 Back
BackAntimicrobial Drugs: Mechanisms, Spectrum, and Resistance
Study Guide - Smart Notes

Antimicrobial Drugs
Introduction to Antimicrobial Agents
Antimicrobial drugs are substances used to treat infections by inhibiting or killing pathogenic microorganisms. They are a subset of chemotherapeutic agents, which are drugs used to combat diseases in general. The development of antimicrobial agents revolutionized medicine, allowing for the effective treatment of bacterial, fungal, and some viral infections.
Antibiotics: Naturally produced by microorganisms to inhibit or kill other microbes.
Semisynthetic drugs: Chemically modified derivatives of natural antibiotics to improve efficacy, stability, or administration.
Synthetic drugs: Entirely synthesized in the laboratory, not derived from natural sources.

Historical Milestones in Antimicrobial Discovery
Paul Ehrlich: Developed the concept of "magic bullets"—compounds that selectively target pathogens (e.g., arsenic compounds).
Alexander Fleming: Discovered penicillin from the mold Penicillium.
Gerhard Domagk: Discovered sulfanilamide, the first widely used synthetic antimicrobial.
Selman Waksman: Coined the term "antibiotics" and discovered streptomycin.
Principles of Antimicrobial Therapy
Selective Toxicity
Selective toxicity is the ability of a drug to target microbial cells without causing significant harm to the host. This principle is fundamental to effective chemotherapy. Drugs exploit differences between microbial and human cells, such as cell wall composition, ribosome structure, or metabolic pathways.
Classification by Mechanism of Action
Antimicrobial drugs are classified based on their target within the microbial cell:
Inhibition of cell wall synthesis
Inhibition of protein synthesis
Disruption of cytoplasmic membrane
Inhibition of metabolic pathways
Inhibition of nucleic acid synthesis
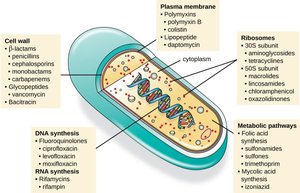
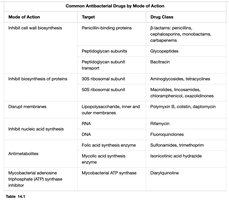
Mechanisms of Antimicrobial Action
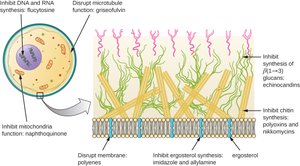
Inhibition of Cell Wall Synthesis
Many bacteria possess a cell wall composed of peptidoglycan, which is absent in human cells. Drugs targeting cell wall synthesis are highly selective for bacteria.
β-lactam antibiotics (e.g., penicillins, cephalosporins): Block cross-linking of peptidoglycan by binding to penicillin-binding proteins, leading to cell lysis.
Vancomycin & Cycloserine: Interfere with the formation of peptide bridges in Gram-positive bacteria.
Bacitracin: Inhibits transport of peptidoglycan precursors.
Isoniazid & Ethambutol: Disrupt mycolic acid synthesis in mycobacteria.
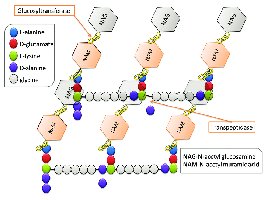
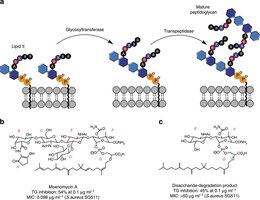
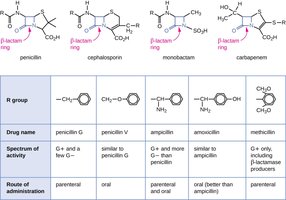
Limitations
Only effective against actively growing bacteria (not dormant cells).
Resistance can develop via β-lactamase enzymes that inactivate the drug.
Inhibition of Fungal Cell Wall Synthesis
Fungal cell walls contain unique polysaccharides (e.g., glucans) absent in humans, making them good drug targets. Echinocandins inhibit glucan synthesis, weakening the fungal cell wall.
