 Back
BackAntimicrobial Drugs: Mechanisms, Spectrum, Resistance, and Clinical Applications
Study Guide - Smart Notes

Antimicrobial Drugs: Introduction
Overview and Selective Toxicity
Antimicrobial drugs are essential when the body's immune defenses cannot overcome infectious diseases. These drugs must exhibit selective toxicity, meaning they target microbial pathogens without harming the host.
Chemotherapy: Use of chemicals to treat disease, originally termed by Paul Ehrlich.
Antibiotic: Substance produced by a microbe that inhibits another microbe.
Antimicrobial drugs: Synthetic substances that interfere with microbial growth.
Selective toxicity: Ability to destroy pathogens without damaging host cells.
Spectrum of Antimicrobial Activity
Narrow vs. Broad Spectrum
The spectrum of activity refers to the range of microbes affected by a drug.
Narrow-spectrum antibiotics: Target a limited group of microbes, reducing risk of disrupting normal flora.
Broad-spectrum antibiotics: Affect a wide range of gram-positive and gram-negative bacteria; useful when the pathogen is unknown but may cause superinfections.
Superinfection
A superinfection occurs when a second infection arises during treatment, often by resistant microbes such as Clostridium difficile or Candida albicans.

Mechanisms of Action of Antimicrobial Drugs
Bactericidal vs. Bacteriostatic
Antimicrobial drugs can be classified by their effect on microbes:
Bactericidal: Kill microbes directly.
Bacteriostatic: Prevent microbial growth, allowing the immune system to eliminate pathogens.
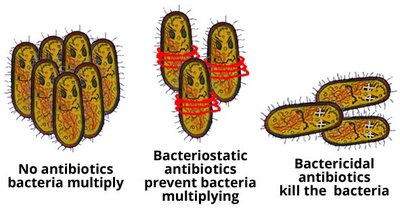
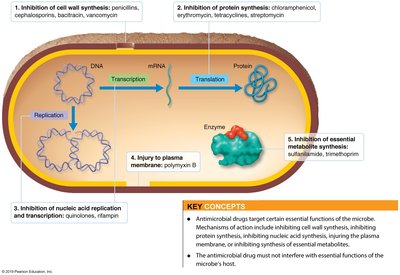
Major Modes of Action
There are five primary mechanisms by which antimicrobial drugs act:
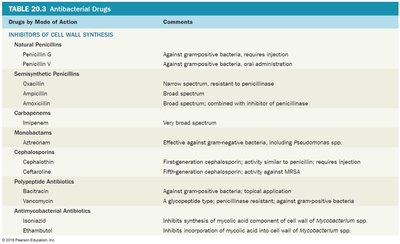
Inhibiting cell wall synthesis: Drugs like penicillins prevent peptidoglycan synthesis, leading to cell lysis.
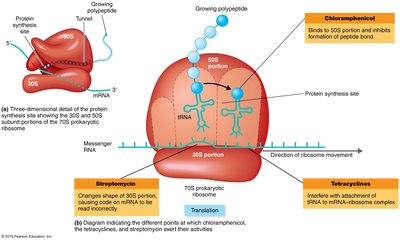
Inhibiting protein synthesis: Target bacterial 70S ribosomes, interfering with translation (e.g., streptomycin, tetracyclines).
Injuring the plasma membrane: Polypeptide antibiotics alter membrane permeability (e.g., polymyxin B).
Inhibiting nucleic acid synthesis: Interfere with DNA replication and transcription (e.g., quinolones, rifampicin).
Inhibiting synthesis of essential metabolites: Antimetabolites compete with substrates, blocking metabolic pathways (e.g., sulfanilamide inhibits folic acid synthesis).
