 Back
BackAdaptive Immunity: T Cells, B Cells, and Antibody-Mediated Responses
Study Guide - Smart Notes

Adaptive Immunity: Overview
Introduction to Adaptive Immunity
The adaptive immune system is a highly specialized defense mechanism that targets specific pathogens through the coordinated actions of lymphocytes, primarily T cells and B cells. This system is characterized by its ability to remember previous encounters with antigens, leading to a faster and stronger response upon re-exposure.
Lymphocytes are the main cells of adaptive immunity, including T cells (CD4+ helper and CD8+ cytotoxic) and B cells.
Adaptive immunity is divided into cellular (T cell-mediated) and h umoral (antibody-mediated) branches.
Both branches progress through four general stages: antigen presentation, lymphocyte activation, proliferation and differentiation, and antigen elimination/memory.

T and B Lymphocytes
Development and Maturation
T and B cells originate from hematopoietic stem cells in the bone marrow but mature in different locations:
T cells: Produced in the bone marrow, mature in the thymus.
B cells: Produced and mature in the bone marrow.
Antigen Receptors
Both T and B cells express unique antigen receptors on their surfaces:
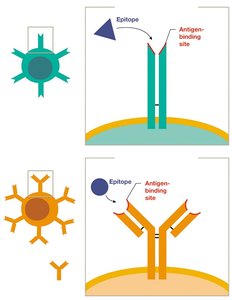
T cell receptors (TCRs) recognize antigens presented by other cells.
B cell receptors (BCRs) can bind directly to free antigens.
Each lymphocyte expresses thousands of identical receptors, each specific for a single epitope.
The diversity of receptors ensures the immune system can recognize virtually any antigen.
Stages of the Adaptive Immune Response
Stage 1: Antigen Presentation
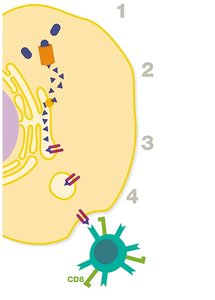
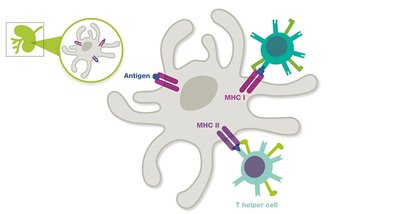
Antigen-presenting cells (APCs), such as dendritic cells, macrophages, and B cells, process and present antigens to T cells using major histocompatibility complex (MHC) molecules.
MHC I: Present on all nucleated cells; presents intracellular antigens to CD8+ cytotoxic T cells.
MHC II: Present only on professional APCs; presents extracellular antigens to CD4+ helper T cells.
B cells can recognize and bind antigens directly without the need for APCs.
Type | MHC I | MHC II |
|---|---|---|
Location | All nucleated cells (except RBCs) | APCs only |
Interacts with | CD8+ T cells | CD4+ T cells |
Antigens Presented | Intracellular | Extracellular |
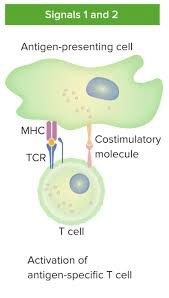
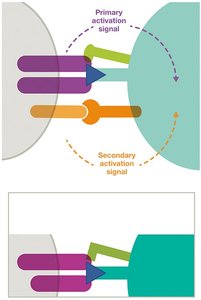
Stage 2: Lymphocyte Activation
Activation of T and B cells requires two signals:
Primary signal: TCR or BCR binding to antigen-MHC complex.
Secondary signal: Co-stimulatory molecules (e.g., B7 on APC binding CD28 on T cell).
Full activation leads to proliferation and differentiation.
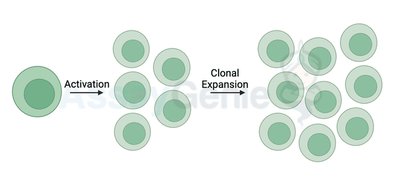
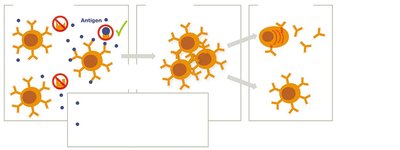
Stage 3: Lymphocyte Proliferation and Differentiation
Activated lymphocytes undergo clonal expansion and differentiate into effector and memory cells:
Effector cells: Actively participate in eliminating the antigen.
Memory cells: Remain in lymphatic tissues for rapid response upon re-exposure.
Stage 4: Antigen Elimination and Memory
Effector cells eliminate the antigen through various mechanisms. Once the threat is cleared, most effector cells die, but memory cells persist, providing long-term immunity.
Memory cells enable a faster and stronger response upon subsequent exposures to the same antigen.
