 Back
BackVisualizing and Interpreting Rate Expressions and Reaction Order from Data Tables
Study Guide - Smart Notes

Q1. What does the expression represent in chemical kinetics?
Background
Topic: Reaction Rates and Rate Expressions
This question is about interpreting the meaning of a rate expression in terms of the change in concentration of a reactant () over time. Understanding how to write and interpret these expressions is fundamental in chemical kinetics.
![Delta[SO2] over Delta t](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/01197dad_image_2.png)
Key Terms and Formulas:
= Change in concentration of (in mol/L)
= Change in time (in seconds, s)
Rate of reaction =
Step-by-Step Guidance
Recognize that describes how the concentration of changes with time during a reaction.
Recall that for a reactant, this value is typically negative because the concentration decreases as the reaction proceeds.
Understand that this expression gives the average rate of change over a time interval .
Try solving on your own before revealing the answer!
Q2. Given the following data table, determine the order of the reaction with respect to [A]:
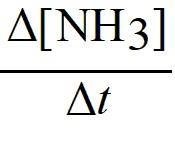
Background
Topic: Determining Reaction Order from Initial Rates
This question tests your ability to use experimental data to deduce the order of a reaction with respect to a single reactant. This is a key skill in chemical kinetics.
Key Terms and Formulas:
Rate law:
Order (): The exponent that shows how the rate depends on the concentration of .
Step-by-Step Guidance
Compare how the initial rate changes as increases. For example, when $[A]$ doubles, what happens to the rate?
Calculate the factor by which changes and the corresponding factor for the rate.
Relate the change in rate to the change in using the general rate law: .
Set up the equation and solve for (but do not finish the calculation yet).
Try solving on your own before revealing the answer!
