 Back
BackChapter 17: Lipids – Structure, Properties, and Biological Roles
Study Guide - Smart Notes

Chapter 17: Lipids
Introduction to Lipids
Lipids are a diverse group of biomolecules that are characterized by their solubility in organic solvents and insolubility in water. They play crucial roles in biological systems, including energy storage, cell membrane structure, and signaling. Lipids are either based on fatty acids or contain a steroid nucleus.
Definition: Lipids are biomolecules containing fatty acids or a steroid nucleus.
Solubility: Soluble in organic solvents, not in water.
Biological Importance: Key components of cell membranes, fat-soluble vitamins, and steroid hormones.
Classification of Lipids
Lipids are classified based on their structure and hydrolysis products:
Hydrolyzable Lipids: Contain esters and can be hydrolyzed to yield fatty acids and other molecules (e.g., waxes, triacylglycerols, glycerophospholipids, sphingolipids).
Nonhydrolyzable Lipids: Do not contain fatty acids and cannot be hydrolyzed (e.g., steroids).
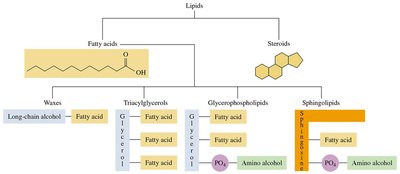
Types of Lipids
Fatty Acids: Long, unbranched carbon chains with a carboxylic acid group at one end. They can be saturated (no double bonds) or unsaturated (one or more double bonds).
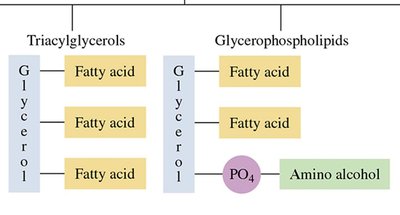
Triacylglycerols: Esters formed from glycerol and three fatty acids; main storage form of energy in animals.
Glycerophospholipids: Contain glycerol, two fatty acids, a phosphate group, and an amino alcohol; major components of cell membranes.
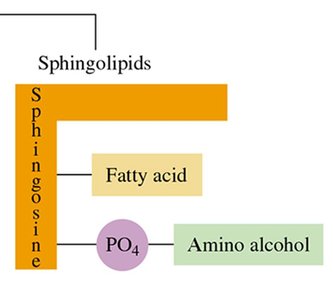
Sphingolipids: Contain sphingosine, a fatty acid, phosphate, and an amino alcohol; important in nerve cell membranes.
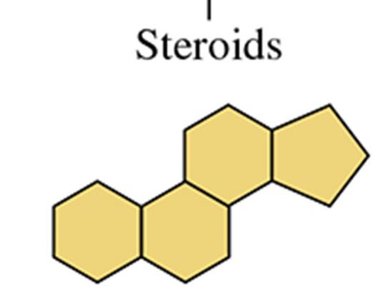
Steroids: Characterized by a four-ring structure; include cholesterol and steroid hormones.
17.2 Fatty Acids
Structure and Types of Fatty Acids
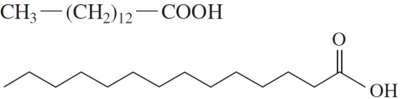
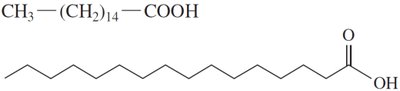
Fatty acids are the simplest lipids, typically containing 12–18 carbon atoms. Their properties depend on the presence and number of double bonds.
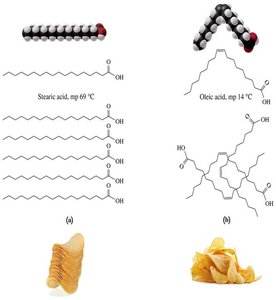
Saturated Fatty Acids: Only single C–C bonds; pack closely together, resulting in higher melting points and solid state at room temperature.
Unsaturated Fatty Acids: Contain one (monounsaturated) or more (polyunsaturated) C=C double bonds; have kinks that prevent tight packing, resulting in lower melting points and liquid state at room temperature.

