 Back
Backlec 26:Adaptive Immunity: Mechanisms, Components, and Clinical Relevance
Study Guide - Smart Notes

Adaptive Immunity: Overview
Definition and Core Functions
Adaptive immunity is a highly specific, acquired defense system in vertebrates that targets particular pathogens (antigens), eliminates them, and retains memory for future protection. Unlike innate immunity, adaptive immunity utilizes B cells and T cells to generate a tailored response, providing long-lasting and highly specific defense.
Antibody production: B cells generate antibodies to neutralize pathogens (humoral immunity).
Cell-mediated killing: T cells destroy infected cells and coordinate immune responses.
Immunological memory: Faster, stronger response upon re-exposure to the same pathogen.
Key Components and Mechanisms
Lymphocyte Development and Differentiation
Lymphocytes, the core cells of adaptive immunity, originate from stem cells in the bone marrow. They differentiate into B cells (mature in bone marrow) and T cells (mature in thymus), each with specialized roles in immune defense.
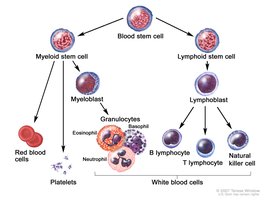
B Cells and Humoral Immunity
B cells mature in the bone marrow and are responsible for humoral immunity. They produce antibodies that directly neutralize antigens in body fluids.
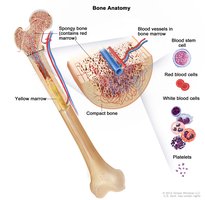
T Cells and Cell-Mediated Immunity
T cells mature in the thymus and are essential for cell-mediated immunity. They kill infected cells and coordinate the overall immune response.
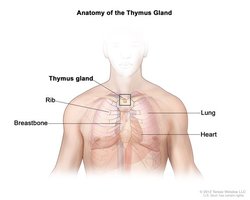
Antigen Recognition and Immune Memory
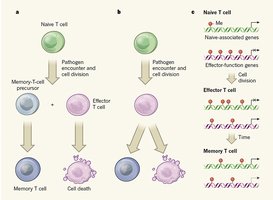
Adaptive immunity relies on the ability to distinguish self from non-self, targeting specific antigens on pathogens. Upon infection, some T and B cells become memory cells, providing long-term protection against future infections by the same pathogen.
Main Features of Adaptive Immunity
Specificity, Versatility, Memory, and Tolerance
Specificity: Antibodies and receptors are uniquely designed to bind a specific antigen, similar to a lock and key.
Versatility: The system can recognize and respond to millions of different antigens.
Memory: Previous infections are remembered, enabling rapid and robust responses upon re-exposure.
Tolerance: The immune system differentiates self from non-self, preventing attacks on the body's own tissues.
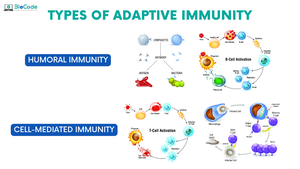
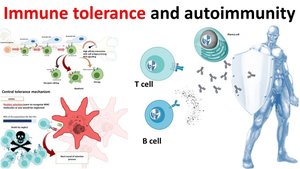
The Adaptive Immune Response Process
Stages of the Adaptive Response
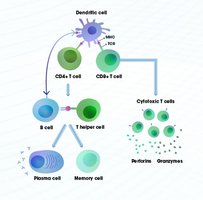
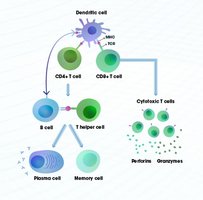
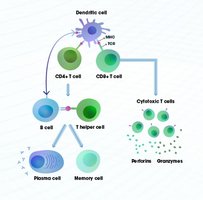
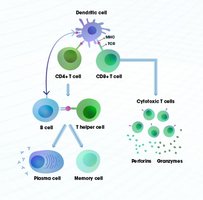
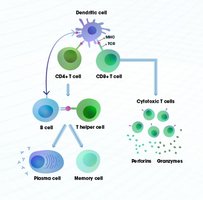
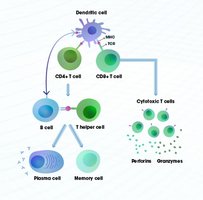
Antigen Detection: Dendritic cells capture antigens and present them to T cells in lymph nodes.
Activation: T cells are activated and subsequently activate B cells.
Expansion and Differentiation: B cells proliferate and differentiate into plasma cells that secrete antibodies.
Targeted Destruction: Antibodies and T cells eliminate the pathogen.
Memory Formation: Long-lived memory B and T cells remain for future protection.
