 Back
BackAdaptive Immunity: Principles and Mechanisms
Study Guide - Smart Notes

Chapter 12
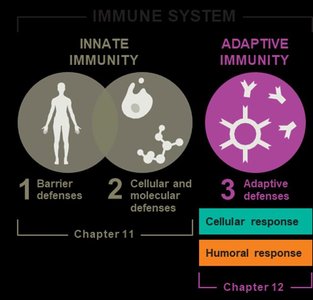
Adaptive Immunity
Overview of Adaptive Immunity
The adaptive immune response is the body's third and final line of defense, following innate immunity. It is highly specific, takes longer to mount, and provides immunological memory for rapid and effective responses upon re-exposure to the same antigen. Adaptive immunity interacts closely with innate immunity and is activated when innate defenses fail to contain a threat.
Specificity: Adaptive responses target particular antigens.
Immunological Memory: Secondary exposure results in rapid, effective elimination of pathogens, often preventing disease symptoms.
Branches of Adaptive Immunity
The adaptive immune system is divided into two interdependent branches: cellular (T cell-mediated) and humoral (antibody-mediated) responses. Both aim to eliminate identified antigens and remember them for faster future responses.
Cellular Response: Mediated by T cells.
Humoral Response: Mediated by B cells and antibodies.
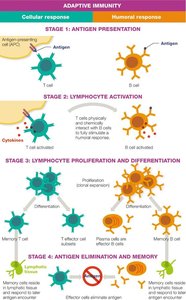
Stages of Adaptive Immune Response
Stage 1: Antigen Presentation
Antigen-presenting cells (APCs), such as dendritic cells and certain white blood cells, display antigens to T cells.
B cells can directly interact with antigens without APCs.
APCs: Present antigens to T cells via MHC molecules.
B Cells: Directly bind antigens.
Stage 2: Lymphocyte Activation
Lymphocytes are activated by cytokines after antigen presentation. Activated T cells influence B cell activation.
Cytokines: Signaling molecules that activate lymphocytes.
T Cell Influence: T cells help activate B cells.
Stage 3: Lymphocyte Proliferation and Differentiation
Activated B and T cells undergo clonal expansion, producing effector cells (engage in response) and memory cells (rapid recognition upon re-exposure).
Effector Cells: Actively respond to antigens.
Memory Cells: Provide long-term immunity.
Stage 4: Antigen Elimination and Memory
Cellular and humoral responses collaborate to eliminate antigens. Effector cells die off after the threat passes, while memory cells persist in lymphatic tissues.
Antigen Elimination: Coordinated action of both branches.
Immunological Memory: Long-lived memory cells ensure rapid response to future exposures.
Lymphocytes: T Cells and B Cells
Origin and Maturation
T cells are produced in the bone marrow and mature in the thymus, while B cells are produced and mature in the bone marrow. Mature lymphocytes mainly reside in lymphoid tissues.
T Cells: Roles in both humoral and cellular immunity.
B Cells: Coordinate humoral response by producing antibodies.
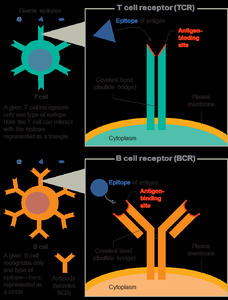
Antigen Recognition
Lymphocytes recognize antigens via specific receptors: T cell receptors (TCRs) and B cell receptors (BCRs). Each cell is monospecific,
recognizing a single epitope, but the immune system generates a vast array of cells for unlimited antigen recognition.
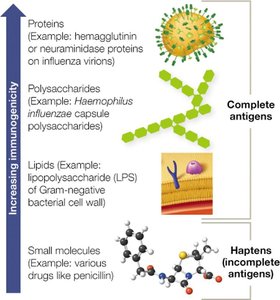
Immunogenicity and Antigen Features
Immunogenicity
An antigen is any substance that can trigger an immune response. Immunogenicity depends on size, complexity, and composition.
Proteins are most immunogenic, followed by polysaccharides, lipids, and haptens (incomplete antigens).
Haptens: Require linkage to larger molecules to be immunogenic.
Epitopes
Epitopes are specific parts of an antigen recognized by B and T cells. Each lymphocyte recognizes a single epitope.
T Cell Subtypes and Functions
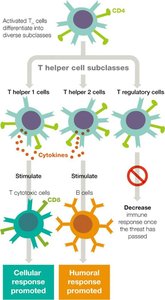
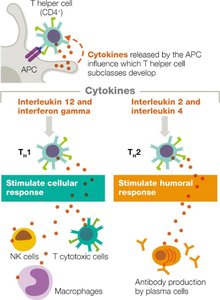
Helper T Cells (TH)
Helper T cells (CD4+) coordinate adaptive immunity by releasing cytokines and activating other white blood cells.
They differentiate into subclasses with specific functions:
TH1: Activate T cytotoxic cells, macrophages, and NK cells (cellular response).
TH2: Stimulate B cells to produce antibodies (humoral response).
Treg: Regulate immune response, reducing risk of autoimmunity.
