 Back
BackAntimicrobial Drugs: Mechanisms, Classes, and Resistance
Study Guide - Smart Notes

Antimicrobial Drugs
Introduction to Antimicrobial Drugs
Antimicrobial drugs are agents designed to interfere with the growth and survival of microorganisms within a host. Unlike disinfectants, these drugs must exhibit selective toxicity, meaning they target pathogens without causing significant harm to the host organism. The effectiveness and safety of these drugs depend on their ability to exploit differences between microbial and host cell structures or metabolic pathways.
Spectrum of Antimicrobial Activity
Narrow-spectrum drugs: Effective against a limited range of microorganisms. Example: Penicillin G, which mainly targets gram-positive bacteria.
Broad-spectrum drugs: Effective against a wide variety of organisms, both gram-positive and gram-negative. Example: Tetracycline.
Advantages of broad-spectrum drugs: Useful when the causative organism is unknown.
Disadvantages: Can disrupt normal microflora, leading to superinfections.
Bactericidal vs. Bacteriostatic Agents
Bactericidal: Drugs that kill bacteria directly.
Bacteriostatic: Drugs that inhibit bacterial growth, allowing the host's immune system to eliminate the pathogen.
Mechanisms of Antimicrobial Action
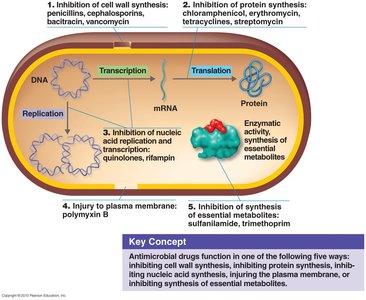
Antimicrobial drugs function by targeting essential processes in microorganisms. The five main mechanisms are:
Inhibition of cell wall synthesis
Inhibition of protein synthesis
Inhibition of nucleic acid synthesis
Disruption of plasma membrane integrity
Inhibition of essential metabolite synthesis
Cell Wall Active Agents
Inhibition of Cell Wall Synthesis
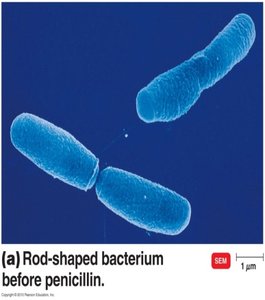
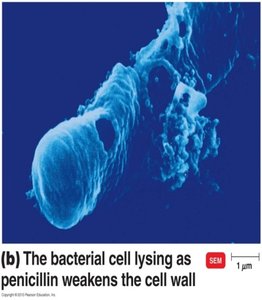
These drugs target the synthesis of peptidoglycan, a molecule unique to bacterial cell walls. Without peptidoglycan, bacteria cannot maintain cell wall integrity and will lyse during growth. This mechanism provides excellent selective toxicity.
Penicillins: Naturally occurring antibiotics containing a beta-lactam ring. They prevent cross-linking of peptidoglycan. Examples: Penicillin G, amoxicillin, ampicillin, methicillin.
Cephalosporins: Similar to penicillins, with a broader spectrum. Examples: Cefixime, cephalothin.
Bacitracin: Polypeptide antibiotic, inhibits an earlier stage of peptidoglycan synthesis. Used topically due to toxicity.
Vancomycin: Glycopeptide with a very narrow spectrum, used as a last resort for MRSA. Resistance (VRE) is a significant clinical concern.
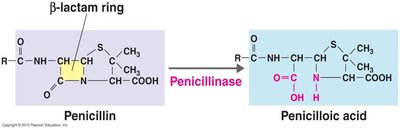
Beta-lactamase Resistance
Many bacteria produce beta-lactamases (e.g., penicillinase) that inactivate penicillins by breaking the beta-lactam ring. Semi-synthetic penicillins (e.g., methicillin) were developed to resist this, but resistance (e.g., MRSA) has emerged.
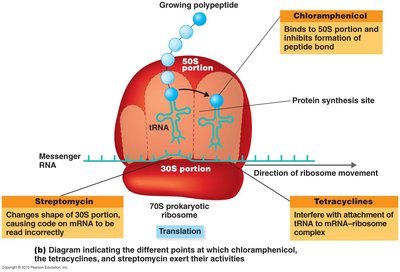
Protein Synthesis Inhibitors
These drugs target bacterial ribosomes (70S), which differ from eukaryotic ribosomes (80S). However, mitochondrial ribosomes resemble bacterial ribosomes, leading to potential toxicity.
