 Back
BackCarbohydrates and Chirality: Structure, Classification, and Biological Significance
Study Guide - Smart Notes

Carbohydrates: Structure and Classification
Overview of Carbohydrates
Carbohydrates are a major source of energy in the human diet and are composed of carbon, hydrogen, and oxygen. They are also known as saccharides or sugars, produced by plants during photosynthesis and oxidized in living cells to release energy.
Key Elements: C, H, O
Examples: Glucose, Fructose, Galactose, Ribose, Deoxyribose
Function: Energy production, structural roles in cells
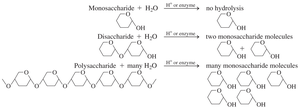
Types of Carbohydrates
Carbohydrates are classified based on the number of sugar units:
Monosaccharides: Simplest carbohydrates, single sugar unit
Disaccharides: Composed of two monosaccharides
Polysaccharides: Composed of many monosaccharides
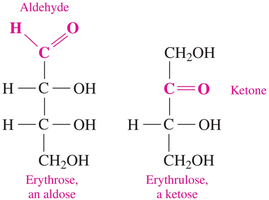
Monosaccharides: Structure and Classification
Monosaccharides contain 3-7 carbon atoms, with one carbon in a carbonyl group. They are further classified as aldoses (aldehyde group) or ketoses (ketone group). Each carbon, except the carbonyl carbon, has a hydroxyl group.
Triose: 3 carbon atoms
Tetrose: 4 carbon atoms
Pentose: 5 carbon atoms
Hexose: 6 carbon atoms
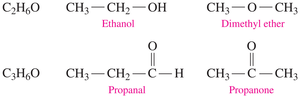
Isomerism in Carbohydrates
Structural Isomers
Structural isomers have the same molecular formula but different bonding arrangements. This concept is important in organic chemistry for distinguishing molecules with identical formulas but different properties.
Stereoisomers
Stereoisomers have identical molecular formulas and bonding sequences but differ in spatial arrangement. They are not structural isomers. When mirror images cannot be superimposed, the molecules are chiral.

Chirality and Chiral Carbon Atoms
Chirality in Molecules
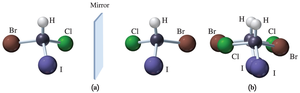
A molecule is chiral if it has a nonsuperimposable mirror image, often due to the presence of a carbon atom bonded to four different groups. Everyday objects like hands are chiral, as their mirror images cannot be superimposed.

Chiral Carbon Atoms
Chiral carbon atoms are central to chirality in organic molecules. A carbon atom bonded to four different groups is chiral, resulting in nonsuperimposable mirror images called enantiomers.
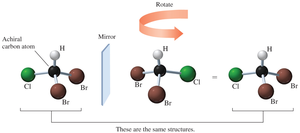
Achiral Carbon Atoms
Achiral molecules have mirror images that are superimposable. If a carbon atom is bonded to at least two identical groups, it is achiral.
Fischer Projections and D/L Notation
Drawing Fischer Projections
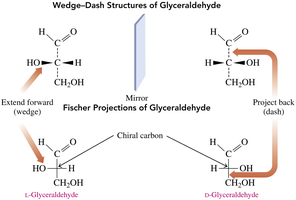
Fischer projections are 2D representations of 3D molecules, commonly used for carbohydrates. The most oxidized carbon is placed at the top, vertical lines represent bonds going back, and horizontal lines represent bonds coming forward.
D and L Notations
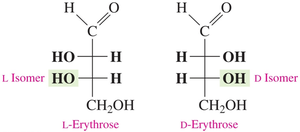
D and L isomers are assigned based on the position of the –OH group on the chiral carbon farthest from the carbonyl carbon. L is assigned if the –OH is on the left, D if it is on the right.
Biological Significance of Enantiomers
Enantiomers in Biology
Enantiomers can have dramatically different biological effects. For example, L-dopa is used to treat Parkinson’s disease, while its enantiomer D-dopa is ineffective.
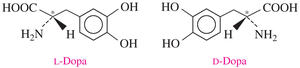
Monosaccharides: Fischer Projections and Haworth Structures
D and L Isomers of Monosaccharides
Common monosaccharides such as glucose, galactose, and fructose can be represented in Fischer projections. The D/L configuration is determined by the –OH group on the chiral carbon farthest from the carbonyl group.
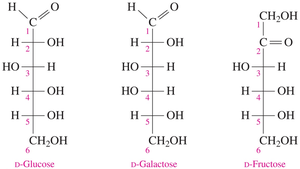
Glucose and Fructose
D-glucose: Aldohexose, formula C6H12O6, found in fruits, syrups, honey, and blood
D-fructose: Ketohexose, formula C6H12O6, sweetest carbohydrate

Galactose
D-galactose: Aldohexose, formula C6H12O6, obtained from lactose
Galactosemia: Genetic disorder where the enzyme to convert galactose to glucose is missing
Haworth Structures: Cyclic Forms of Monosaccharides
Formation of Haworth Structures
Pentose and hexose sugars form stable five- or six-membered rings called Haworth structures. These rings are produced by the reaction of a carbonyl group with a hydroxyl group in the same molecule.
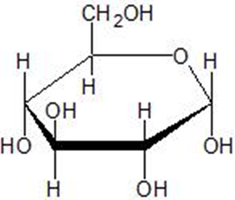
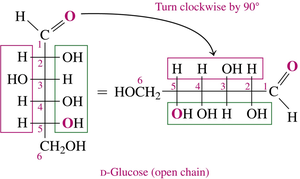
Drawing Cyclic Structures for Glucose
To convert a Fischer projection to a Haworth structure:
Turn the Fischer projection clockwise by 90°
Fold to make a hexagon, rotate groups on carbon 5 so the –OH is close to carbon 1
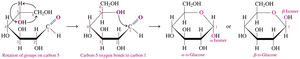
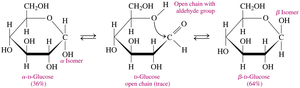
Alpha and Beta Isomers
When forming the ring, the new –OH group on carbon 1 can be below the ring (α isomer) or above the ring (β isomer).
Additional info: The notes cover the classification, structure, and biological relevance of carbohydrates, focusing on chirality and isomerism, which are essential for understanding their function in biochemistry and metabolism.