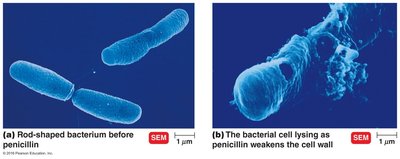
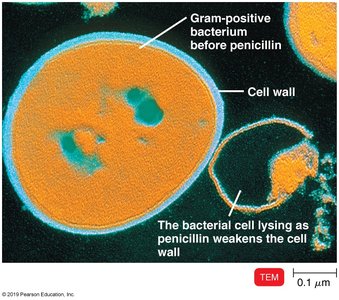
Antibacterial Antibiotics: Inhibitors of Cell Wall Synthesis
Penicillins and β-lactam Antibiotics
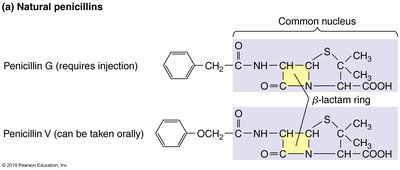
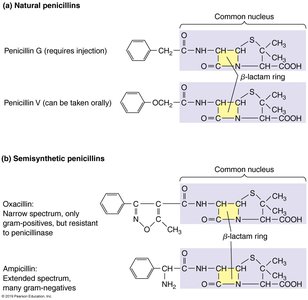
Penicillins contain a β-lactam ring that prevents cross-linking of peptidoglycan, weakening the cell wall and causing lysis.
Primarily affect gram-positive bacteria.
Only effective against actively growing cells.
Types of Penicillins
Natural penicillins: Penicillin G (injected) and Penicillin V (oral); narrow spectrum, susceptible to penicillinases.
Semisynthetic penicillins: Chemically modified to resist penicillinases and broaden spectrum.
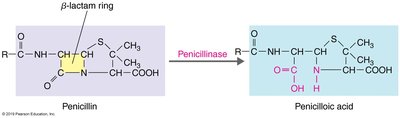
Penicillinases (β-lactamases)
Enzymes produced by bacteria (especially Staphylococcus species) that break the β-lactam ring, rendering penicillin inactive.
Other Cell Wall Synthesis Inhibitors
Bacitracin: Interferes with linear strand formation of peptidoglycan; effective against gram-positive bacteria.
Vancomycin: Inhibits cell wall synthesis; important for treating MRSA.
Inhibitors of Protein Synthesis
Mechanisms and Examples
These drugs exploit differences between prokaryotic and eukaryotic ribosomes for selective toxicity.
Streptomycin: Binds to 30S ribosomal subunit, causing misreading of mRNA.
Tetracycline: Blocks attachment of tRNA to mRNA, preventing amino acid addition.
Chloramphenicol: Inhibits formation of peptide bonds at the 50S subunit.
Antifungal Drugs
Agents Affecting Cell Membrane and Cell Wall
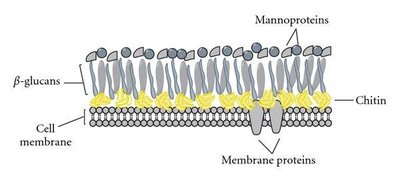
Fungal cell membranes contain ergosterols instead of cholesterol. Many antifungal drugs target ergosterol synthesis, increasing membrane permeability.
Polyenes: Nystatin and Amphotericin B; bind to ergosterol, causing cell leakage.
Azoles: Imidazoles; inhibit ergosterol synthesis, used for cutaneous mycoses.
Echinocandins: Inhibit β-glucan synthesis in cell wall, leading to lysis.
Antiviral Drugs
Mechanisms of Action
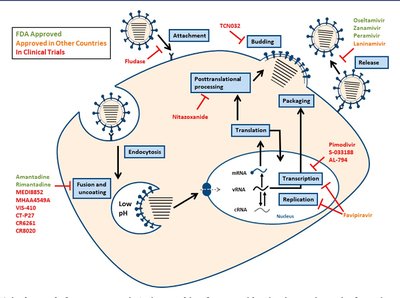
Antiviral drugs are challenging to develop due to viruses' reliance on host cells.
Entry & Fusion inhibitors: Block initial steps of viral infection.
Uncoating and genome integration inhibitors: Prevent viral uncoating and integration into host genome.
Nucleic acid analogs: Inhibit viral RNA or DNA synthesis.
Protease inhibitors: Block cleavage of protein precursors, preventing viral assembly.
Exit inhibitors: Inhibit neuraminidase, blocking viral release (e.g., Tamiflu).
