 Back
BackAlcohols, Phenols, Thiols, and Ethers: Structure, Nomenclature, and Reactions
Study Guide - Smart Notes

Ch.13 Alcohols, Phenols, Thiols, and Ethers
Concept: Naming Alcohols
Alcohols are organic compounds characterized by the presence of a hydroxyl (OH) group attached to an sp3 hybridized carbon atom. The systematic naming of alcohols follows rules similar to those for alkanes, with modifications to indicate the functional group.
Key Point 1: The parent chain must include the OH group and be the longest possible chain containing it.
Key Point 2: The suffix of the parent alkane is changed from -e to -ol to indicate the alcohol functional group.
Key Point 3: Number the chain from the end nearest the OH group; if a tie occurs, use the next closest substituent or lowest alphabetical order.
Key Point 4: Assign numbers to indicate the position of the OH group and other substituents.
Example: 2-chloro-3-hydroxyhexane (where the OH group is on the third carbon and a Cl is on the second carbon).
Additional info: The IUPAC system ensures clarity in naming, especially for compounds with multiple substituents or functional groups.
Alcohol Classification
Alcohols are classified based on the number of alkyl groups attached to the carbon bearing the OH group:
Primary (1°) Alcohol: The carbon with the OH group is attached to one other carbon.
Secondary (2°) Alcohol: The carbon with the OH group is attached to two other carbons.
Tertiary (3°) Alcohol: The carbon with the OH group is attached to three other carbons.
Example: Ethanol is a primary alcohol; isopropanol is a secondary alcohol; tert-butanol is a tertiary alcohol.
Concept: Naming Thiols
Thiols (mercaptans) are organic compounds containing a sulfhydryl (SH) group attached to a carbon atom. The naming rules for thiols are similar to those for alcohols, with the suffix -thiol added to the parent chain name.
Key Point 1: The parent chain must include the SH group and be the longest possible chain containing it.
Key Point 2: Number the chain from the end nearest the SH group.
Key Point 3: Assign numbers to indicate the position of the SH group and other substituents.
Example: 2-bromopentanethiol (where the SH group is on the first carbon and Br is on the second carbon).
Concept: Naming Ethers
Ethers are compounds in which an oxygen atom is bonded to two alkyl or aryl groups. Ethers have both common and IUPAC naming systems.
Common Naming: Name the two alkyl groups alphabetically followed by the word "ether" (e.g., methyl propyl ether).
IUPAC Naming: The smaller alkyl group is named as an alkoxy substituent attached to the parent chain (e.g., methoxypropane).
Key Point: If both alkyl groups are identical, use a numerical prefix (e.g., diethyl ether).
Example: CH3CH2OCH3 is called ethyl methyl ether (common) or methoxyethane (IUPAC).
Concept: Alcohol Reactions – Dehydration Reactions
Alcohols can undergo dehydration reactions in the presence of a strong acid (such as H2SO4), resulting in the formation of an alkene and water.
Key Point 1: The OH group is removed from the alcohol carbon, and a hydrogen atom is removed from an adjacent carbon.
Key Point 2: Zaitsev’s Rule applies: the hydrogen is removed from the neighboring carbon with the greatest number of hydrogens, favoring the formation of the more substituted (stable) alkene.
Equation:
Example: Dehydration of 2-butanol yields 2-butene as the major product.
Concept: Introduction to Redox Reactions
Redox (oxidation-reduction) reactions in organic chemistry involve changes in the number of oxygen or hydrogen atoms bonded to a molecule.
Oxidation: Increases the number of C–O bonds (or decreases C–H bonds).
Reduction: Increases the number of C–H bonds (or decreases C–O bonds).
Example: Oxidation of a primary alcohol to an aldehyde, then to a carboxylic acid.
Concept: Alcohol Reactions – Oxidation Reactions
Alcohols can be oxidized using agents such as potassium dichromate (K2Cr2O7) in sulfuric acid. The product depends on the type of alcohol:
Primary Alcohols: Oxidized to aldehydes, then to carboxylic acids.
Secondary Alcohols: Oxidized to ketones.
Tertiary Alcohols: Generally do not undergo oxidation under these conditions.
Equation:
Example: Oxidation of ethanol yields acetaldehyde, then acetic acid.
Concept: Reactions of Thiols
Thiols undergo oxidation to form disulfides, and reduction of disulfides regenerates thiols. This redox process is important in biological systems, such as protein structure.
Key Point 1: Two moles of thiol react to form one mole of disulfide.
Key Point 2: S–H bonds are broken, and an S–S bond is formed during oxidation.
Equation:
Reduction: Disulfides can be reduced back to thiols.
Example: Oxidation of two molecules of ethanethiol yields diethyl disulfide.

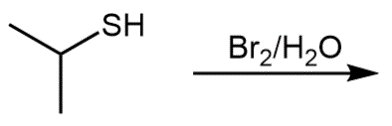
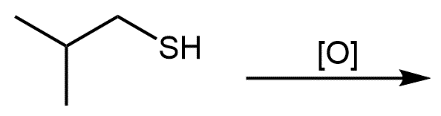
Additional info: Disulfide bonds are crucial for the tertiary and quaternary structure of proteins, stabilizing their folded forms.
