 Back
BackAdaptive Immunity: Mechanisms, Cells, and Clinical Implications
Study Guide - Smart Notes

Adaptive Immunity
Overview of Adaptive Immunity
Adaptive immunity is a highly specialized and specific defense mechanism that develops in response to exposure to foreign substances. It is characterized by recognition, response, and memory, allowing the immune system to mount stronger attacks upon repeated exposure to the same pathogen.
Recognition: Identifies anything foreign to the body.
Defense: Activates T and B lymphocytes to eliminate or neutralize foreign material.
Memory: Remembers previous invaders for a faster, more robust response upon re-exposure.
Key Characteristics:
Discrimination: Selectively responds to non-self antigens.
Specificity: T and B cells respond to specific antigens.
Diversity: Generates a vast array of receptors and antibodies.
Timing: Activated after the innate immune response.
Memory: Rapid and effective secondary response prevents illness.
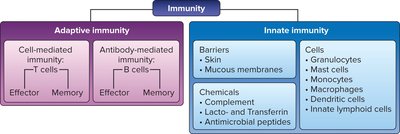
Types of Adaptive Immunity
Humoral (Antibody-Mediated) Immunity: B cells produce antibodies that neutralize or destroy extracellular pathogens and toxins.
Cell-Mediated Immunity: T cells (especially cytotoxic T lymphocytes) target and destroy infected or abnormal cells.
Antigens and Immunogenicity
Antigen Structure and Function
Antigens are substances that elicit an immune response. Most are complex molecules with specific regions called epitopes (antigenic determinant sites) to which antibodies bind. The valence of an antigen refers to the number of epitopes it possesses.
Antibody affinity: Strength of binding between an antibody and its specific epitope.
Avidity: Overall strength of binding between an antibody and an antigen with multiple epitopes.
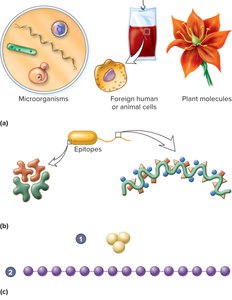
Haptens
Haptens are small organic molecules that are not immunogenic by themselves but become immunogenic when attached to a larger carrier protein (e.g., penicillin bound to serum proteins can trigger an allergic reaction).
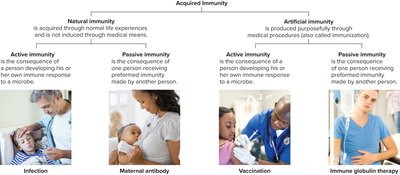
Acquired Immunity: Natural and Artificial
Types of Acquired Immunity
Naturally Acquired Active Immunity: Results from infection; the body produces its own antibodies.
Naturally Acquired Passive Immunity: Transfer of antibodies from mother to child (placenta or breast milk).
Artificially Acquired Active Immunity: Vaccination with antigens stimulates antibody production.
Artificially Acquired Passive Immunity: Injection of preformed antibodies or lymphocytes (e.g., antitoxins).
Recognition of Foreignness: The Major Histocompatibility Complex (MHC)
MHC Molecules
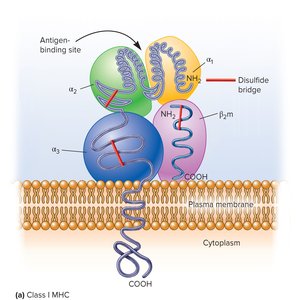
The major histocompatibility complex (MHC) is a group of genes encoding proteins that help the immune system distinguish self from non-self. In humans, these are called human leukocyte antigens (HLA) and are located on chromosome 6.
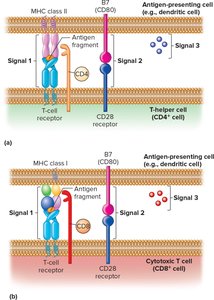
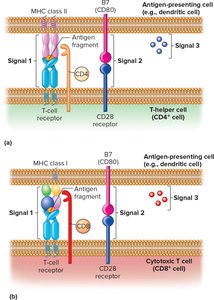
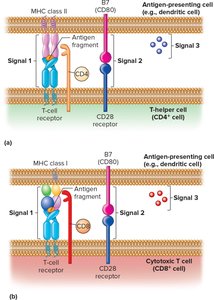
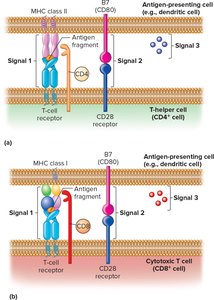
Class I MHC: Present on all nucleated cells; present endogenous antigens to CD8+ T cells.
