 Back
BackAntimicrobial Drugs: Mechanisms, Clinical Considerations, and Resistance
Study Guide - Smart Notes

History and Types of Antimicrobial Agents
Introduction to Antimicrobial Agents
Antimicrobial agents are chemicals used to treat infections by inhibiting or killing microorganisms. Their development revolutionized medicine, enabling effective treatment of bacterial, fungal, and viral diseases.
Chemotherapeutic agents: Drugs that act against diseases, including infections.
Antimicrobial agents: Drugs specifically used to treat infections.
Semi-synthetics: Chemically modified antibiotics for improved efficacy.
Synthetics: Completely synthesized antimicrobials in laboratories.
Historical Figures in Antimicrobial Discovery
Paul Ehrlich: Developed arsenic compounds as "magic bullets" to kill microbes.
Alexander Fleming: Discovered penicillin from Penicillium mold.
Gerhard Domagk: Discovered sulfanilamide, a synthetic antimicrobial.
Selman Waksman: Coined the term "antibiotics" for naturally produced antimicrobial agents.
Example: Antibiotic Effect of Penicillium chrysogenum
Penicillium chrysogenum releases penicillin, which inhibits the growth of nearby bacteria such as Staphylococcus aureus. 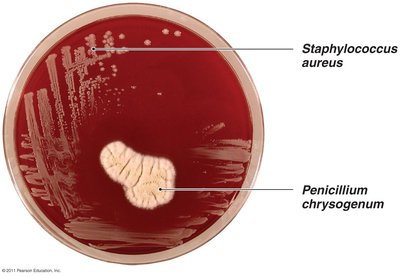
Mechanisms of Antimicrobial Action
Overview of Mechanisms
Antimicrobial drugs act through several distinct mechanisms, targeting specific structures or processes in microorganisms. Selective toxicity is key, allowing drugs to harm pathogens without damaging host cells.
Inhibition of cell wall synthesis
Inhibition of protein synthesis
Disruption of cytoplasmic membranes
Inhibition of metabolic pathways
Inhibition of nucleic acid synthesis
Prevention of virus attachment
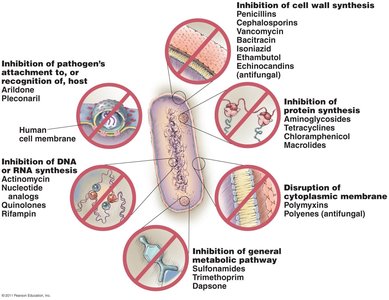
Summary Diagram of Drug Targets
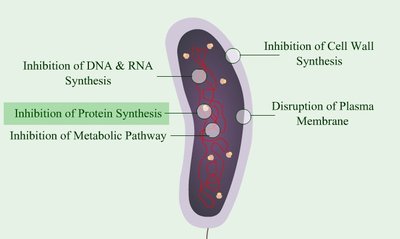
Inhibition of Cell Wall Synthesis
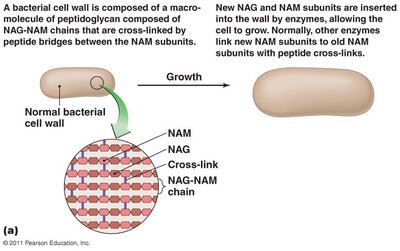
Many antibacterial agents target the synthesis of peptidoglycan, a key component of bacterial cell walls.
Beta-lactams: Bind to enzymes that cross-link NAM subunits, weakening the cell wall and causing lysis.
Vancomycin and cycloserine: Interfere with bridges linking NAM subunits in Gram-positive bacteria.
Bacitracin: Blocks secretion of NAG and NAM from the cytoplasm.
Isoniazid and ethambutol: Disrupt mycolic acid formation in mycobacteria.
Peptidoglycan Structure and Growth
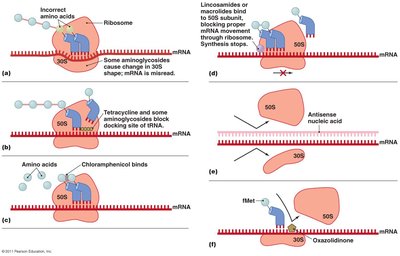
Inhibition of Protein Synthesis
Antimicrobials can selectively target prokaryotic ribosomes (70S), inhibiting translation and protein production.
Aminoglycosides: Cause misreading of mRNA.
