 Back
BackWeak Acid-Strong Base and Weak Base-Strong Acid Titrations: Principles, Curves, and Indicators
Study Guide - Smart Notes

Weak Acid-Strong Base and Weak Base-Strong Acid Titrations
Qualitative and Quantitative Principles
Titrations involving weak acids and strong bases, or weak bases and strong acids, are fundamental in analytical chemistry. These titrations differ significantly from strong acid-strong base titrations in their titration curves, buffer regions, and equivalence point characteristics.
Weak Acid-Strong Base Titration: The titration curve shows a gradual increase in pH, a buffer region, and an equivalence point above pH 7.
Weak Base-Strong Acid Titration: The titration curve features a buffer region and an equivalence point below pH 7.
Buffer Region: Occurs when both the weak acid/base and its conjugate are present, resisting changes in pH.
Comparison: Weak Acid-Strong Base vs. Strong Acid-Strong Base Titration Curves
Three key differences distinguish weak acid-strong base titration curves from strong acid-strong base curves:
Initial pH: Weak acids start at a higher initial pH than strong acids.
Buffer Region: Weak acid titrations display a buffer region before the equivalence point, absent in strong acid titrations.
Equivalence Point: The equivalence point for weak acid-strong base titrations is above pH 7, while for strong acid-strong base titrations it is at pH 7.
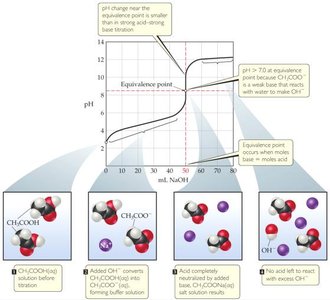
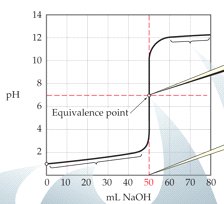
Buffer Region and Half-Equivalence Point
The buffer region is a segment of the titration curve where the solution resists changes in pH. The half-equivalence point is particularly important:
Half-Equivalence Point: Occurs when half of the weak acid has been neutralized. At this point, the concentration of acid equals its conjugate base, and pH = pKa.
Buffer Capacity: The solution is most effective at resisting pH changes near the half-equivalence point.
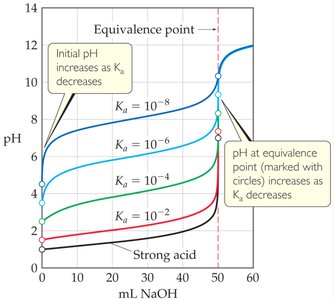
Variations in Titration Curves Due to Acid Strength
The strength of the weak acid (as measured by its Ka) affects the titration curve:
Initial pH: Increases as Ka decreases (weaker acid).
Equivalence Point pH: Also increases as Ka decreases.
Quantitative Calculations in Weak Acid-Strong Base Titrations
Initial pH Calculation
For a weak acid solution, the initial pH is calculated using the acid dissociation constant (Ka):
Example: 0.0250 M benzoic acid (C6H5COOH, Ka = 6.3 × 10–5)
Equation:
Use ICE tables to solve for [H+] and calculate pH.
Buffer Zone Calculation
When a strong base is added to a weak acid, the solution forms a buffer. The pH can be calculated using the Henderson-Hasselbalch equation:
Equation:
Example: Adding 10.0 mL of 0.050 M NaOH to 40.0 mL of 0.0250 M benzoic acid.
Equivalence Point Calculation
At the equivalence point, all the weak acid has been neutralized, and the conjugate base is present. The pH is determined by the hydrolysis of the conjugate base:
Equation:
Calculate [OH–] produced by the conjugate base and determine pH.
Example: 40.0 mL of 0.025 M benzoic acid titrated with 0.050 M NaOH.
