 Back
BackAntimicrobial Drugs: Mechanisms, Clinical Considerations, and Resistance
Study Guide - Smart Notes

Controlling Microbial Growth in the Body: Antimicrobial Drugs
The History of Antimicrobial Agents
The development of antimicrobial agents revolutionized the treatment of infectious diseases. These agents include chemicals that affect physiology, chemotherapeutic agents (drugs acting against diseases), and antimicrobial agents (drugs treating infections).
Paul Ehrlich: Introduced the concept of “magic bullets” and developed arsenic compounds to kill microbes.
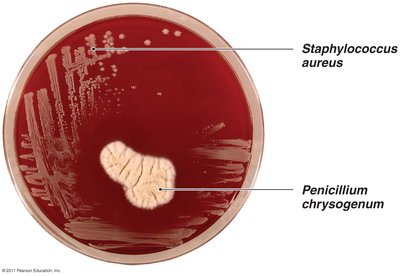
Alexander Fleming: Discovered Penicillin released from Penicillium.
Gerhard Domagk: Discovered sulfanilamide, the first widely used antimicrobial.
Selman Waksman: Coined the term “antibiotics” for naturally produced antimicrobial agents.
Semi-synthetics: Chemically altered antibiotics for improved efficacy.
Synthetics: Completely synthesized antimicrobials in laboratories.
Example: The antibiotic effect of Penicillium chrysogenum on Staphylococcus aureus demonstrates the principle of microbial antagonism.
Mechanisms of Antimicrobial Action
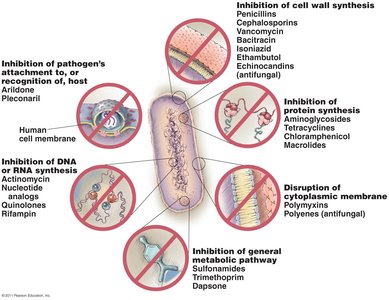
Antimicrobial drugs act through several mechanisms, targeting specific features of pathogens to achieve selective toxicity. The largest number of drugs are antibacterial, with fewer available for eukaryotic and viral infections.
Inhibition of cell wall synthesis
Inhibition of protein synthesis
Disruption of cytoplasmic membranes
Inhibition of metabolic pathways
Inhibition of nucleic acid synthesis
Prevention of virus attachment
Summary Diagram of Drug Action

Inhibition of Cell Wall Synthesis
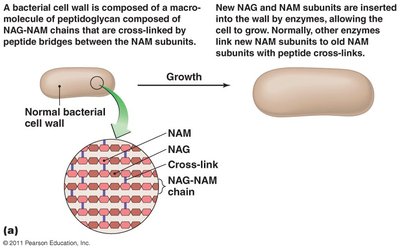
Drugs targeting cell wall synthesis are most effective against bacteria, especially during growth. They prevent cross-linkage of peptidoglycan subunits, weakening the cell wall and causing lysis.
Beta-lactams: Bind to enzymes that cross-link NAM subunits. Their functional group is the beta-lactam ring.
Semi-synthetic beta-lactams: More stable, better absorbed, less susceptible to deactivation, and active against more bacteria.
Vancomycin and cycloserine: Interfere with bridges linking NAM subunits in Gram-positives.
Bacitracin: Blocks secretion of NAG and NAM from cytoplasm.
Isoniazid and ethambutol: Disrupt mycolic acid formation in mycobacteria.
Example: Peptidoglycan structure and cell wall growth.
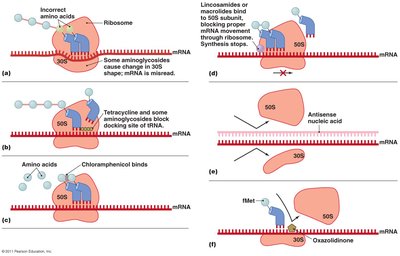
Inhibition of Protein Synthesis
Drugs can selectively target prokaryotic ribosomes (70S) without affecting eukaryotic ribosomes (80S), though mitochondrial ribosomes are similar to prokaryotic ones and may be affected.
Aminoglycosides: Cause misreading of mRNA.
