 Back
Back5- 3
Study Guide - Smart Notes

Controlling Microbial Growth in the Environment and the Body
Food Preservation and Microbial Control
Food preservation relies on chemical and physical methods to inhibit or kill microbes, thereby extending shelf life and ensuring safety. Understanding the mechanisms by which these methods work is essential for microbiology students.
Key Preservatives:
Sugar and Salt: Increase solute concentration, causing dehydration of microbes via osmosis, which inhibits their growth.
Sodium Nitrite: Inhibits microbial growth, especially Clostridium botulinum, by interfering with cellular respiration.
Potassium Sorbate: The salt of sorbic acid, lowers pH and inhibits the growth of molds, yeasts, and some bacteria.
Physical Methods:
Freezing: Slows or stops microbial metabolism and growth.
Desiccation: Removes water, halting metabolic activities.
Filtration: Removes microbes from liquids based on size exclusion.
Ionizing Radiation: Damages DNA, leading to microbial death.
Non-ionizing Radiation: Causes DNA damage (e.g., UV light), preventing microbial replication.


Example: The ingredient label for beef jerky lists salt, sugar, sodium nitrite, and potassium sorbate, all of which contribute to microbial inhibition and food preservation.
Antimicrobial Agents: Definitions and Types
Basic Terminology
Antimicrobial agents are chemicals that kill or inhibit the growth of microorganisms. They are classified based on their effect and target organism.
Antibiotics: Naturally occurring compounds produced by fungi or bacteria; can be modified or synthesized.
Antimicrobial Agents: Includes antibacterials, antifungals, antivirals, and antiparasitics.
-cidal: Agents that kill microorganisms (e.g., bactericidal, fungicidal).
-static: Agents that inhibit growth without killing (e.g., bacteriostatic).
-lytic: Agents that kill by lysing cells (e.g., bacteriolytic).
Selectivity: Effective antimicrobials exhibit selective toxicity, targeting pathogens while sparing host cells due to structural differences.

Example: Neosporin contains three antimicrobials (Bacitracin, Neomycin, Polymyxin B) to target a broad range of bacteria.
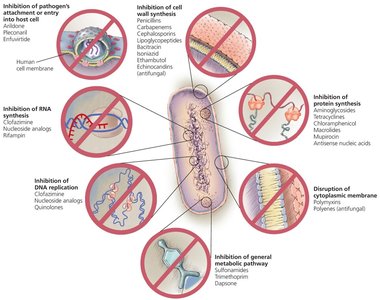
Mechanisms of Action
Antimicrobials are classified by their mechanism of action, which determines how they inhibit or kill microbes. There are seven major groups:
Inhibition of DNA replication
Inhibition of RNA synthesis
Inhibition of protein synthesis
Inhibition of cell wall synthesis
Disruption of cell membrane
Disruption of metabolism
Prevention of attachment/entry into host
Classification of Antimicrobial Activity
Bacteriostatic, Bactericidal, and Bacteriolytic Agents
Understanding the differences between these agents is crucial for selecting appropriate treatments.
Bacteriostatic: Inhibit growth by interfering with processes like protein synthesis; effect is reversible.
Bactericidal: Kill bacteria by tightly binding to essential targets, halting all cellular function.
Bacteriolytic: Kill bacteria by lysing the cell, leading to cell death and breakdown.
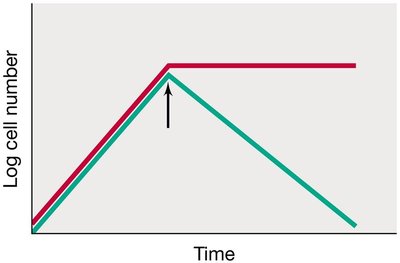
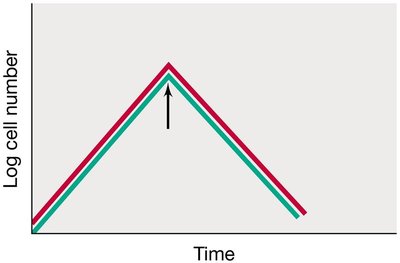
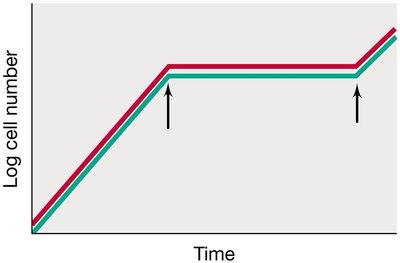
Example: Bacteriostatic agents pause growth, but cells can resume multiplying if the agent is removed. Bactericidal and bacteriolytic agents result in cell death, with bacteriolytic agents causing cell lysis.
Spectrum of Action
Specificity and Broad-Spectrum Agents
Antimicrobials vary in their spectrum of activity, which is determined by the structural differences among microbes and their susceptibility to the drug.
Narrow-spectrum: Target specific groups (e.g., only Gram-positive bacteria).
Broad-spectrum: Effective against a wide range of microbes (e.g., both Gram-positive and Gram-negative bacteria).
Structural Differences: Gram-positive and Gram-negative bacteria differ in cell envelope structure, affecting drug sensitivity.
Drug | Mycobacteria | Gram-negative bacteria | Gram-positive bacteria | Chlamydias/Rickettsias | Protozoa | Fungi | Helminths | Viruses |
|---|---|---|---|---|---|---|---|---|
Isoniazid | X | |||||||
Polymyxin | X | |||||||
Penicillin | X | |||||||
Streptomycin | X | X | X | |||||
Erythromycin | X | X | X | |||||
Tetracycline | X | X | X | X | ||||
Sulfonamides | X | X | X | X | ||||
Azoles | X | |||||||
Nicosamide | X | |||||||
Praziquantel | X | |||||||
Arildone, Ribavirin, Acyclovir | X |