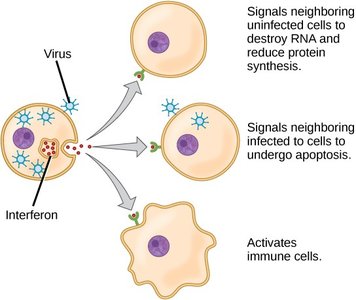
Interferons
Produced by virus-infected cells to inhibit further spread.
Alpha interferon is used to treat viral hepatitis.
Imiquimod promotes interferon production for HPV treatment.
Antiprotozoan and Antihelminthic Drugs
Antiprotozoan Drugs
Quinine and chloroquine: Treat malaria.
Artemisinin: Treat malaria; resistance is increasing.
Metronidazole: Interferes with DNA synthesis; treats trichomoniasis, giardiasis, amebic dysentery, and anaerobic bacteria.
Antihelminthic Drugs
Niclosamide: Prevents ATP production; treats tapeworms.
Praziquantel: Alters membrane permeability; treats tapeworms and flukes.
Mebendazole: Interferes with nutrient absorption; treats intestinal helminths.
Tests to Guide Chemotherapy
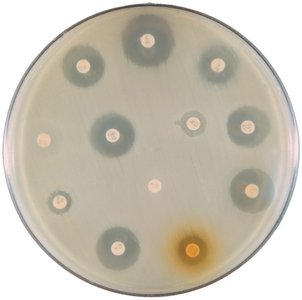
Disk-Diffusion Method (Kirby-Bauer Test)
Paper disks with drugs are placed on agar inoculated with the test organism.
Zone of inhibition indicates sensitivity to the antibiotic.
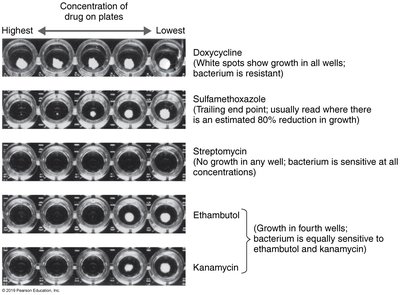
Broth Dilution Tests
Determine MIC (minimal inhibitory concentration) and MBC (minimal bactericidal concentration).
Test organism is placed in wells with varying drug concentrations; growth is assessed.
Resistance to Antimicrobial Drugs
Mechanisms of Resistance
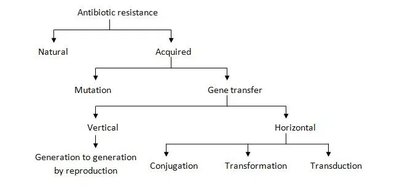
Microbes can be resistant by innate mechanisms or acquired genetic changes.
Innate resistance: Lack of drug target.
Acquired resistance: Mutations, plasmids, transposons; spread by transformation, conjugation, or transduction.
Persister cells: Survive antibiotic exposure due to genetic traits.
Superbugs: Resistant to multiple antibiotics.
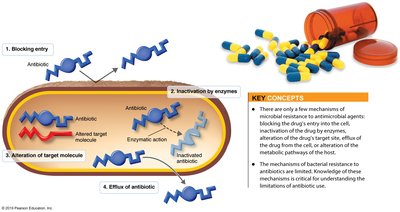
Mechanisms of Resistance
Enzymatic destruction or inactivation: e.g., β-lactamases.
Prevention of penetration: Gram-negative bacteria restrict entry.
Alteration of target site: Changes in ribosome or other targets.
Rapid efflux: Pumps remove antibiotics from cell.
Antibiotic Safety and Drug Combinations
Therapeutic Index and Safety
Therapeutic index: Ratio of risk to benefit; higher index is safer.
Possible adverse effects: organ damage, fetal risk, hypersensitivity, drug interactions.
Effects of Drug Combinations
Synergism: Combined effect greater than individual drugs.
Antagonism: Combined effect less than individual drugs.
Future of Chemotherapeutic Agents
Emerging Strategies
Targeting virulence factors (e.g., toxins).
Sequestering iron required for microbial growth.
Antimicrobial peptides (bacteriocins).
Phage therapy: Using bacteriophages to target bacteria.
----------------------------------------
