orbitals (trigonal planar, 120°) and one unhybridized p orbital, which forms a π bond.

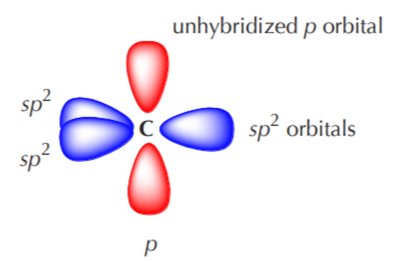
Acetylene (C2H2): sp Hybridization
Each carbon forms two sp hybrid orbitals (linear, 180°) and two unhybridized p orbitals, which form two π bonds.
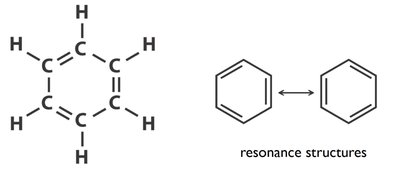
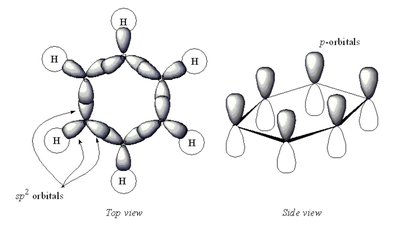
Benzene (C6H6): sp2 Hybridization and Resonance
Each carbon in benzene is sp2 hybridized, forming a planar hexagonal ring with delocalized π electrons, leading to resonance stabilization.
Isomerism and Conformers
Definition of Isomers
Isomers are compounds with the same molecular formula but different arrangements of atoms, resulting in different properties.
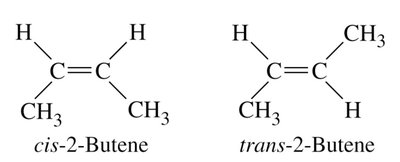
Geometric (cis-trans) Isomerism
Occurs due to restricted rotation around double bonds, leading to different spatial arrangements (cis and trans forms). Example: 2-butene.

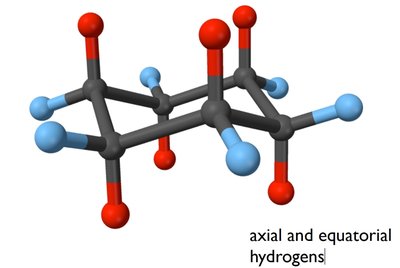
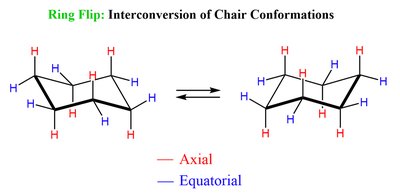
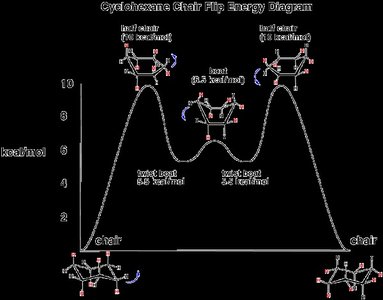
Conformers and Cyclohexane Chair Conformation
Conformers are different spatial arrangements of a molecule that can be interconverted by rotation around single bonds. Cyclohexane exhibits chair conformations, which are more stable due to minimized steric strain.
Calculating Hybridization: Stepwise Method
To determine the hybridization of a central atom:
Add the number of valence electrons of all atoms in the species.
Adjust for charge (add for anions, subtract for cations).
Divide the result as follows:
If less than 8, divide by 2; sum quotient and remainder.
If between 9 and 56, divide by 8 (Q1), then divide the remainder by 2 (Q2), and sum Q1, Q2, and the final remainder.
The final value (X) determines hybridization:
X | Hybridization |
|---|---|
2 | sp |
3 | sp2 |
4 | sp3 |
5 | sp3d |
6 | sp3d2 |
7 | sp3d3 |
Summary Table: Hybridization and Geometry
Species | Hybridization | Geometry | Bond Angle |
|---|---|---|---|
BeCl2 | sp | Linear | 180° |
BF3 | sp2 | Trigonal planar | 120° |
NH3 | sp3 | Tetrahedral (pyramidal) | 107° |
SF6 | sp3d2 | Octahedral | 90° |
IF7 | sp3d3 | Pentagonal bipyramidal | 72°, 90° |
Additional info:
For more complex molecules, Molecular Orbital Theory provides a more complete description of bonding and stability, especially for delocalized systems like benzene.
Lewis and VBT do not account for the relative stability of conformers; this is better explained by energy diagrams and molecular orbital considerations.