Chloramphenicol: Broad-spectrum, inexpensive, but toxic. Used in eye drops for conjunctivitis.
Aminoglycosides: Bactericidal, bind to the 30S subunit. Examples: gentamycin, neomycin, streptomycin. Effective against gram-negatives but can cause auditory and kidney damage.
Tetracyclines: Broad-spectrum, bacteriostatic. Used for UTIs, gonorrhea, and chlamydial infections. Can cause superinfections and toxicity (e.g., teeth discoloration, liver damage).
Macrolides: Erythromycin, azithromycin, clarithromycin. Good for gram-positives and intracellular bacteria, but limited activity against gram-negatives.
Plasma Membrane Disruption
These drugs damage the bacterial plasma membrane, which is similar to that of human cells, resulting in high toxicity. Polymyxin B is used topically to avoid systemic toxicity.
Inhibition of Nucleic Acid Synthesis
Rifamycins: Block transcription (e.g., rifampin), used for tuberculosis and leprosy.
Fluoroquinolones: Inhibit DNA gyrase (e.g., ciprofloxacin), essential for DNA replication. Useful for severe infections but can affect cartilage development in children.
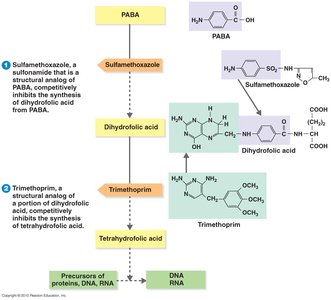
Inhibition of Essential Metabolite Synthesis
These drugs act as competitive inhibitors of key metabolic pathways. Sulfonamides (sulfa drugs) are structural analogs of PABA, blocking folic acid synthesis, which is essential for bacterial growth. Often used in combination (e.g., TMP-SMZ) for UTIs and burn patients.
Anti-Mycobacterial, Antifungal, Antiprotozoan, and Antiviral Agents
Anti-Mycobacterial Antibiotics
Very narrow spectrum, targeting Mycobacterium spp.
Inhibit mycolic acid synthesis (e.g., isoniazid, often combined with rifampin).
Combination therapy reduces resistance development.
Antifungal Agents
Target ergosterol in fungal membranes (not present in animal cells).
Polyenes: Fungicidal, create membrane pores (e.g., nystatin, amphotericin B).
Azoles: Fungistatic, inhibit sterol synthesis (e.g., miconazole, ketoconazole).
Griseofulvin: Interferes with mitosis, used for skin and hair infections.
Antiprotozoan Agents
Few available due to similarities with human cells.
Quinine: Anti-malarial, interferes with DNA replication.
Metronidazole (Flagyl): Inhibits fermentation in anaerobes, used for Giardia lamblia and some bacteria.
Antiviral Agents
Antibiotics are ineffective against viruses.
Most antivirals are specific (e.g., for HIV, herpes, influenza).
Nucleoside/nucleotide analogs: Block viral replication (e.g., acyclovir for herpes, AZT for HIV).
Interferons: Stimulate antiviral protein production, used for hepatitis.
Protease and neuraminidase inhibitors: Block viral assembly/release (e.g., Tamiflu for influenza).
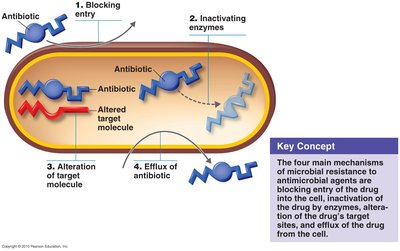
Antibiotic Resistance
Microorganisms can develop resistance to antibiotics through several mechanisms, often encoded on plasmids that can be transferred between bacteria. The four main mechanisms are:
Drug inactivation by microbial enzymes: Example: beta-lactamases (penicillinase) inactivate penicillins.
Decreased drug uptake: Prevents antibiotics from reaching their target.
Altered drug target: Mutation changes the antibiotic's binding site.
Efflux pumps: Actively expel antibiotics from the cell.
Additional info: Resistance genes are often carried on R plasmids, facilitating rapid spread among bacterial populations. The emergence of multidrug-resistant organisms (e.g., MRSA, VRE) is a major public health concern.