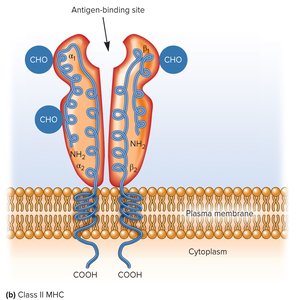
Class II MHC: Present on antigen-presenting cells (APCs) such as dendritic cells, macrophages, and B cells; present exogenous antigens to CD4+ T-helper cells.
Class III MHC: Encode secreted proteins with immune functions (not involved in antigen presentation).
Antigen Processing and Presentation
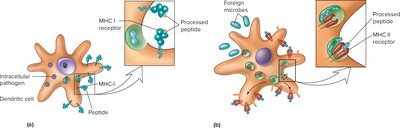
Endogenous pathway (Class I): Presents peptides from cytoplasmic proteins to CD8+ T cells.
Exogenous pathway (Class II): Presents peptides from extracellular proteins to CD4+ T-helper cells.
T Cells and Their Activation
T Cell Development and Selection
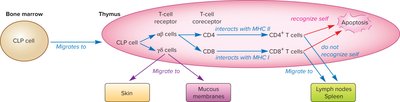
T cells develop in the thymus, where they undergo positive and negative selection to ensure self-tolerance and functional competence. T-cell receptors (TCRs) are heterodimeric proteins that recognize specific antigens presented by MHC molecules.
Positive selection: Ensures T cells can recognize self-MHC.
Negative selection: Eliminates T cells that react strongly to self-antigens.
T Cell Activation: Three Signals
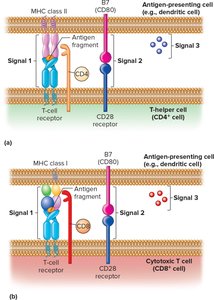
Signal 1: TCR binds to antigen-MHC complex on APC.
Signal 2: Costimulatory signal (e.g., B7 on APC binds CD28 on T cell); absence leads to anergy.
Signal 3: Cytokines released by APC and T cell direct differentiation into effector or memory cells.
Types of T Cells
Effector T cells: Actively respond to antigens (e.g., cytotoxic T lymphocytes, helper T cells).
Memory T cells: Persist long-term for rapid response upon re-exposure.
T Helper Cells (CD4+)
TH0: Naïve, undifferentiated T helper cells.
TH1: Promote cytotoxic T cell activity and activate macrophages.
TH2: Stimulate B cell antibody production.
TH17: Recruit neutrophils via IL-17 secretion.
Treg: Regulate immune response and prevent autoimmunity.
Cytotoxic T Lymphocytes (CD8+)
Destroy infected or abnormal host cells by releasing perforin and granzymes or activating apoptotic pathways (e.g., CD95).
B Cells and Antibody Production
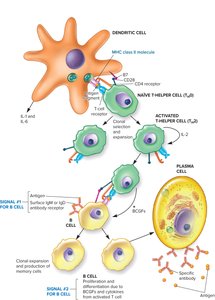
B Cell Activation
B cells mature in the bone marrow and are activated by antigen binding. They differentiate into plasma cells (antibody-secreting) and memory B cells.
T-dependent activation: Requires help from T cells; produces high-affinity antibodies and memory cells.
T-independent activation: Triggered by polymeric antigens (e.g., bacterial LPS); produces low-affinity antibodies, no memory.
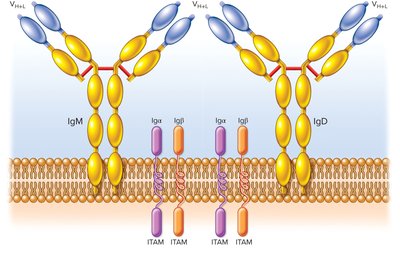
Antibody Structure and Function
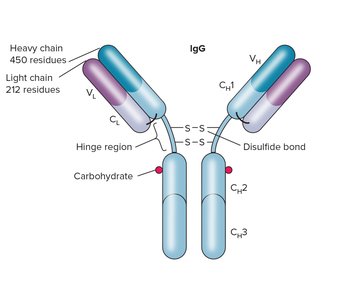
Antibody Structure
Antibodies (immunoglobulins, Ig) are Y-shaped glycoproteins composed of two identical heavy chains and two identical light chains, connected by disulfide bonds. Each antibody has:
Constant (C) region: Determines antibody class and effector function.
Variable (V) region: Forms the antigen-binding site; provides specificity.
Fab fragment: Binds antigen.
Fc fragment: Binds to immune cell receptors and complement proteins.