Tetracyclines: Block docking site of tRNA.
Chloramphenicol: Binds to 50S subunit, inhibiting peptide bond formation.
Macrolides and lincosamides: Block movement of ribosome.
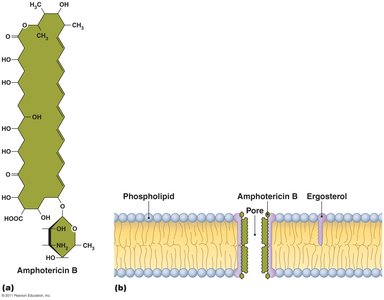
Disruption of Cytoplasmic Membranes
Some drugs damage membrane integrity, leading to cell death.
Amphotericin B: Attaches to ergosterol in fungal membranes, forming pores.
Azoles and allyamines: Inhibit ergosterol synthesis.
Polymyxin: Disrupts Gram-negative bacterial membranes; toxic to human kidneys.
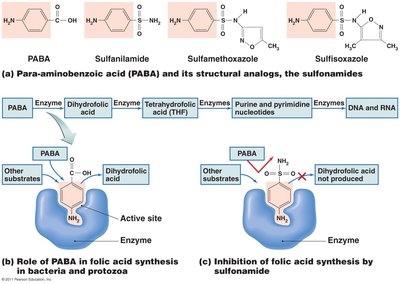
Inhibition of Metabolic Pathways
Antimetabolic agents block unique metabolic processes in pathogens.
Quinolones: Interfere with malaria parasite metabolism.
Heavy metals: Inactivate enzymes.
Sulfonamides: Block folic acid synthesis in bacteria and protozoa.
Inhibition of Nucleic Acid Synthesis
Drugs may block DNA replication or mRNA transcription, often affecting both prokaryotic and eukaryotic cells.
Nucleotide analogs: Distort nucleic acid shapes, preventing replication and transcription.
Quinolones and fluoroquinolones: Act against prokaryotic DNA gyrase.
Reverse transcriptase inhibitors: Target HIV replication.
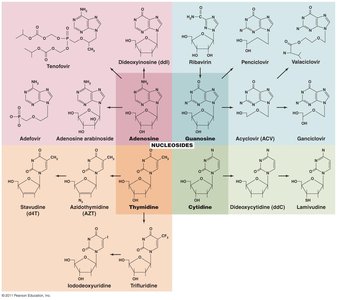
Prevention of Virus Attachment
Attachment antagonists: Block viral attachment or receptor proteins, preventing infection.
Clinical Considerations in Prescribing Antimicrobial Drugs
Ideal Antimicrobial Agent
The ideal drug is readily available, inexpensive, chemically stable, easily administered, nontoxic, nonallergenic, and selectively toxic against a wide range of pathogens.
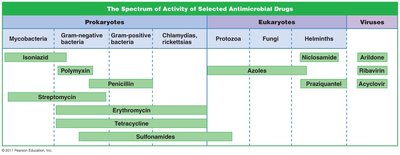
Spectrum of Action
Narrow-spectrum: Effective against a few organisms.
Broad-spectrum: Effective against many organisms; may cause superinfections by killing normal flora.
Spectrum of Activity Table
Drug | Prokaryotes | Eukaryotes | Viruses |
|---|---|---|---|
Isoniazid | Mycobacteria | ||
Polymyxin | Gram-negative bacteria | ||
Penicillin | Gram-positive bacteria | ||
Azoles | Fungi | ||
Acyclovir | Viruses |
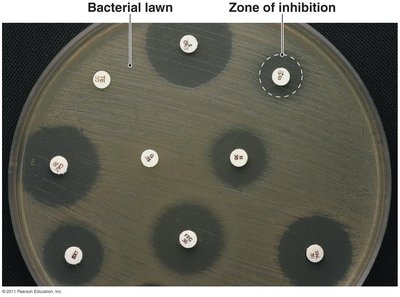
Efficacy Testing
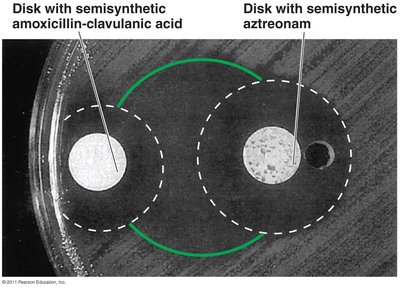
Diffusion susceptibility test: Measures zone of inhibition around drug disks.