Antibody Structure and Synthesis
Antibody Structure
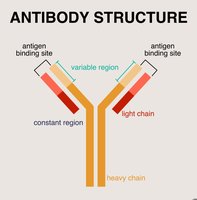
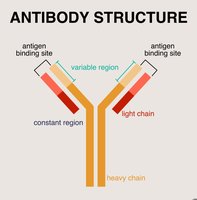
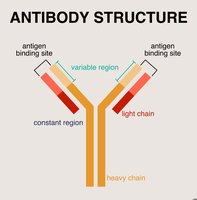
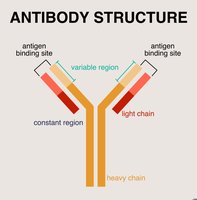
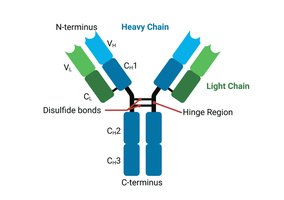
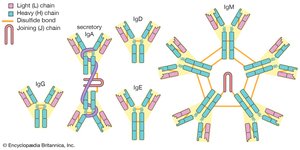
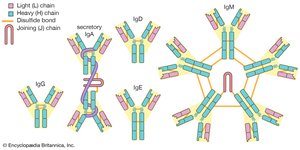
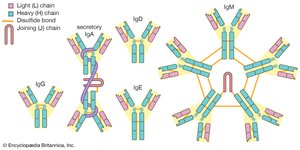
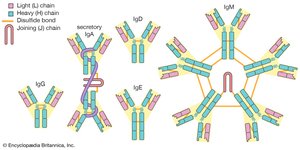
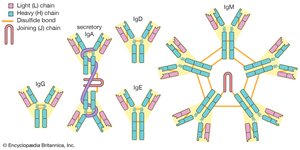
Antibodies (immunoglobulins) are Y-shaped proteins produced by B cells. Each antibody consists of four polypeptide chains (two heavy and two light) held together by disulfide bonds. The variable region at the tips of the Y forms the antigen-binding site, while the constant region determines the antibody's class and function.
Synthesis and Secretion
Activation: Naïve B cells encounter their specific antigen and receive help from Helper T cells.
Differentiation: Activated B cells proliferate and mature into plasma cells, which are specialized for antibody production.
Production: Plasma cells can secrete thousands of antibody molecules per second.
Class Switching: B cells initially produce IgM, but can switch to other classes (IgG, IgA, etc.) depending on the immune context.
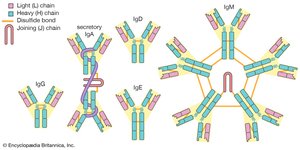
Classes of Antibodies (Immunoglobulins)
Overview of Antibody Isotypes
Class | Main Function | Structure |
|---|---|---|
IgG | Opsonization, neutralization, secondary response | Monomer |
IgM | Agglutination, complement activation, primary response | Pentamer |
IgA | Mucosal defense, found in secretions | Dimer (secretory form) |
IgE | Allergic responses, binds mast cells/basophils | Monomer |
IgD | B cell receptor, rarely secreted | Monomer |
Primary vs. Secondary Immune Response
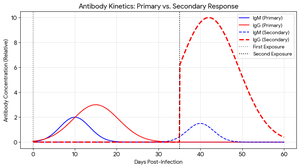
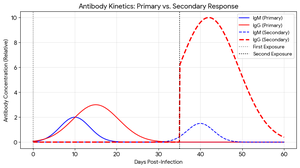
Antibody Kinetics During Infection
Upon first exposure to a pathogen, the adaptive immune system produces a primary response characterized by a slow rise in IgM, followed by IgG. Upon subsequent exposures, the secondary response is faster and more robust, dominated by high-affinity IgG antibodies.
Coordination Between Humoral and Cellular Immunity
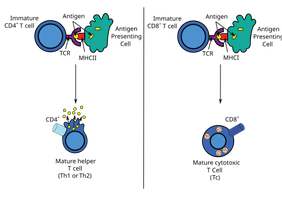
Role of Helper T Cells (CD4+)
Helper T cells act as coordinators, linking the cellular and humoral branches of adaptive immunity. They recognize antigens presented by antigen-presenting cells (APCs) and provide essential signals (cytokines) to activate B cells and promote antibody production, class switching, and memory formation.
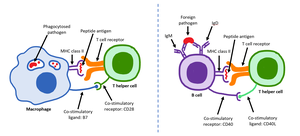
Cytotoxic T Cells (CD8+)
Cytotoxic T cells are responsible for killing infected or abnormal cells by inducing apoptosis. They recognize antigens presented on MHC Class I molecules and are crucial for controlling intracellular infections, such as viruses.
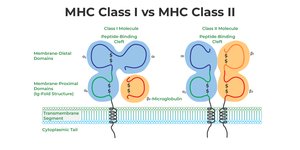
Major Histocompatibility Complex (MHC)
Structure and Function
MHC molecules (called HLA in humans) are cell surface glycoproteins that present peptide fragments to T cells, enabling the immune system to distinguish self from non-self. There are two main classes:
MHC Class I: Present on all nucleated cells; present endogenous antigens to CD8+ cytotoxic T cells.
MHC Class II: Present on antigen-presenting cells (APCs); present exogenous antigens to CD4+ helper T cells.
Key Concepts in MHC Biology
MHC Restriction: T cells recognize antigens only when presented by self-MHC molecules.
Extreme Polymorphism: High genetic diversity ensures population-level protection against diverse pathogens.
Codominant Expression: Both maternal and paternal MHC alleles are expressed, increasing antigen presentation diversity.
Cross-Presentation: Dendritic cells can present exogenous antigens on MHC Class I to activate cytotoxic T cells.
Clinical Relevance: MHC molecules are central to organ transplantation compatibility and are linked to autoimmune diseases.
Summary Table: Adaptive Immunity Key Points
Component | Origin/Maturation | Main Function |
|---|---|---|
B cell | Bone marrow | Antibody production (humoral immunity) |
T cell | Thymus | Cell-mediated immunity, coordination |
Antibody | B cell/plasma cell | Neutralization, opsonization, complement activation |
MHC I | All nucleated cells | Present to CD8+ T cells |
MHC II | APCs | Present to CD4+ T cells |
Additional info: This guide covers the essential mechanisms, cell types, and clinical relevance of adaptive immunity, suitable for exam preparation in a college-level microbiology course.