Tetracyclines: Block docking site of tRNA.
Chloramphenicol: Binds to 50S subunit, inhibiting peptide bond formation.
Macrolides and lincosamides: Block 50S subunit.
Oxazolidinones: Block initiation of translation.
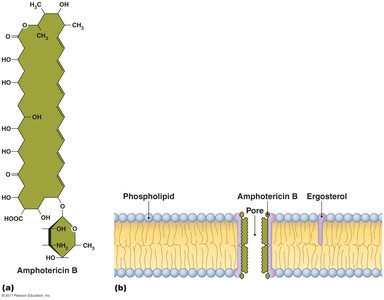
Disruption of Cytoplasmic Membranes
Some drugs damage membrane integrity by forming channels or inhibiting sterol synthesis.
Amphotericin B: Attaches to ergosterol in fungal membranes, forming pores.
Azoles and allyamines: Inhibit ergosterol synthesis.
Polymyxin: Disrupts Gram-negative bacterial membranes; toxic to human kidneys.
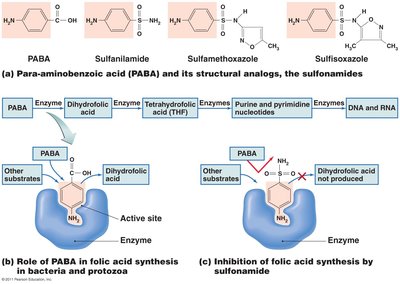
Inhibition of Metabolic Pathways
Antimetabolic agents are effective when pathogen and host metabolic processes differ.
Quinolones: Interfere with malaria parasite metabolism.
Heavy metals: Inactivate enzymes.
Sulfonamides: Block folic acid synthesis in bacteria and protozoa.
Antiviral agents: Target unique aspects of viral metabolism (e.g., amantadine prevents viral uncoating).
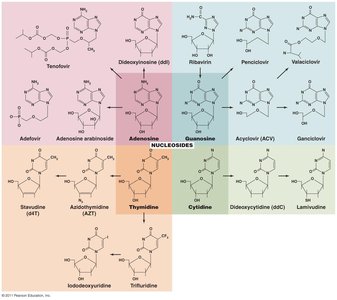
Inhibition of Nucleic Acid Synthesis
Drugs in this category block DNA replication or mRNA transcription, often affecting both eukaryotic and prokaryotic cells. Nucleotide analogs distort nucleic acid shapes, preventing replication, transcription, or translation.
Quinolones and fluoroquinolones: Act against prokaryotic DNA gyrase.
Reverse transcriptase inhibitors: Target HIV replication.
Prevention of Virus Attachment
Attachment antagonists block viral attachment or receptor proteins, representing a new area of drug development.
Clinical Considerations in Prescribing Antimicrobial Drugs
Ideal Antimicrobial Agent
The ideal agent is readily available, inexpensive, chemically stable, easily administered, nontoxic, nonallergenic, and selectively toxic against a wide range of pathogens.
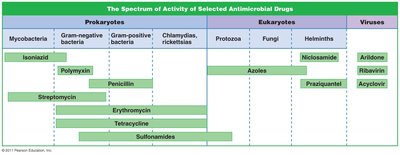
Spectrum of Action
The spectrum of action refers to the range of pathogens a drug affects. Narrow-spectrum drugs target few organisms, while broad-spectrum drugs target many. Broad-spectrum drugs may allow secondary or superinfections by killing normal flora.
Efficacy Testing
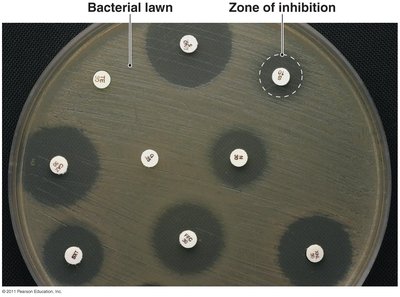
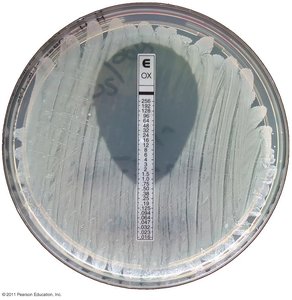
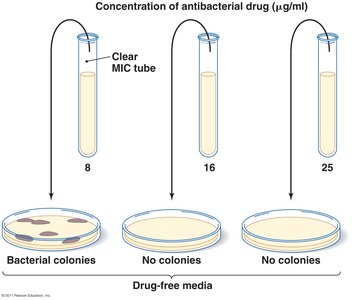
Efficacy is determined by laboratory tests:
Diffusion susceptibility test: Measures zone of inhibition.