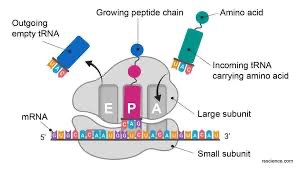
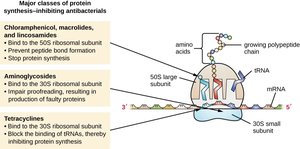
Inhibition of Protein Synthesis
Prokaryotic ribosomes (70S) differ from eukaryotic ribosomes (80S), allowing selective inhibition of bacterial translation. However, mitochondrial ribosomes resemble bacterial ribosomes, so some toxicity may occur.
Aminoglycosides: Bind 30S subunit, causing misreading of mRNA.
Tetracyclines: Block tRNA binding to the ribosome.
Macrolides, Chloramphenicol: Bind 50S subunit, inhibiting peptide bond formation or translocation.
Oxazolidinones: Inhibit initiation complex formation.
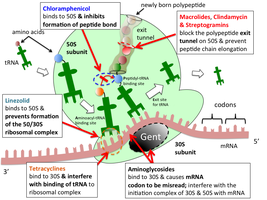
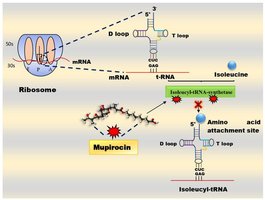
Special Case: Mupirocin
Mupirocin selectively inhibits isoleucyl-tRNA synthetase, blocking the attachment of isoleucine to tRNA, and is effective against Gram-positive bacteria.
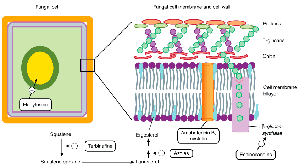
Disruption of Cytoplasmic Membranes
Certain drugs disrupt the integrity of microbial membranes, leading to cell death.
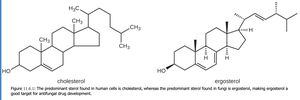
Nystatin & Amphotericin B: Bind ergosterol in fungal membranes, forming pores.
Polymyxins: Disrupt Gram-negative bacterial membranes; toxic to human kidneys.
Azoles & Allylamines: Inhibit ergosterol synthesis in fungi.
Inhibition of Metabolic Pathways
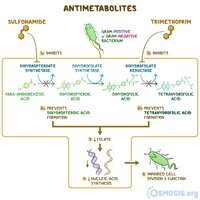
Some drugs act as metabolic antagonists, interfering with pathways unique to microbes.
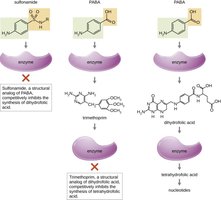
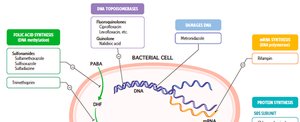
Sulfonamides: Inhibit folic acid synthesis by mimicking para-aminobenzoic acid (PABA).
Trimethoprim: Inhibits a later step in folic acid synthesis.
Atovaquone: Disrupts electron transport in protozoa and fungi.
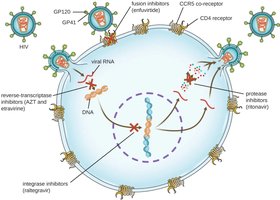
Antiviral agents: Block viral uncoating or enzyme activity (e.g., amantadine, protease inhibitors).
Inhibition of Nucleic Acid Synthesis
These drugs block DNA replication or RNA transcription, often affecting both prokaryotic and eukaryotic cells, so their use is limited to specific cases.
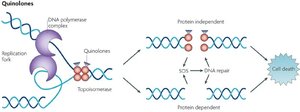
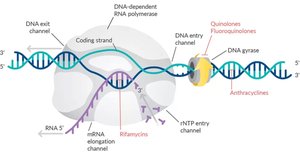
Quinolones & Fluoroquinolones: Inhibit DNA gyrase in bacteria.
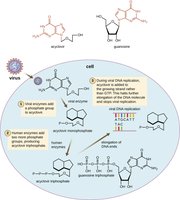
Nucleotide/Nucleoside analogs: Mimic DNA/RNA building blocks, causing chain termination (e.g., acyclovir for viruses).
Rifamycins: Inhibit bacterial RNA polymerase.
