 Back
BackAntimicrobial Drugs: Mechanisms, Classes, and Resistance
Study Guide - Smart Notes

Antimicrobial Treatment
Overview of Antimicrobial Drugs
Antimicrobial drugs are therapeutic compounds designed to kill or inhibit the growth of microbes. They are classified based on the type of pathogen they target:
Antibacterial drugs: Treat bacterial infections
Antiviral drugs: Target viral infections
Antifungal drugs: Target fungal infections
Antiparasitic drugs: Treat protozoan and helminthic (worm) infections
The primary goal of antimicrobial chemotherapy is to destroy the infective agent without harming the host’s cells.
Characteristics of the Ideal Antimicrobial Drug
Toxic to the microbe but nontoxic to the host
Microbicidal rather than microbiostatic
Relatively soluble and functions even when highly diluted in body fluids
Remains potent long enough to act and is not broken down or excreted prematurely
Does not lead to the development of antimicrobial resistance
Complements or assists the activities of the host’s defenses
Remains active in tissues and body fluids
Readily delivered to the site of infection
Reasonably priced
Does not disrupt the host’s health by causing allergies or predisposing the host to other infections
Characteristics of Antimicrobials
Spectrum of Activity
Broad-spectrum drugs: Effective against more than one group of bacteria (e.g., tetracyclines target both gram-positive and gram-negative bacteria).
Narrow-spectrum drugs: Target a specific group of bacteria (e.g., penicillins mainly target gram-positive bacteria).
Bacteriostatic vs. Bactericidal
Bacteriostatic: Prevent bacteria from growing, often by targeting protein synthesis and metabolic pathways.
Bactericidal: Kill bacteria, typically by targeting cell walls, cell membranes, or nucleic acids. May also kill normal microbiota, potentially leading to harmful toxin release (e.g., LPS).
Drug Safety and Selective Toxicity
Selective Toxicity
Selective toxicity is the central concept in antibiotic treatment. The best drugs block the actions or synthesis of molecules in microorganisms but not in vertebrate cells. As the infectious agent becomes more similar to the host, selective toxicity becomes harder to achieve, increasing the risk of side effects.
Therapeutic Index
The therapeutic index is the ratio of the maximum tolerated (safe) dose to the minimum effective (therapeutic) dose.
A high therapeutic index is safer than a narrow one.
The kidneys and liver are especially susceptible to drug toxicity, as they metabolize and eliminate drugs.
History and Sources of Antimicrobials
Paul Ehrlich (1910): Discovered the first antimicrobial drug (Salvarsan) for syphilis.
Alexander Fleming (1928): Discovered penicillin from Penicillium.
Gerhard Domagk (1932): Discovered sulfanilamide for antibacterial use.
Antibiotics are commonly produced by bacteria (e.g., Streptomyces, Bacillus) and fungi (e.g., Penicillium, Cephalosporium). Semisynthetic drugs are chemically modified natural antibiotics, while synthetic drugs are entirely lab-created.
Mechanisms of Antimicrobial Action
Five Major Targets of Antimicrobial Agents
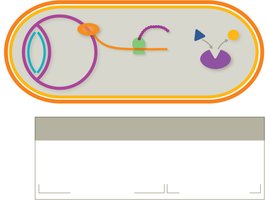
Inhibition of cell wall synthesis
Inhibition of nucleic acid (DNA and RNA) structure and function
Inhibition of protein synthesis
Interference with cell membrane structure or function
Inhibition of folic acid synthesis
Ideal drug targets are structures and processes unique to bacteria, minimizing harm to human cells.
Examples of Drug Classes and Their Targets
Target | Examples of Drugs | Typical Effect |
|---|---|---|
Cell wall synthesis | Penicillins, Cephalosporins, Carbapenems, Monobactams, Glycopeptides | Bactericidal |
Plasma membrane | Polypeptide drugs | Bactericidal |
Nucleic acids | Quinolones, Rifamycins | Bactericidal |
Protein synthesis | Macrolides, Tetracyclines, Aminoglycosides, Lincosamides, Phenicols | Often bacteriostatic |
Folic acid synthesis | Sulfa drugs, Trimethoprim | Usually bacteriostatic |
Antibacterial Drugs: Mechanisms and Examples
Targeting the Cell Wall
Drugs: Penicillins, Cephalosporins, Bacitracin, Isoniazid, Vancomycin
Mechanism: Block peptidoglycan production, weakening the cell wall and causing cell lysis.
