Back
BackCarbohydrates, Lipids, and Proteins: Structure, Function, and Biological Importance
Study Guide - Smart Notes

Carbohydrates
Introduction to Carbohydrates
Carbohydrates are a major source of energy in the human diet and are composed of carbon, hydrogen, and oxygen. They are also known as saccharides, meaning "sugars." Carbohydrates are produced by plants through photosynthesis and are oxidized in living cells to provide energy.
Key Elements: Carbon (C), Hydrogen (H), Oxygen (O)
Dietary Sources: Bread, potatoes, pasta

Photosynthesis Equation:
Cellular Respiration Equation:

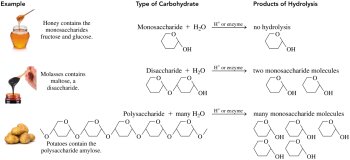
Types of Carbohydrates
Carbohydrates are classified based on the number of sugar units:
Monosaccharides: The simplest carbohydrates (single sugar units)
Disaccharides: Composed of two monosaccharide units
Polysaccharides: Composed of many monosaccharide units
Monosaccharides
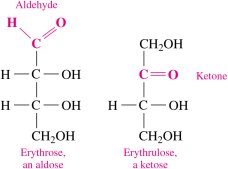
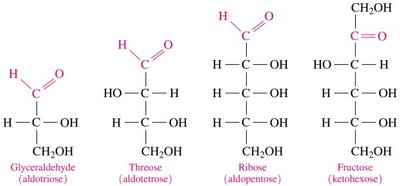
Monosaccharides contain several hydroxyl groups attached to a chain of three to eight carbon atoms. They are further classified by their functional group and the number of carbon atoms:
Aldoses: Contain an aldehyde group
Ketoses: Contain a ketone group
Classification by Carbon Number: Triose (3C), Tetrose (4C), Pentose (5C), Hexose (6C)
Chemical Properties of Monosaccharides
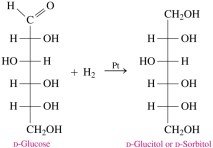
Monosaccharides can be reduced to sugar alcohols, which are used as sweeteners in sugar-free products. For example, D-glucose can be reduced to D-glucitol (sorbitol).
Sugar Alcohols: D-sorbitol, D-xylitol, D-mannitol
Applications: Diet drinks, sugarless gum, diabetic products
Disaccharides
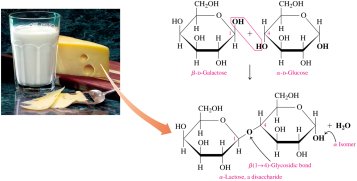
Disaccharides are carbohydrates composed of two monosaccharide units joined by a glycosidic bond. Common examples include lactose and maltose.
Lactose: Found in milk; composed of galactose and glucose
Maltose: Found in cereals and candies; composed of two glucose units

Polysaccharides
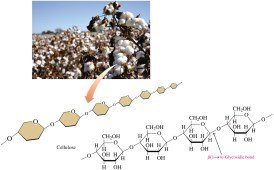
Polysaccharides are large molecules formed by joining many monosaccharide units. They serve as energy storage or structural components in living organisms.
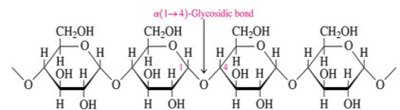
Cellulose: Composed of glucose units connected by β(1→4) glycosidic bonds; structural component in plants
Starch: Storage form of glucose in plants; consists of amylose (unbranched) and amylopectin (branched)
Glycogen: Storage form of glucose in animals
Lipids
Introduction to Lipids
Lipids are biomolecules that contain fatty acids or a steroid nucleus. They are soluble in organic solvents but not in water. Lipids are essential for cell membranes, fat-soluble vitamins, and steroid hormones.
Types of Lipids: Waxes, triacylglycerols, glycerophospholipids, sphingolipids, steroids
Hydrolyzable Lipids: Contain ester bonds and can be broken down to fatty acids
Nonhydrolyzable Lipids: Steroids, which do not contain fatty acids
Fatty Acids
Fatty acids are long, unbranched carbon chains with a carboxylic acid group at one end. They are classified as saturated (no double bonds) or unsaturated (one or more double bonds).
Saturated Fatty Acids: Pack closely together, leading to higher melting points
Unsaturated Fatty Acids: Have kinks due to double bonds, preventing tight packing

Essential Fatty Acids
Essential fatty acids, such as linoleic acid, linolenic acid, and arachidonic acid, cannot be synthesized by the human body and must be obtained from the diet.
Waxes and Triacylglycerols
Waxes are esters of long-chain fatty acids and long-chain alcohols, providing protective coatings for plants and animals. Triacylglycerols (triglycerides) are the main storage form of fatty acids in the body, formed by esterification of glycerol with three fatty acids.