T Cell Subsets and Functions
T Helper Cells (CD4+)
T helper cells coordinate immune responses by releasing cytokines that activate other immune cells. They differentiate into several subclasses:
TH1 cells: Promote cellular immunity (activate cytotoxic T cells, macrophages, NK cells).
TH2 cells: Promote humoral immunity (stimulate B cell maturation and antibody production).
T regulatory (Treg) cells: Suppress immune responses to maintain tolerance and prevent autoimmunity.
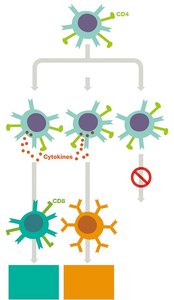
Cytotoxic T Cells (CD8+)
Cytotoxic T cells (also called killer T cells or CTLs) directly destroy infected, damaged, or cancerous cells by recognizing antigens presented on MHC I molecules.
Release perforins to form pores in target cell membranes.
Release granzymes to induce apoptosis in target cells.
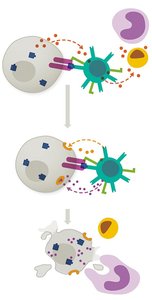
B Cells and Humoral Immunity
B Cell Activation
B cells can be activated by two types of antigens:
T-independent antigens: Directly activate B cells (e.g., polysaccharides).
T-dependent antigens: Require help from T helper cells (especially TH2) for full activation (e.g., proteins).
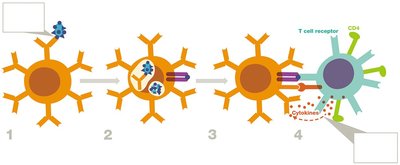
B Cell Proliferation and Differentiation
Activated B cells proliferate and differentiate into:
Plasma cells: Secrete antibodies specific to the antigen.
Memory B cells: Provide long-term immunity.
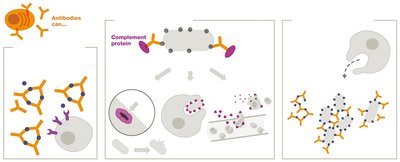
Antibody Functions
Antibodies (immunoglobulins) help eliminate antigens through several mechanisms:
Neutralization: Block toxins or pathogens from binding to host cells.
Opsonization: Enhance phagocytosis by marking antigens for destruction.
Agglutination/Precipitation: Clump antigens together for easier removal.
Complement activation: Trigger the complement cascade, leading to cell lysis and inflammation.
Antibody Structure and Isotypes
Structure of Antibodies
Antibodies are Y-shaped molecules composed of two heavy chains and two light chains, held together by disulfide bonds. The tips of the Y form the antigen-binding sites (Fab region), while the stem (Fc region) determines the antibody's class and function.
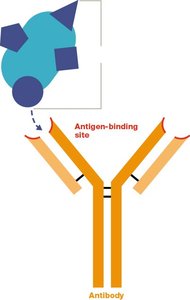
Antibody Isotypes
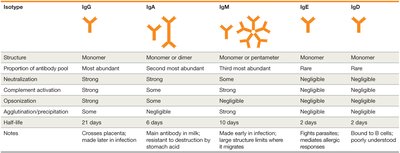
There are five main antibody isotypes, each with distinct functions and properties:
Isotype | Structure | Proportion | Key Functions | Half-life |
|---|---|---|---|---|
IgG | Monomer | Most abundant | Neutralization, opsonization, complement activation, crosses placenta | 21 days |
IgA | Monomer or dimer | Second most abundant | Neutralization, mucosal immunity, found in secretions | 6 days |
IgM | Monomer or pentamer | Third most abundant | Agglutination, complement activation, first made in infection | 10 days |
IgE | Monomer | Rare | Allergic responses, defense against parasites | 2 days |
IgD | Monomer | Rare | B cell receptor, poorly understood | 2 days |
Immunological Memory
Primary vs. Secondary Immune Response
Upon first exposure to an antigen, the adaptive immune response is slower and less robust (primary response). Memory cells generated during this response enable a much faster and stronger reaction upon subsequent exposures (secondary response), providing the basis for long-term immunity and vaccine effectiveness.
Summary Table: Major Histocompatibility Complex Features
Type | MHC I | MHC II |
|---|---|---|
Location | All nucleated cells (except RBCs) | APCs only |
Interacts with | CD8+ T cells | CD4+ T cells |
Antigens Presented | Intracellular | Extracellular |
Additional info: This guide covers the core concepts of adaptive immunity, including the roles of T and B cells, antigen presentation, lymphocyte activation, antibody structure and function, and immunological memory. Understanding these principles is essential for further study in immunology, infectious disease, and vaccine development.