Most effective during the exponential growth phase when bacteria are actively building cell walls.
Targeting Protein Synthesis
Drugs: Aminoglycosides (gentamicin, kanamycin, streptomycin), Tetracyclines, Macrolides (erythromycin, clarithromycin, azithromycin)
Mechanism: Target bacterial ribosomes, often broad spectrum, usually bacteriostatic.
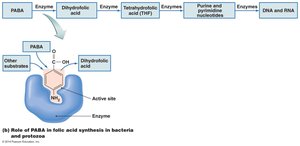
Targeting Folic Acid Synthesis
Drugs: Sulfonamides (sulfa drugs), Trimethoprim
Mechanism: Sulfa drugs act as competitive inhibitors, blocking enzymes required for folic acid production. Mammalian cells are unaffected because they do not synthesize their own folic acid.
Often administered together (e.g., TMP-SMX) for a synergistic effect, targeting different steps in the pathway.
Targeting DNA or RNA
Drugs: Fluoroquinolones
Mechanism: Inhibit nucleic acid synthesis, leading to bacterial cell death.
Targeting Cell Membranes
Drugs: Polymyxins
Mechanism: Interact with lipopolysaccharide and destabilize the outer membrane of gram-negative bacteria, causing cytoplasmic leakage and cell lysis. Noted for kidney toxicity.
Antifungal, Antiprotozoan, and Antiviral Drugs
Antifungal Drugs
Amphotericin B: Binds to fungal membranes, causing loss of selective permeability; highly toxic, administered intravenously.
Azoles (miconazole, clotrimazole): Interfere with sterol synthesis in fungi; common over-the-counter antifungals.
Antiprotozoan Drugs
Quinine (chloroquine), Metronidazole (Flagyl): Used to treat protozoan infections.
Antiviral Drugs
More difficult to design due to viruses relying on host cell machinery.
Often block replication of viral proteins, but may cause severe side effects due to effects on host cells.
Antimicrobial Resistance and Stewardship
Overview of Resistance
Antimicrobial resistance occurs when a microbe is not affected by a drug therapy intended to inhibit or eliminate it. Resistant microbes are called superbugs. The "antibiotic crisis" is largely due to overuse and misuse of antibiotics in medicine and agriculture.
Patients may miss doses, not finish regimens, or self-medicate with leftovers.
Antibiotics are often added to animal feed to promote growth.
Inappropriate prescribing practices contribute to resistance.
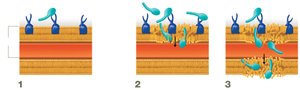
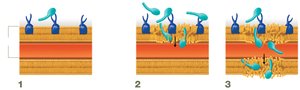
Mechanisms of Microbial Drug Resistance
Altering the drug’s target (mutation or acquisition of new genes)
Inactivating the drug (e.g., by producing enzymes such as beta-lactamases)
Reducing drug concentrations inside the cell (blocking entry or pumping the drug out via efflux pumps)
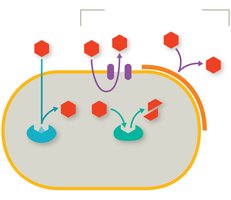
Target Alterations
Microbes may resist antimicrobials by altering the drug’s target, decreasing the number of binding sites or their affinity, thus preventing drug binding.
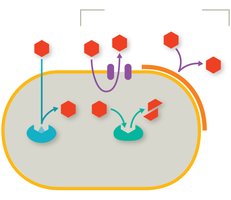
Drug Inactivation
Bacteria may produce enzymes that inactivate drugs by breaking them down (e.g., beta-lactamases) or by adding chemical groups. Genes encoding these enzymes are often carried on plasmids, facilitating horizontal gene transfer.
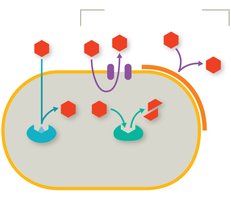
Reducing Drug Concentrations Inside the Cell
Bacteria can lower the concentration of a drug in their cytoplasm by limiting drug entry or actively pumping drugs out (efflux pumps).
Combating Drug Resistance: Stewardship
Healthcare Worker Responsibilities
Follow proper hand hygiene and enforce contact precautions
Limit unnecessary antimicrobial prescriptions
Use narrow-spectrum drugs when possible
Educate patients about the importance of following drug regimens
Patient Responsibilities
Follow all drug-dosing instructions
Attend follow-up appointments as recommended
Properly store medications
Do not demand antibiotics from physicians
