 Back
BackAdaptive (Specific) Immunity: Mechanisms, Cells, and Applications
Study Guide - Smart Notes

Specific Immunity: The Adaptive Line of Defense
Overview of Adaptive Immunity
Adaptive (specific) immunity is the third line of defense in the immune system, characterized by its ability to recognize and remember specific pathogens. This system is mediated by B and T lymphocytes, which develop immunocompetence—the ability to interact with a wide spectrum of foreign substances known as antigens.
Specificity: Antibodies and immune cells target only the specific antigen that triggered their production.
Memory: Lymphocytes "recall" their first encounter with an antigen and respond more rapidly upon subsequent exposures.


Development and Differentiation of the Immune System
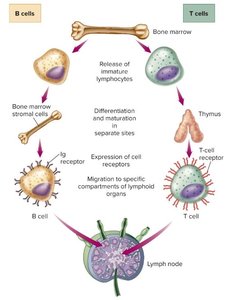
Lymphocyte Maturation and Migration
Lymphocytes originate from stem cells in the bone marrow and differentiate into either B cells or T cells. B cells mature in the bone marrow, while T cells mature in the thymus. Both cell types migrate to secondary lymphoid tissues, such as lymph nodes and spleen, where they encounter antigens.
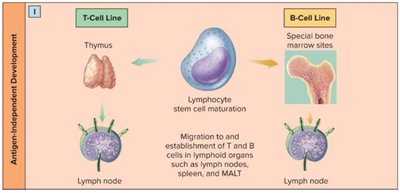
Antigen-Dependent and Antigen-Independent Development
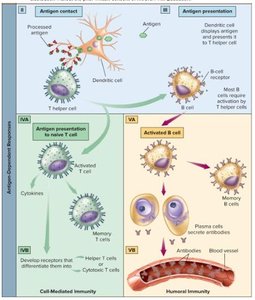
After maturation, lymphocytes populate lymphoid organs and are ready to respond to antigens. The immune response involves antigen contact, antigen presentation, and the activation of B and T cells, leading to either cell-mediated or humoral immunity.
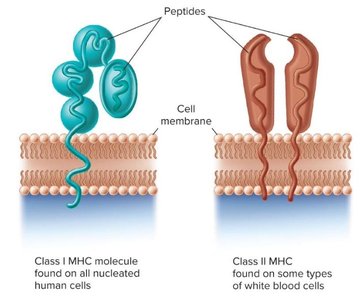
Major Histocompatibility Complex (MHC)
Structure and Function of MHC Molecules
The Major Histocompatibility Complex (MHC) is a set of cell surface proteins essential for acquired immunity. MHC molecules help the immune system distinguish self from non-self and are divided into two main classes:
Class I MHC: Found on all nucleated cells; present antigens to cytotoxic T cells (CD8+).
Class II MHC: Found on antigen-presenting cells (APCs) such as macrophages, dendritic cells, and B cells; present antigens to helper T cells (CD4+).
Lymphocyte Receptors and Clonal Selection
B and T Cell Receptors
Lymphocytes recognize antigens through highly specific receptors:
B cell receptors (BCR): Bind free antigens directly.
T cell receptors (TCR): Bind processed antigens presented with MHC molecules on APCs.
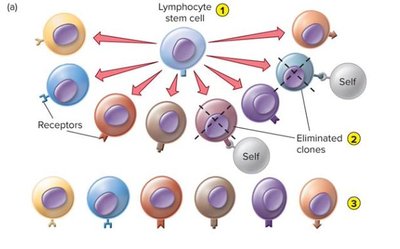
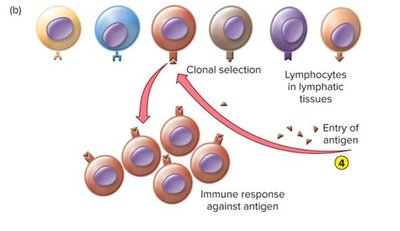
Clonal Selection Theory
Each lymphocyte expresses a unique receptor specificity, determined during development. When an antigen enters the body, it "selects" the lymphocyte clone with the matching receptor, leading to clonal expansion and a targeted immune response.
Immunoglobulins: Structure and Diversity
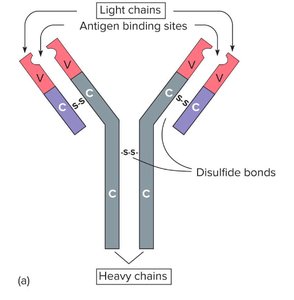
Structure of Immunoglobulins (Antibodies)
Immunoglobulins are Y-shaped glycoproteins composed of two identical heavy chains and two identical light chains, connected by disulfide bonds. The variable (V) regions at the tips of the "Y" form the antigen-binding sites, while the constant (C) regions determine the antibody class.