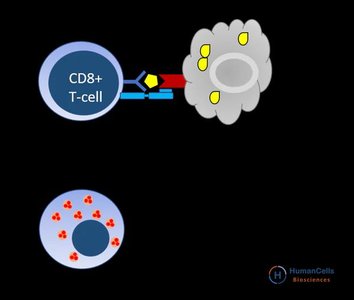
Cytotoxic T Cells (TC)
Cytotoxic T cells (CD8+) directly destroy infected, damaged, foreign, or cancerous cells.
They consist of effector and memory cells.
Self-Tolerance Screening
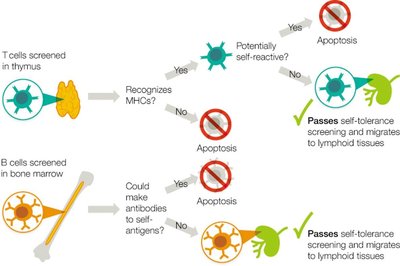
Mechanisms of Self-Tolerance
T and B cells are screened for self-tolerance to prevent autoimmunity.
T cells are tested in the thymus for recognition of MHC proteins, while
B cells are screened in the bone marrow to ensure antibodies do not cross-react with self-antigens.
Cells failing self-tolerance undergo apoptosis.
Comparison of T Cells and B Cells
Table: T Cells vs B Cells
Feature | T Cells | B Cells |
|---|---|---|
Adaptive immunity branch | Cellular | Humoral |
Site of maturation | Thymus | Bone marrow |
Antigen recognition receptors | TCRs | BCRs (antibodies) |
Require APC for activation | Yes | No |
Memory cells made | Yes | Yes |
MHC proteins present | MHC I | MHC I & II |
Considered APCs | No | Yes |
Antigen Presentation: MHC Molecules
MHC I and MHC II
MHC I is found on all nucleated cells and presents intracellular antigens to cytotoxic T cells.
MHC II is found only on APCs and presents extracellular antigens to helper T cells.
Type | Location | Interacts with | Antigens Presented |
|---|---|---|---|
MHC I | All body cells except RBCs | CD8+ T cells | Intracellular |
MHC II | APCs | CD4+ T cells | Extracellular |
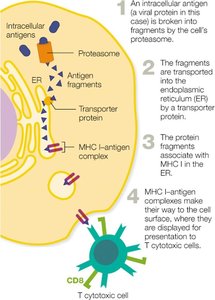
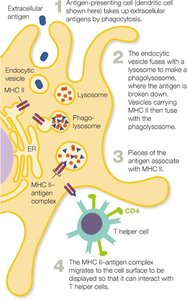
T Cell Activation and Differentiation
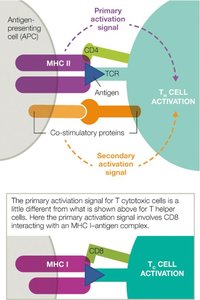
Activation Signals
T cells require two signals for activation:
primary (TCR-MHC-antigen interaction) and
secondary (co-stimulatory proteins).
The signaling cascade determines T cell subclass specialization.
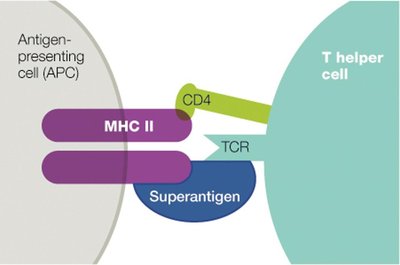
Superantigens
Superantigens are potent activators of T helper cells, causing broad, nonspecific activation and dangerous cytokine release, potentially leading to shock and death.
Proliferation and Differentiation
Activated T cells undergo clonal expansion and differentiate into effector and memory cells.
Cytokines released by APCs influence the development of T helper cell subclasses.
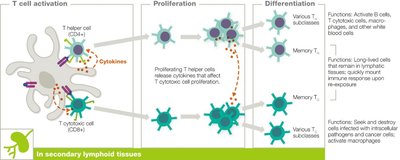
Effector Functions and Memory
T Cytotoxic Cell Roles
T cytotoxic cells eliminate infected or cancerous cells by releasing perforins (form pores) and granzymes (induce apoptosis).
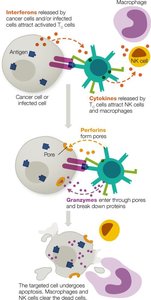
T Helper Cell Roles
T helper cells support B cells, T cytotoxic cells, and innate leukocytes.
TH1 cells favor cellular response,
TH2 cells promote humoral response.
B Cell Activation and Antibody Production
B Cell Activation
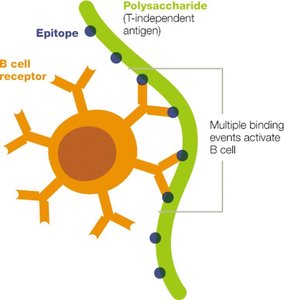
B cells can be activated by T-dependent or T-independent antigens.
Most antigens are T-dependent, requiring T helper cells for full activation.
T-independent activation is limited in memory and long-term protection.
