 Back
BackAtoms and Elements: Structure, Properties, and Periodicity
Study Guide - Smart Notes

Atoms and Elements
Definition and Nature of Elements
Elements are pure substances from which all other matter is constructed. They cannot be broken down into simpler substances by chemical means. Each element is composed of atoms, which are the smallest units retaining the chemical properties of the element.
Example: Sulfur is an element, as shown below.

Naming and Symbols of Elements
Element names often derive from Greek, Latin, or German words, locations, or are named in honor of scientists. Each element is represented by a one- or two-letter chemical symbol; the first letter is always capitalized, and the second (if present) is lowercase.
Examples: Co (cobalt), C (carbon), Si (silicon), S (sulfur), Ne (neon), N (nitrogen), He (helium), H (hydrogen).

Common Elements and Their Symbols
Below is a table of common elements and their chemical symbols:
Symbol | Name | Symbol | Name |
|---|---|---|---|
Al | Aluminum | Cu | Copper |
Ar | Argon | F | Fluorine |
As | Arsenic | Ga | Gallium |
Ba | Barium | Au | Gold |
B | Boron | He | Helium |
Br | Bromine | H | Hydrogen |
Cd | Cadmium | I | Iodine |
Ca | Calcium | Fe | Iron |
C | Carbon | Pb | Lead |
Cl | Chlorine | Li | Lithium |
Cr | Chromium | Mg | Magnesium |
Co | Cobalt | Mn | Manganese |
The Periodic Table
Organization and Groups
The periodic table, created by Dmitri Mendeleev, arranges elements into groups (vertical columns) and periods (horizontal rows) based on similar properties. Groups are numbered 1A to 8A (or 1 to 18), and periods are numbered from top to bottom.
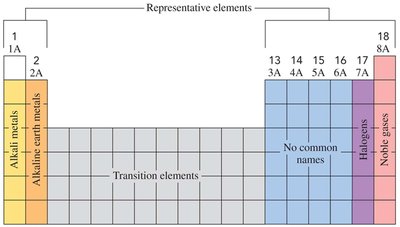
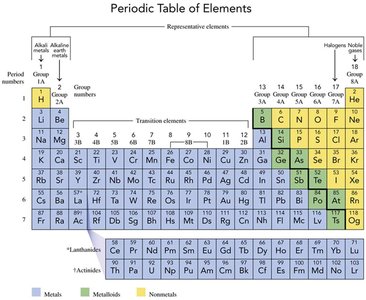
Metals, Nonmetals, and Metalloids
Elements are classified as metals, nonmetals, or metalloids based on their physical and chemical properties:
Metals: Shiny, ductile, malleable, and good conductors of electricity (e.g., copper, gold, silver).
Nonmetals: Dull, brittle, poor conductors of heat and electricity (e.g., hydrogen, carbon, nitrogen, oxygen, chlorine, sulfur).
Metalloids: Exhibit properties intermediate between metals and nonmetals; better conductors than nonmetals but not as good as metals.
Atomic Structure
Dalton’s Atomic Theory
Dalton proposed that elements are composed of tiny, indivisible particles called atoms. Atoms of the same element are identical, and atoms of different elements combine in fixed ratios to form compounds. However, later discoveries showed that atoms are divisible and can have isotopes.
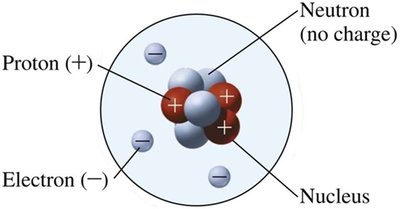
Subatomic Particles
Atoms are composed of three main subatomic particles:
Particle | Location | Mass (amu) | Charge | Symbol |
|---|---|---|---|---|
Proton | Nucleus | 1.007 | +1 | p or p+ |
Neutron | Nucleus | 1.008 | 0 | n or n0 |
Electron | Outside nucleus | 0.00055 | -1 | e- |
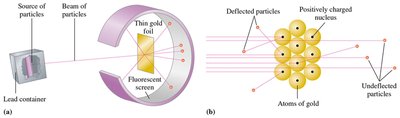
Rutherford’s Gold Foil Experiment
Rutherford’s experiment demonstrated that atoms have a small, dense, positively charged nucleus surrounded by electrons in mostly empty space.
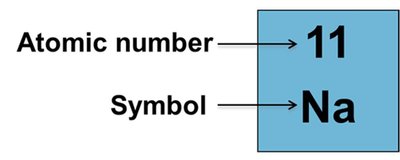
Atomic Number and Mass Number
Atomic Number (Z): Number of protons in the nucleus; determines the identity of the element.
Mass Number (A): Total number of protons and neutrons in the nucleus.
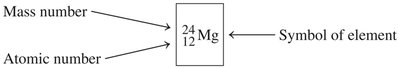
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, and thus different mass numbers. The atomic symbol notation shows the element, mass number (superscript), and atomic number (subscript):
Example: , ,