Quantitative Calculations in Weak Base-Strong Acid Titrations
Initial pH Calculation
For a weak base solution, the initial pH is calculated using the base dissociation constant (Kb):
Example: 0.100 M NH3 (Kb = 1.8 × 10–5)
Equation:
Use ICE tables to solve for [OH–] and calculate pOH, then pH.
Buffer Zone Calculation
When a strong acid is added to a weak base, the solution forms a buffer. The pH can be calculated using the Henderson-Hasselbalch equation for bases:
Equation:
Convert pOH to pH.
Example: Adding 10.0 mL of 0.100 M HCl to 20.0 mL of 0.100 M NH3.
Equivalence Point Calculation
At the equivalence point, all the weak base has been neutralized, and the conjugate acid is present. The pH is determined by the hydrolysis of the conjugate acid:
Equation:
Calculate [H+] produced by the conjugate acid and determine pH.
Example: 20.0 mL of 0.100 M NH3 titrated with 0.100 M HCl.
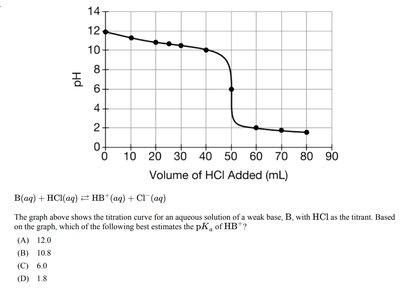
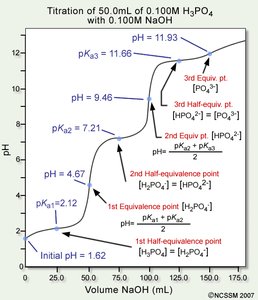
Polyprotic Acid Titrations
Polyprotic acids, such as H3PO4, undergo multiple deprotonations, resulting in multiple equivalence points and buffer regions in their titration curves.
Each equivalence point: Corresponds to the neutralization of one proton.
Half-equivalence points: Occur between equivalence points, where the concentration of acid and its conjugate base are equal.
pH Calculation: At each half-equivalence point, pH = pKa for that proton.
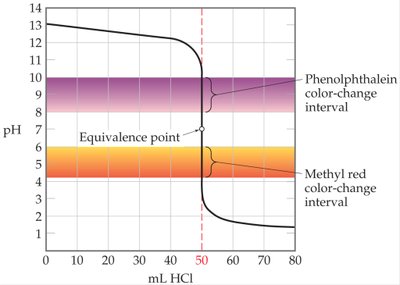
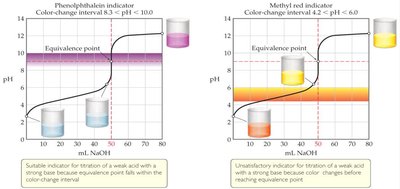
Choosing Indicators for Acid-Base Titrations
Indicators are weak acids that change color depending on the pH of the solution. The choice of indicator depends on the expected pH at the equivalence point.
Indicator Range: Each indicator has a specific pH range over which it changes color.
Equivalence Point Detection: The indicator should change color in the region where the pH changes rapidly.
Example: Phenolphthalein is suitable for weak acid-strong base titrations, while methyl red is not.

Summary Table: Key Features of Titration Curves
Titration Type | Initial pH | Buffer Region | Equivalence Point pH | Indicator Choice |
|---|---|---|---|---|
Strong Acid-Strong Base | Low | None | 7 | Phenolphthalein, Methyl Red |
Weak Acid-Strong Base | Moderate | Present | >7 | Phenolphthalein |
Weak Base-Strong Acid | High | Present | <7 | Methyl Red |
Polyprotic Acid | Low | Multiple | Multiple | Depends on equivalence point |
Additional info:
ICE tables (Initial, Change, Equilibrium) are commonly used for calculating pH in titration problems.
For polyprotic acids, each deprotonation step has its own Ka and equivalence point.
Indicators must be chosen based on the expected pH at the equivalence point, not just the initial or final pH.