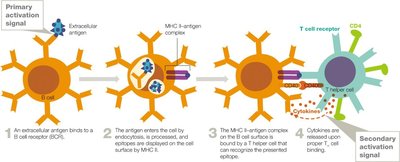
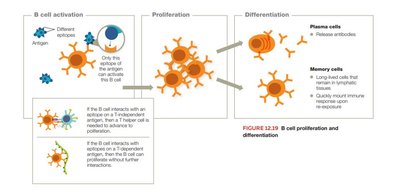
B Cell Proliferation and Differentiation
Activated B cells proliferate and differentiate into plasma cells (antibody producers) and memory B cells (long-term immunity).
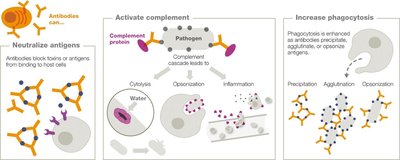
Antibody Functions
Antibodies (immunoglobulins) secreted by plasma cells bind antigens, activate complement, neutralize antigens, and promote phagocytosis.
Complement activation leads to cytolysis, inflammation, and opsonization.
Antibodies increase phagocytosis by precipitation, agglutination, and opsonization.
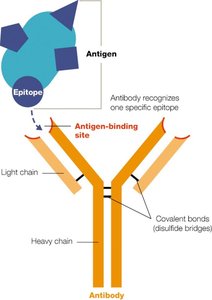
Antibody Structure and Isotypes
Structure
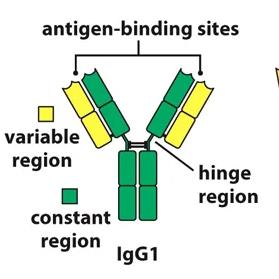
Antibodies are composed of two heavy and two light chains, held together by disulfide bonds.
The variable region forms the antigen-binding site, while the constant region forms the stem.
Isotypes
There are five antibody isotypes: IgG, IgA, IgM, IgE, and IgD. Each has distinct functions and locations in the body.
Isotype | Structure | Proportion | Functions | Notes |
|---|---|---|---|---|
IgG | Monomer | Most abundant | Neutralization, complement activation, opsonization | Crosses placenta, made later in infection |
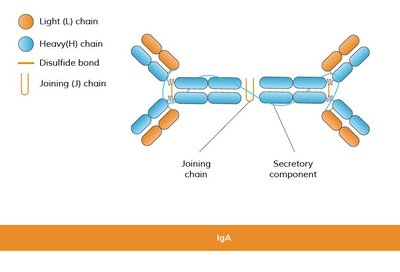
IgA | Monomer/dimer | Second most abundant | Neutralization, opsonization | Main antibody in breast milk, resistant to stomach acid |
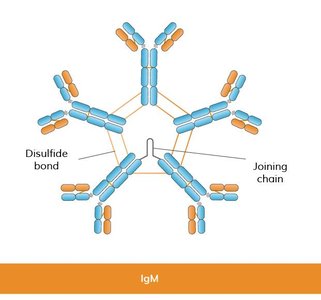
IgM | Monomer/pentamer | Third most abundant | Agglutination, precipitation, complement activation | Made early in infection |
IgE | Monomer | Rare | Fights parasites, mediates allergic responses | Bound to mast cells and basophils |
IgD | Monomer | Rare | Poorly understood | Bound to B cells |
Immunological Memory
Primary vs Secondary Response
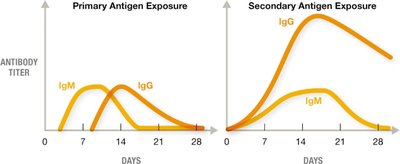
Effector cells die off after the threat subsides, but memory cells persist, allowing rapid and amplified responses upon re-exposure.
Primary exposure generates IgM first, then IgG. Secondary exposure rapidly produces high-affinity IgG.
Acquisition of Humoral Immunity
Types of Adaptive Immunity
Naturally Acquired Active Immunity: Infection triggers immune response; long-term protection.
Artificially Acquired Active Immunity: Vaccination triggers immune response; long-term protection.
Naturally Acquired Passive Immunity: Antibodies received non-medically (e.g., maternal); temporary protection.
Artificially Acquired Passive Immunity: Antibodies received medically (e.g., antiserum); temporary protection.
Clinical Application: Case Study
Acute Lymphoblastic Leukemia (ALL) and Immunity
Patients with ALL have increased abnormal lymphocytes, leading to anemia, fatigue, and bone pain. Bone marrow transplants require compatible tissue donors to reduce risk of graft-versus-host disease (GVHD). Revaccination is necessary after immune system reconstitution to restore protective antibody titers.
Microscopic Abnormalities: Increased immature lymphocytes, low RBC count.
Low Antibody Titers: Abnormal B cells do not produce functional antibodies.
Compatible Tissue Donor: Closely matched MHC proteins reduce GVHD risk.
Immunological Response to Incompatible Transplant: Donor cells attack host tissues, leading to GVHD.
Siblings as Donors: Not guaranteed due to genetic variability in MHC.