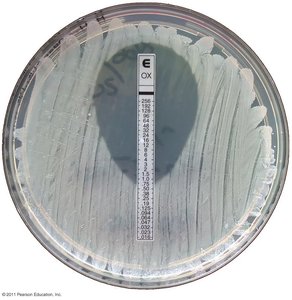
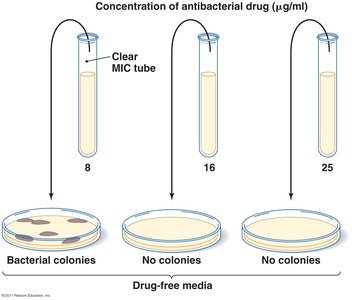
Minimum inhibitory concentration (MIC) test: Determines lowest concentration preventing growth.
Minimum bactericidal concentration (MBC) test: Determines lowest concentration killing bacteria.

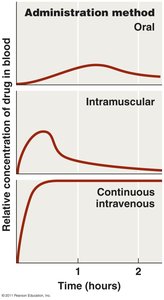
Routes of Administration
Topical: For external infections.
Oral: Self-administered, no needles.
Intramuscular: Injection into muscle.
Intravenous: Directly into bloodstream.
Safety and Side Effects
Toxicity: May affect kidneys, liver, or nerves; special consideration for pregnant women.
Allergies: Rare but potentially life-threatening (anaphylactic shock).
Disruption of normal microbiota: Can lead to secondary infections and superinfections, especially in hospitalized patients.
Resistance to Antimicrobial Drugs
Development of Resistance
Resistance arises naturally or is acquired through mutations or horizontal gene transfer (R-plasmids via transformation, transduction, conjugation). 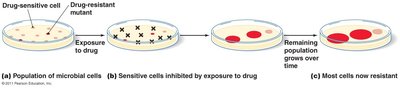
Mechanisms of Resistance
Enzyme production to destroy or deactivate drug (e.g., beta-lactamase).
Prevent drug entry into cell.
Alter drug target for reduced binding.
Change metabolic pathways.
Pump drug out of cell (efflux pumps).
Specific proteins (e.g., MfpA in Mycobacterium tuberculosis) bind targets to prevent drug action.
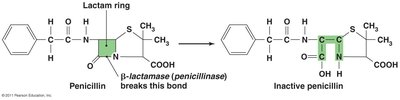
Multiple Resistance and Cross Resistance
Pathogens may acquire resistance to multiple drugs, especially in healthcare settings.
Cross resistance occurs when resistance to one drug confers resistance to similar drugs.
Superbugs are highly resistant strains.
Retarding Resistance
Maintain high drug concentration to kill sensitive cells.
Use drug combinations (synergism vs. antagonism).
Use antimicrobials only when necessary.
Develop new drug variations and search for new agents.
Design drugs to target microbial proteins specifically.
Summary Table: Mechanisms of Antimicrobial Action
Mechanism | Example Drugs | Target |
|---|---|---|
Cell Wall Synthesis Inhibition | Penicillins, Cephalosporins, Vancomycin | Peptidoglycan |
Protein Synthesis Inhibition | Aminoglycosides, Tetracyclines | Ribosomes |
Membrane Disruption | Polymyxin, Amphotericin B | Cytoplasmic membrane |
Metabolic Pathway Inhibition | Sulfonamides, Trimethoprim | Enzymes |
Nucleic Acid Synthesis Inhibition | Quinolones, Nucleotide analogs | DNA/RNA |
Virus Attachment Prevention | Arildone, Pleconaril | Viral receptors |
Key Equations and Concepts
Minimum Inhibitory Concentration (MIC)
The MIC is the lowest concentration of an antimicrobial agent that prevents visible growth of a microorganism.
Minimum Bactericidal Concentration (MBC)
The MBC is the lowest concentration of an antimicrobial agent that kills 99.9% of the original inoculum.
Zone of Inhibition
The diameter of the zone of inhibition in a diffusion test indicates the susceptibility of the microorganism to the drug.
Synergism vs. Antagonism
Synergism occurs when two drugs together have a greater effect than either alone; antagonism is when one drug reduces the effect of another.
Conclusion
Understanding the mechanisms, clinical considerations, and resistance patterns of antimicrobial drugs is essential for effective treatment and stewardship in microbiology and medicine. Continued research and careful use of these agents are vital to combat emerging resistance.