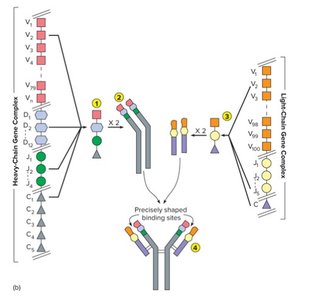
Genetic Basis of Antibody Diversity
Antibody diversity arises from the recombination of multiple gene segments encoding the variable and constant regions of heavy and light chains. This process allows the immune system to generate billions of unique antigen-binding sites.
T Cell Receptors and Maturation
T Cell Receptor Structure and CD Markers
T cell receptors are formed by genetic recombination and consist of variable and constant regions. T cells express specific surface markers known as clusters of differentiation (CD), such as CD4 (on helper T cells) and CD8 (on cytotoxic T cells).
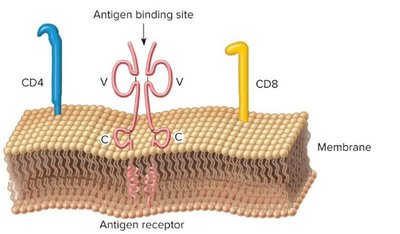
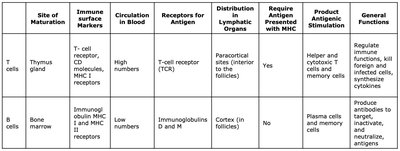
Comparison of B Cells and T Cells
Site of Maturation | Immune Surface Markers | Circulation in Blood | Receptors for Antigen | Distribution in Lymphatic Organs | Require Antigen Presented with MHC | Product after Antigenic Stimulation | General Functions |
|---|---|---|---|---|---|---|---|
Thymus gland (T cells) Bone marrow (B cells) | T cell receptor, CD molecules, MHC I receptors (T cells) Immunoglobulin, MHC I and II receptors (B cells) | High (T cells) Low (B cells) | T-cell receptor (TCR) (T cells) Immunoglobulins D and M (B cells) | Paracortical sites (T cells) Cortex (B cells) | Yes (T cells) No (B cells) | Helper and cytotoxic T cells, memory cells (T cells) Plasma cells and memory cells (B cells) | Regulate immune functions, kill foreign and infected cells, synthesize cytokines (T cells) Produce antibodies to target, inactivate, and neutralize antigens (B cells) |
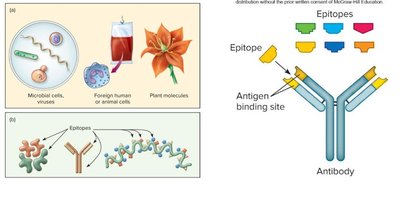
Antigens and Immunogenicity
Characteristics of Antigens
An antigen is any substance that elicits an immune response in specific lymphocytes. The property of behaving as an antigen is called antigenicity, which depends on factors such as foreignness, size, shape, and accessibility. Antigens contain multiple epitopes (antigenic determinants) recognized by lymphocytes.
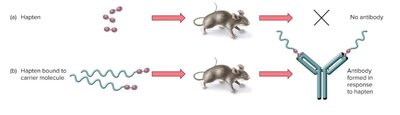
Haptens and Carrier Molecules
Haptens are small molecules that cannot elicit an immune response unless attached to a larger carrier molecule. The carrier increases the size and enhances the orientation of the antigen, while the hapten serves as the epitope.
Antigen Processing and Presentation
Role of Antigen-Presenting Cells (APCs)
Antigen-presenting cells (APCs) such as macrophages, dendritic cells, and B cells process antigens and present them on their surface bound to MHC class II molecules. This presentation is essential for the activation of T helper cells, which then secrete cytokines to activate other immune cells.