Electromagnetic Radiation and Atomic Spectra
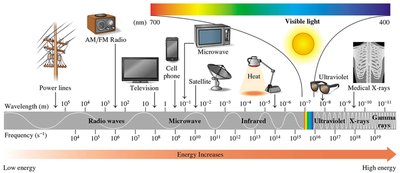
Electromagnetic Spectrum
Electromagnetic radiation includes radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, and gamma rays. All travel as waves, and the energy increases as wavelength decreases.
Atomic Spectrum
When light from a heated element passes through a prism, it separates into distinct lines called an atomic spectrum. Each element has a unique atomic spectrum, which is used for identification.

Electron Energy Levels
Electrons in atoms occupy specific energy levels, designated by principal quantum numbers (n = 1, 2, 3, ...). Energy increases with increasing n, and electrons farther from the nucleus have higher energy.

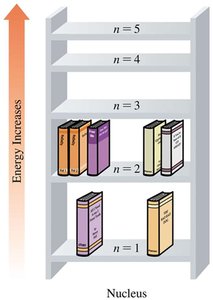
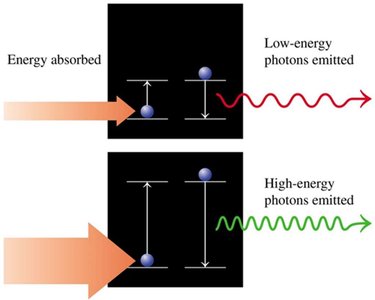
Electron Transitions
When electrons absorb energy, they move to higher energy levels; when they emit energy, they fall to lower levels. The energy change corresponds to the difference between the two levels.
Electron Configuration and Valence Electrons
Electron Configuration
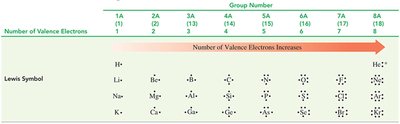
The arrangement of electrons in an atom is called its electron configuration. The outermost electrons, or valence electrons, determine chemical properties. For representative elements, the group number equals the number of valence electrons.
Lewis Symbols
Lewis symbols represent valence electrons as dots around the element symbol. This notation helps visualize bonding and reactivity.

Periodic Trends
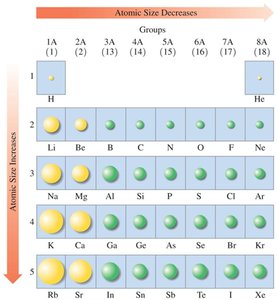
Atomic Size
Atomic size increases down a group and decreases across a period from left to right. This trend is due to the increasing number of energy levels and the increasing nuclear charge.
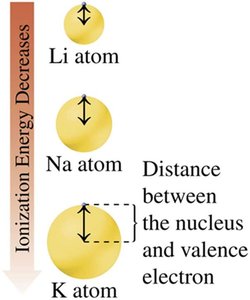
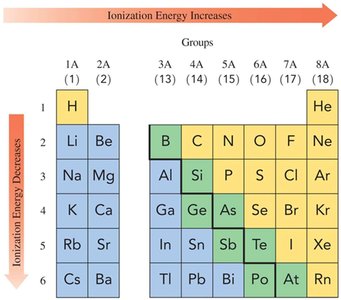
Ionization Energy
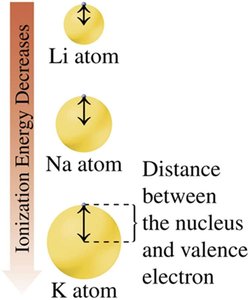
Ionization energy is the energy required to remove an outermost electron from an atom. It decreases down a group and increases across a period from left to right.
Equation:
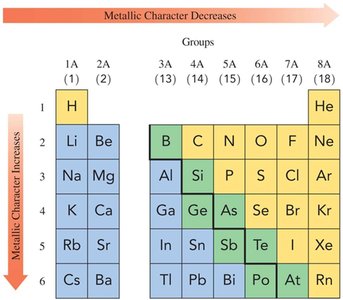
Metallic Character
Metallic character refers to the tendency of an element to lose valence electrons. It increases down a group and decreases across a period from left to right.
Summary Table: Periodic Trends
Trend | Down a Group | Across a Period (Left to Right) |
|---|---|---|
Atomic Size | Increases | Decreases |
Ionization Energy | Decreases | Increases |
Metallic Character | Increases | Decreases |
Additional info: These notes cover the foundational concepts of atomic structure, periodicity, and the classification of elements, which are essential for understanding chemical reactivity and bonding in general, organic, and biological chemistry.