Minimum inhibitory concentration (MIC) test: Determines lowest concentration preventing growth.
Minimum bactericidal concentration (MBC) test: Determines lowest concentration killing bacteria.

Routes of Administration
Drugs can be administered topically, orally, intramuscularly, or intravenously. The route affects drug distribution and concentration in tissues.
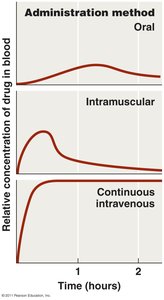
Safety and Side Effects
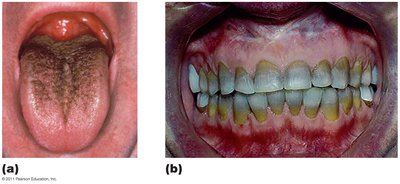
Adverse effects include toxicity (to kidneys, liver, nerves), allergies (rare but potentially life-threatening), and disruption of normal microbiota (leading to secondary infections or superinfections).
Resistance to Antimicrobial Drugs
The Development of Resistance in Populations
Resistance can be natural or acquired through mutations or acquisition of R-plasmids via transformation, transduction, or conjugation.
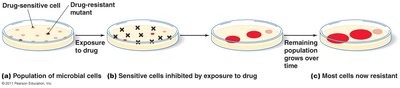
Mechanisms of Resistance
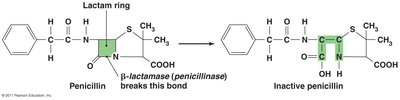
Microbes resist drugs by:
Producing enzymes that destroy or deactivate drugs (e.g., beta-lactamase).
Preventing drug entry into the cell.
Altering drug targets.
Changing metabolic chemistry.
Pumping drugs out of the cell.
Producing proteins that protect drug targets (e.g., MfpA protein in Mycobacterium tuberculosis).
Multiple Resistance and Cross Resistance
Pathogens may acquire resistance to multiple drugs, especially in hospital settings. Cross resistance occurs when resistance to one drug confers resistance to similar drugs.
Retarding Resistance
Strategies to slow resistance include maintaining high drug concentrations, using combinations of drugs (synergism vs. antagonism), limiting use to necessary cases, developing new drug variations, and designing drugs to target microbial proteins.
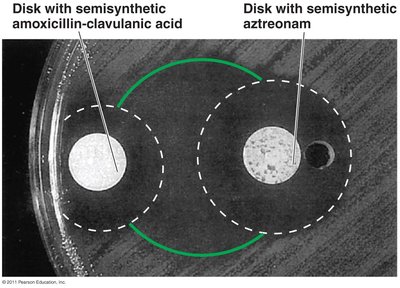
Table: Mechanisms of Antimicrobial Action
Mechanism | Example Drugs | Target Pathogen |
|---|---|---|
Inhibition of Cell Wall Synthesis | Penicillins, Cephalosporins, Vancomycin | Bacteria |
Inhibition of Protein Synthesis | Aminoglycosides, Tetracyclines | Bacteria |
Disruption of Cytoplasmic Membrane | Polymyxin, Amphotericin B | Bacteria, Fungi |
Inhibition of Metabolic Pathways | Sulfonamides, Trimethoprim | Bacteria, Protozoa |
Inhibition of Nucleic Acid Synthesis | Quinolones, Nucleotide analogs | Bacteria, Viruses |
Prevention of Virus Attachment | Arildone, Pleconaril | Viruses |
Additional info: This summary expands on brief points with academic context, definitions, and examples to provide a comprehensive study guide for antimicrobial drugs, their mechanisms, clinical considerations, and resistance.