Chemical Properties of Triacylglycerols
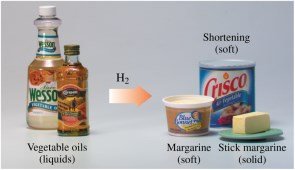
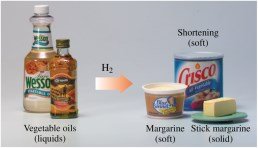
Triacylglycerols can undergo hydrogenation, where double bonds in unsaturated fatty acids react with hydrogen gas to form saturated fatty acids. This process is used to convert vegetable oils into margarine and shortening.
Hydrogenation: Increases saturation and melting point
Partial Hydrogenation: Produces soft, semisolid fats
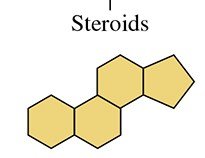
Steroids: Cholesterol, Bile Salts, and Steroid Hormones
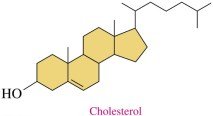
Steroids are lipids with a characteristic structure of four fused rings. Cholesterol is the most important steroid in the body, essential for cell membranes, hormone synthesis, and vitamin D production. Excess cholesterol can lead to plaque formation in arteries, increasing the risk of heart attack.
Cholesterol: Obtained from diet and synthesized in the liver
Bile Salts: Synthesized from cholesterol, aid in fat digestion
Steroid Hormones: Chemical messengers derived from cholesterol
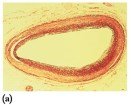
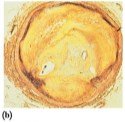
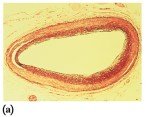
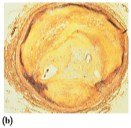

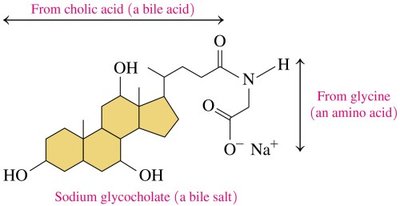
Steroid Hormones and Adrenal Corticosteroids
Steroid hormones include sex hormones (testosterone, estrogens, progesterone) and adrenal corticosteroids (aldosterone, cortisone). These hormones regulate metabolism, electrolyte balance, and other physiological functions.
Adrenal Corticosteroids: Produced by adrenal glands; regulate electrolytes and glucose levels
Aldosterone: Regulates water and electrolyte balance
Cortisone: Increases blood glucose and stimulates glycogen synthesis
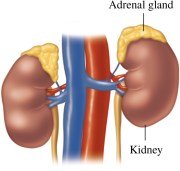
Proteins and Amino Acids
Introduction to Proteins
Proteins are large biomolecules made from 20 different amino acids. They serve a wide range of functions, including structural support, catalysis, transport, storage, regulation, and protection.
Structural Proteins: Collagen, keratin
Contractile Proteins: Myosin, actin
Transport Proteins: Hemoglobin, lipoproteins
Storage Proteins: Casein, ferritin
Hormonal Proteins: Insulin, growth hormone
Enzymes: Sucrase, trypsin
Protective Proteins: Immunoglobulins

Class of Protein | Function | Examples |
|---|---|---|
Structural | Provide structural components | Collagen, Keratin |
Contractile | Make muscles move | Myosin, Actin |
Transport | Carry essential substances | Hemoglobin, Lipoproteins |
Storage | Store nutrients | Casein, Ferritin |
Hormone | Regulate metabolism and nervous system | Insulin, Growth hormone |
Enzyme | Catalyze biochemical reactions | Sucrase, Trypsin |
Protection | Immune response | Immunoglobulins |
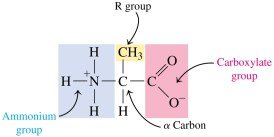
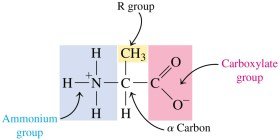
Amino Acids
Amino acids are the building blocks of proteins. Each amino acid contains a central (α) carbon atom bonded to an amino group, a carboxyl group, a hydrogen atom, and a unique side chain (R group). At physiological pH, amino acids exist as zwitterions, carrying both positive and negative charges but having an overall neutral charge.