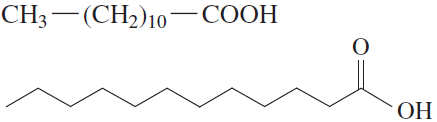
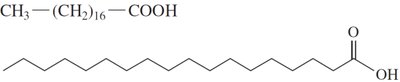
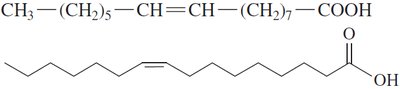
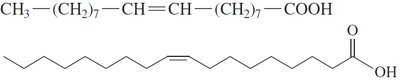
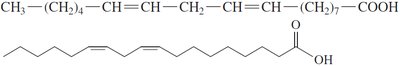
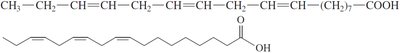
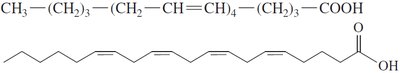
Cis and Trans Isomers
Unsaturated fatty acids can exist as cis or trans isomers. Most naturally occurring unsaturated fatty acids are in the cis form, which introduces a bend in the chain.
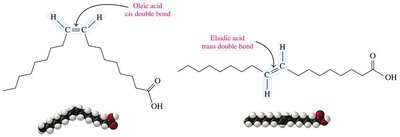
Physical Properties
Saturated fatty acids: Higher melting points, solid at room temperature.
Unsaturated fatty acids: Lower melting points, liquid at room temperature due to kinks from cis double bonds.
Essential Fatty Acids
Essential fatty acids (e.g., linoleic, linolenic, and arachidonic acids) cannot be synthesized by the human body and must be obtained from the diet.
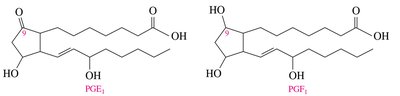
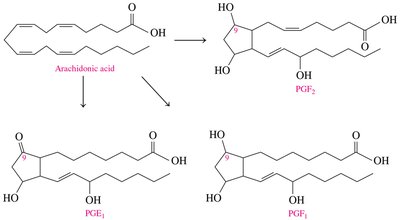
Prostaglandins
Prostaglandins are hormone-like substances derived from arachidonic acid. They regulate physiological processes such as inflammation, blood pressure, and smooth muscle contraction.
Omega Fatty Acids
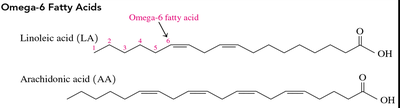
Omega-6 Fatty Acids: First double bond at the sixth carbon from the methyl end (e.g., linoleic acid, arachidonic acid).
Omega-3 Fatty Acids: First double bond at the third carbon from the methyl end (e.g., linolenic acid, EPA, DHA); found in fish oils and beneficial for cardiovascular health.
17.3 Waxes and Triacylglycerols
Waxes
Waxes are esters of long-chain fatty acids and long-chain alcohols. They serve as protective coatings in plants and animals.
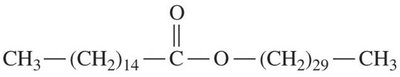
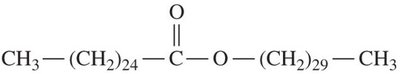
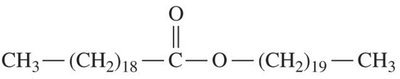
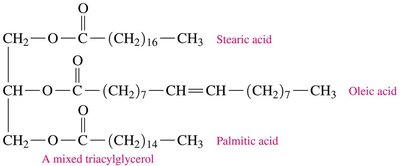
Triacylglycerols (Triglycerides)
Triacylglycerols are the main storage form of energy in animals. They are formed by esterification of glycerol with three fatty acids.
Simple triacylglycerols: Contain three identical fatty acids.
Mixed triacylglycerols: Contain two or three different fatty acids.