Classes of Immunoglobulins
Class | Main Functions |
|---|---|
IgG | Most abundant; crosses placenta; opsonization; neutralizes toxins/viruses |
IgM | First produced; pentamer; agglutination; complement activation |
IgA | Secreted at mucosal surfaces; found in breast milk |
IgD | Part of B cell receptor complex; signals B cell activation |
IgE | Allergic responses; defense against parasites |
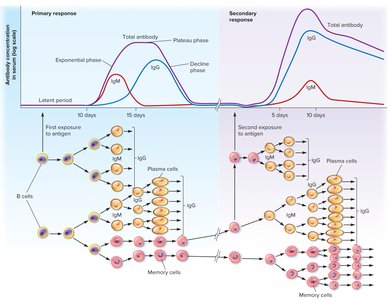
Antibody Kinetics
Primary response: Slow, with initial production of IgM followed by IgG.
Secondary response: Faster, stronger, and longer-lasting due to memory cells; mainly IgG.
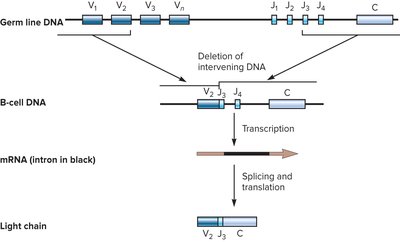
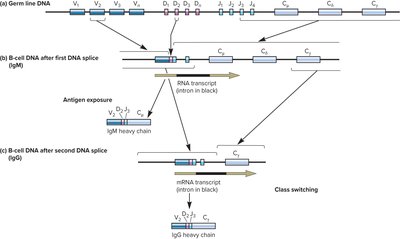
Generation of Antibody Diversity
Combinatorial joining: V, D, and J gene segments recombine to create diverse antibodies.
Splice site variability: Additional diversity from variable joining of gene segments.
Somatic hypermutation: Point mutations in variable regions increase diversity and affinity.
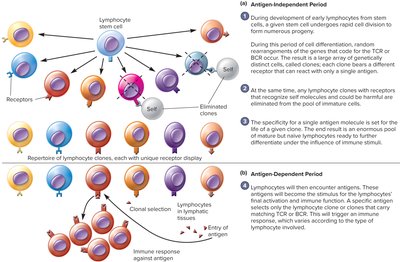
Clonal Selection Theory
Each B cell produces a unique antibody. Upon antigen encounter, only B cells with receptors specific to that antigen proliferate and differentiate into plasma and memory cells. Self-reactive cells are eliminated during development (negative selection).
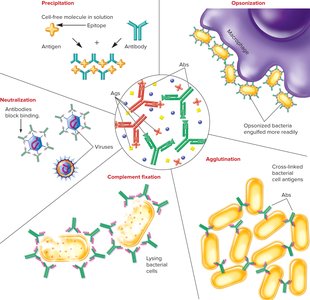
Antibody Effector Functions
Neutralization
Antibodies bind to pathogens or toxins, blocking their interaction with host cells and neutralizing their effects.
Opsonization
Antibodies (especially IgG and IgM) coat pathogens, enhancing their recognition and uptake by phagocytes.
Immune Complex Formation
Antibodies cross-link antigens, forming complexes that can precipitate or agglutinate, facilitating clearance but potentially causing tissue damage if excessive.
Immune System Malfunctions
Hypersensitivities
Type I (Immediate, Allergy): IgE-mediated; mast cell degranulation (e.g., hay fever, anaphylaxis).
Type II (Cytotoxic): IgG/IgM-mediated destruction of cells (e.g., transfusion reactions, erythroblastosis fetalis).
Type III (Immune Complex): Excess immune complexes cause inflammation (e.g., vasculitis, glomerulonephritis).
Type IV (Delayed-Type): T cell-mediated (e.g., contact dermatitis, tuberculin reaction).
Autoimmunity and Autoimmune Diseases
Autoimmunity involves the presence of antibodies or T cells that react with self-antigens. Autoimmune diseases result from the activation of self-reactive lymphocytes, causing chronic tissue damage.
Transplant Rejection and Graft-Versus-Host Disease
Allograft: Transplant between genetically different individuals of the same species.
Xenograft: Transplant between different species.
Host-versus-graft: Host immune system attacks transplanted tissue.
Graft-versus-host: Donor immune cells attack recipient tissues (e.g., after bone marrow transplant).
Immunodeficiencies
Primary (congenital): Genetic defects in immune function.
Acquired: Result from infections (e.g., HIV) or immunosuppressive treatments.