Example: Tetracycline is a broad-spectrum antibiotic effective against many bacteria and some protozoa, while penicillin is more specific for Gram-positive bacteria.
Special Considerations for Unique Microbes
Mycobacteria
Mycobacteria, such as Mycobacterium tuberculosis, have unique cell envelopes with a lipid-rich outer leaflet and mycolic acids, making them resistant to many drugs and requiring specialized treatments.
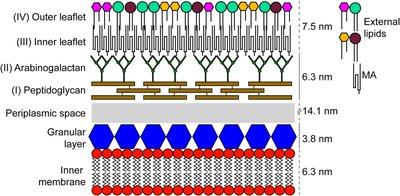
Example: Isoniazid is used specifically to target mycobacteria due to their unique cell wall structure.
Antifungal Drugs
Fungi are eukaryotes, so antifungal drugs must target features unique to fungi to avoid harming human cells. Common targets include ergosterol in the plasma membrane and enzymes involved in cell wall synthesis.
Ergosterol Inhibitors: Disrupt fungal membranes (e.g., azoles, polyenes).
Echinocandins: Inhibit glucan synthase, blocking cell wall synthesis.
Topical Application: Used to minimize toxicity to host tissues.
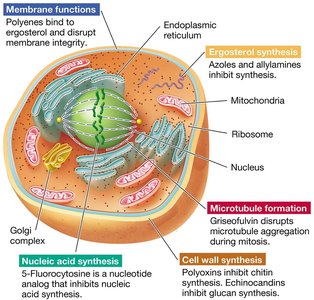
Example: Topical antifungal creams target ergosterol or chitin, structures not found in human cells, reducing side effects.
Antiviral Drugs
Viruses rely on host cells for replication, so antiviral drugs must target viral-specific enzymes or processes to avoid host toxicity.
Nucleoside Analogs: Block viral DNA synthesis (e.g., AZT for HIV).
Protease Inhibitors: Prevent processing of viral proteins.
Neuraminidase Inhibitors: Block viral release from host cells (e.g., Tamiflu).
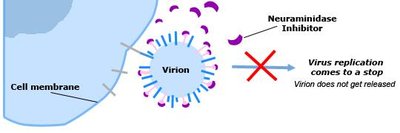
Example: Tamiflu inhibits neuraminidase, preventing influenza virus from spreading to new cells.
Routes of Administration and Side Effects
Drug Administration Methods
The route of administration affects drug concentration, efficacy, and side effects.
Topical: Applied to skin or mucous membranes for local infections.
Oral: Swallowed; convenient but may result in lower blood concentrations.
Intramuscular (IM): Injected into muscle; absorbed into bloodstream over time.
Intravenous (IV): Directly into bloodstream; achieves high concentrations quickly.
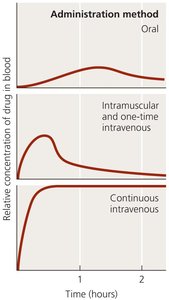
Example: Topical antifungals are preferred for skin infections to minimize systemic side effects.
Safety and Side Effects
Antimicrobial drugs can cause side effects, including allergic reactions, toxicity to organs, and disruption of normal microbiota, which may allow opportunistic pathogens to proliferate.
Allergic Reactions: Range from mild to severe (anaphylaxis).
Organ Toxicity: Liver and kidneys are most at risk.
Microbiota Disruption: Can lead to secondary infections.
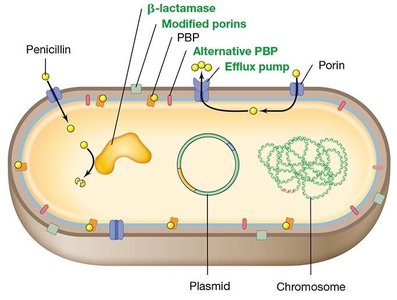
Mechanisms and Spread of Antimicrobial Resistance
Mechanisms of Resistance
Microbes can acquire resistance to antimicrobials through several mechanisms, often encoded by resistance genes.
Impermeability to the drug
Modification of the drug target
Enzymatic inactivation of the drug
Efflux pumps that expel the drug
Metabolic bypasses
Biofilm formation and decoy molecules
Development and Spread of Resistance
Resistance arises from genetic variation within microbial populations. Exposure to antimicrobials selects for resistant cells, which survive and multiply, leading to a population shift toward resistance. This process does not induce new mutations but selects for pre-existing resistant variants.
Selection Pressure: Use of antimicrobials kills sensitive cells, allowing resistant ones to thrive.
Genetic Transmission: Resistance genes can spread via horizontal gene transfer (e.g., plasmids).
Practice Questions and Applications
Sample Questions
Which agent permanently kills bacteria? Bactericidal and bacteriolytic agents; bacteriostatic agents only pause growth.
Why can't a drug targeting bacterial DNA polymerase stop a virus with an RNA genome? Viruses may use different enzymes (e.g., RNA polymerase), so the drug is ineffective.
What antifungal drug targets minimize toxicity? Drugs targeting ergosterol, chitin, or cell wall assembly enzymes are effective because these structures are not found in human cells.
Which administration route minimizes side effects? Topical application is best for limiting systemic side effects.
Additional info: For further study, review mechanisms of action for antimicrobials and Koch’s Postulates as referenced in the homework section.