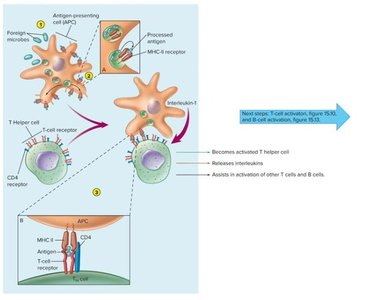
T Cell Responses and Cell-Mediated Immunity
Types and Functions of T Cells
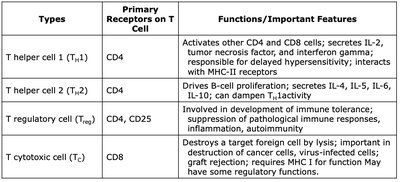
T cells are central to cell-mediated immunity. The main types include:
Type | Primary Receptors | Functions/Features |
|---|---|---|
T helper cell 1 (TH1) | CD4 | Activates CD4 and CD8 cells; secretes IL-2, TNF, IFN-γ; interacts with MHC-II; delayed hypersensitivity. |
T helper cell 2 (TH2) | CD4 | Drives B-cell proliferation; secretes IL-4, IL-5, IL-6, IL-10; can dampen TH1 activity. |
T regulatory cell (Treg) | CD4, CD25 | Immune tolerance; suppresses pathological responses, inflammation, autoimmunity. |
T cytotoxic cell (TC) | CD8 | Destroys target cells by lysis; important in cancer, virus-infected cells, graft rejection; requires MHC I. |
T Cell Activation and Effector Functions
Upon activation by antigen-MHC complexes, T cells differentiate into effector and memory cells. Helper T cells (CD4+) regulate immune responses, while cytotoxic T cells (CD8+) destroy infected or abnormal cells by releasing perforins and granzymes.
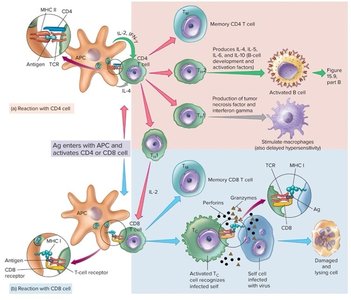
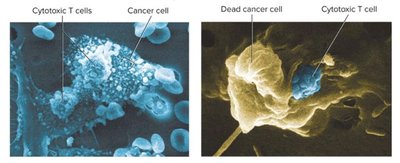
B Cell Responses and Antibody Production
Activation and Differentiation of B Cells
B cells are activated when they encounter their specific antigen, process it, and receive help from T helper cells. Activated B cells proliferate and differentiate into plasma cells (which secrete antibodies) and memory B cells (which provide long-term immunity).
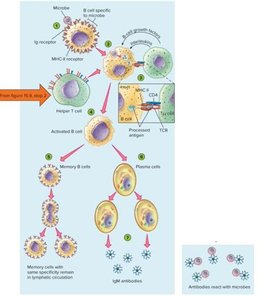
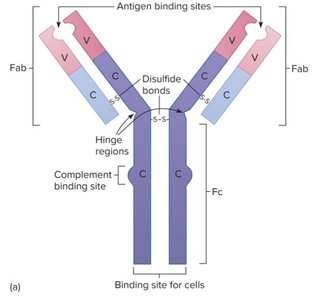
Antibody Structure and Function
Functional Regions of Antibodies
Antibodies have two main functional regions:
Fab (antigen-binding) fragments: Bind to specific antigens via hypervariable regions.
Fc (crystallizable) fragment: Binds to immune cells and mediates effector functions such as complement activation.
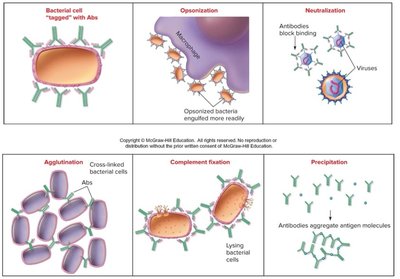
Antibody-Antigen Interactions
Antibodies neutralize, opsonize, agglutinate, and precipitate antigens, and can activate the complement system. These mechanisms help eliminate pathogens from the body.