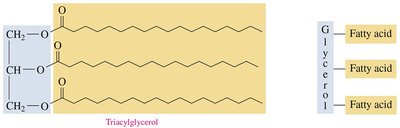
Energy Storage
Triacylglycerols are the major energy reserve in animals, especially important for hibernating species.
17.4 Chemical Properties of Triacylglycerols
Hydrogenation
Hydrogenation is the process of adding hydrogen to unsaturated fatty acids, converting double bonds to single bonds and increasing the melting point. Partial hydrogenation produces trans fats.
Hydrolysis
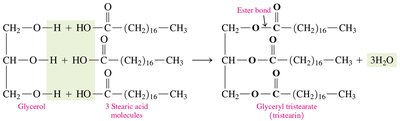
Hydrolysis of triacylglycerols in the presence of acid or enzymes (lipases) produces glycerol and fatty acids.
Saponification
Saponification is the reaction of a fat with a strong base (NaOH or KOH) to produce glycerol and the salts of fatty acids (soaps).
Summary Table of Lipid Reactions
Reaction | Reactants and Products |
|---|---|
Hydrogenation | Unsaturated fat + H2 (Ni catalyst) → Saturated fat |
Hydrolysis | Triacylglycerol + 3 H2O (enzyme/acid) → Glycerol + 3 fatty acids |
Saponification | Triacylglycerol + 3 NaOH (heat) → Glycerol + 3 soap molecules |
17.5 Phospholipids
Glycerophospholipids
Glycerophospholipids are major components of cell membranes, containing glycerol, two fatty acids, a phosphate group, and an amino alcohol (e.g., choline, serine, ethanolamine).

Sphingomyelin
Sphingomyelin contains sphingosine instead of glycerol and is abundant in the myelin sheath of nerve cells.

Structure and Polarity
Phospholipids have both polar (hydrophilic) heads and nonpolar (hydrophobic) tails, allowing them to form bilayers in cell membranes.
17.6 Steroids: Cholesterol, Bile Salts, and Steroid Hormones
Steroid Structure
Steroids have a core structure of four fused rings (three cyclohexane and one cyclopentane). Cholesterol is the most abundant steroid in the body.

Cholesterol
Obtained from diet and synthesized in the liver.
Essential for cell membranes, steroid hormone synthesis, and vitamin D production.
High levels can lead to plaque formation and cardiovascular disease.
Bile Salts
Bile salts are derived from cholesterol and aid in the digestion and absorption of fats by emulsifying them in the intestine.
Lipoproteins
Lipoproteins are complexes of lipids and proteins that transport nonpolar lipids through the bloodstream. Types include chylomicrons, VLDL, LDL, and HDL, differing in density and function.
Type | Density (g/mL) | Triacylglycerols (%) | Phospholipids (%) | Cholesterol (%) | Protein (%) |
|---|---|---|---|---|---|
Chylomicrons | 0.940 | 86 | 7 | 2 | 2 |
VLDL | 0.940–1.006 | 55 | 18 | 7 | 8 |
LDL | 1.006–1.063 | 6 | 22 | 8 | 22 |
HDL | 1.063–1.210 | 4 | 24 | 2 | 55 |
Steroid Hormones
Sex hormones: Testosterone, estrogens, progesterone.
Adrenal corticosteroids: Aldosterone (electrolyte balance), cortisone (glucose regulation).
17.7 Cell Membranes
Structure and Function
Cell membranes are composed of a lipid bilayer with embedded proteins and cholesterol. They are semipermeable, allowing selective transport of substances.
Fluid Mosaic Model: Describes the dynamic and flexible nature of the membrane, with proteins and lipids moving laterally within the layer.
Transport Mechanisms:
Diffusion: Passive movement from high to low concentration.
Facilitated Transport: Uses protein channels to increase diffusion rate.
Active Transport: Moves substances against a concentration gradient using energy.
Additional info: This summary covers the structure, classification, and biological roles of lipids, including their chemical properties, importance in health, and their role in cell membranes. It is suitable for exam preparation in a general, organic, and biological chemistry course.