Reverse transcriptase inhibitors: Block HIV replication; safe for humans as we lack this enzyme.
Spectrum of Antimicrobial Activity
Narrow vs. Broad-Spectrum Drugs
The spectrum of action refers to the range of pathogens a drug can affect.
Narrow-spectrum: Effective against specific families of microbes (e.g., only Gram-positive bacteria).
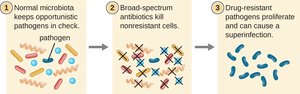
Broad-spectrum: Effective against a wide variety of organisms; may disrupt normal microbiota, leading to superinfections.
Clinical Considerations in Prescribing Antimicrobial Drugs
Evaluating Effectiveness
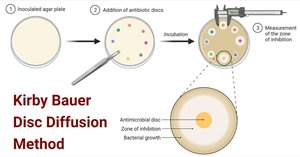
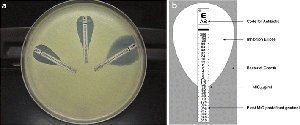
Diffusion susceptibility (Kirby-Bauer) test: Measures zone of inhibition around antibiotic discs on agar plates.
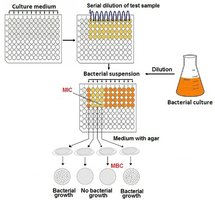
Minimum Inhibitory Concentration (MIC): Lowest concentration preventing visible growth.
Minimum Bactericidal Concentration (MBC): Lowest concentration that kills the microbe.


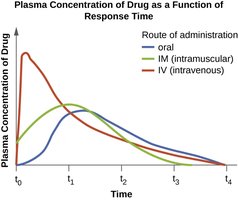
Routes of Administration
Topical: Applied to skin or mucous membranes.
Oral: Taken by mouth; convenient but variable absorption.
Intramuscular (IM): Injected into muscle; slower absorption.
Intravenous (IV): Directly into bloodstream; rapid effect.
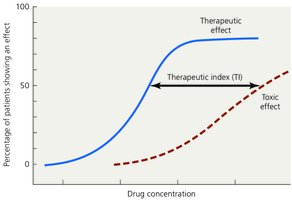
Therapeutic Index and Safety
Therapeutic Index (TI): Ratio of the highest tolerated dose to the effective dose; higher TI indicates greater safety.
Toxicity: Some drugs may harm organs (kidneys, liver, nervous system) or cause fetal harm.
Allergies: Rare but can be severe (e.g., anaphylactic shock).
Disruption of normal microbiota: May lead to secondary infections or overgrowth of opportunistic pathogens.
Antimicrobial Drug Resistance
Development and Spread of Resistance
Natural resistance: Some microbes are inherently resistant to certain drugs.
Acquired resistance: Results from chromosomal mutations or acquisition of resistance genes (R plasmids) via transformation, transduction, or conjugation.
Mechanisms of Resistance
Enzyme production (e.g., β-lactamase destroys penicillin)
Reduced permeability or increased efflux of drug
Alteration of drug target site
Metabolic pathway changes
Biofilm formation
Multiple and Cross Resistance
Multiple resistance: Resistance to several drugs, often due to R plasmids; common in hospital settings.
Cross resistance: Resistance to drugs with similar structures or mechanisms.
Strategies to Retard Resistance
Use high concentrations of drugs for the full course of treatment.
Use combinations of drugs (synergism) to reduce the likelihood of resistance.
Limit use of antimicrobials to necessary cases.
Develop new drugs and modify existing ones.
Clinical Case Study: Antibiotic Overkill
Case Summary
Patient developed oral and vaginal candidiasis after antibiotic treatment for a urinary tract infection.
Antibiotics disrupted normal microbiota, allowing Candida overgrowth.
Restoration of normal flora and targeted antifungal therapy are necessary to resolve the infection and prevent recurrence.
Prevention
Use antibiotics only when necessary and as prescribed.
Consider probiotics or other measures to restore normal microbiota after antibiotic therapy.
Conclusion
Antimicrobial drugs are essential tools in modern medicine, but their effectiveness is threatened by the development of resistance. Understanding their mechanisms, spectrum, and clinical considerations is crucial for their responsible use and for combating infectious diseases.