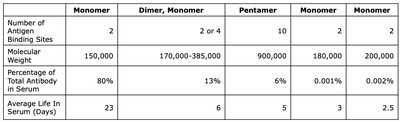
Classes of Immunoglobulins
Comparison of Immunoglobulin Classes
There are five main classes of immunoglobulins, each with distinct structural and functional properties:
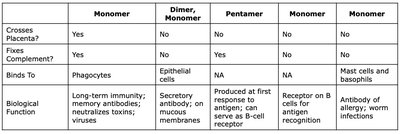
Class | Structure | Antigen Binding Sites | Molecular Weight | Serum % | Avg. Life (days) | Crosses Placenta? | Fixes Complement? | Binds To | Biological Function |
|---|---|---|---|---|---|---|---|---|---|
IgG | Monomer | 2 | 150,000 | 80% | 23 | Yes | Yes | Phagocytes | Long-term immunity; memory antibodies; neutralizes toxins/viruses |
IgA | Dimer, Monomer | 2 or 4 | 170,000-385,000 | 13% | 6 | No | No | Epithelial cells | Secretory antibody; on mucous membranes |
IgM | Pentamer | 10 | 900,000 | 6% | 5 | No | Yes | NA | First response to antigen; can serve as B-cell receptor |
IgD | Monomer | 2 | 180,000 | 0.001% | 3 | No | No | NA | Receptor on B cells for antigen recognition |
IgE | Monomer | 2 | 200,000 | 0.002% | 2.5 | No | No | Mast cells, basophils | Antibody of allergy; worm infections |
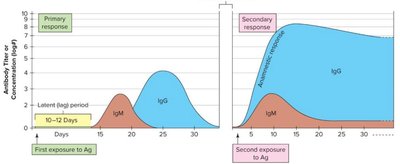
Antibody Production Over Time
Primary and Secondary Immune Responses
Upon first exposure to an antigen, the primary immune response is characterized by a lag period and the production of IgM, followed by IgG. Subsequent exposures elicit a faster and stronger secondary (anamnestic) response, dominated by IgG.
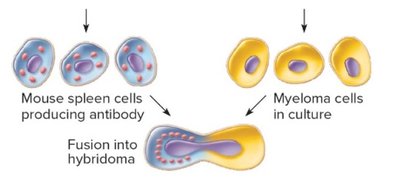
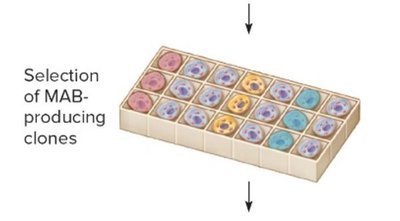
Monoclonal Antibodies
Production and Applications
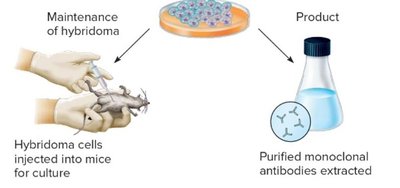
Monoclonal antibodies are produced from a single clone of B cells fused with myeloma cells to form hybridomas. These antibodies are highly specific and are used in diagnostics, therapy, and research.

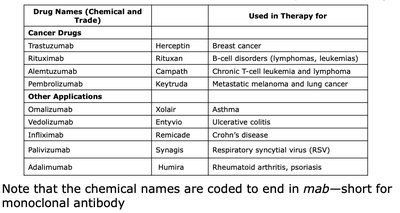
Drug Name | Used in Therapy for |
|---|---|
Trastuzumab (Herceptin) | Breast cancer |
Rituximab (Rituxan) | B-cell disorders (lymphomas, leukemias) |
Alemtuzumab (Campath) | Chronic T-cell leukemia and lymphoma |
Pembrolizumab (Keytruda) | Metastatic melanoma and lung cancer |
Omalizumab (Xolair) | Asthma |
Vedolizumab (Entyvio) | Ulcerative colitis |
Infliximab (Remicade) | Crohn's disease |
Palivizumab (Synagis) | Respiratory syncytial virus (RSV) |
Adalimumab (Humira) | Rheumatoid arthritis, psoriasis |
Immunity Categories and Immunization
Types of Immunity
Active immunity: Produced by exposure to antigen; creates memory; long-lasting.
Passive immunity: Preformed antibodies are transferred; no memory; short-lived.
Natural immunity: Acquired through normal life events (e.g., infection, maternal antibodies).
Artificial immunity: Acquired through medical intervention (e.g., vaccination, immunotherapy).
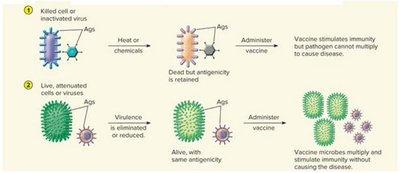
Principles of Vaccine Preparation
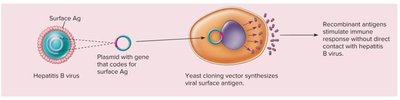
Vaccines can be prepared from killed or inactivated microbes, live attenuated organisms, acellular components, or recombinant antigens. An effective vaccine should be safe, provide long-term protection, and stimulate both B and T cell responses.
Herd Immunity
When a significant portion of a population is immune to a pathogen, the spread of disease is reduced, protecting non-immune individuals—a phenomenon known as herd immunity.
Additional info: These notes provide a comprehensive overview of adaptive immunity, including the development and function of B and T lymphocytes, antigen recognition, antibody structure and function, and the principles of immunization. The included tables and images reinforce key concepts and support exam preparation for microbiology students.
