 Back
BackAntimicrobial Drugs: Mechanisms, Spectrum, Resistance, and Clinical Applications
Study Guide - Smart Notes

Antimicrobial Drugs: Introduction
History and Principles of Antimicrobial Drugs
Antimicrobial drugs are essential tools in modern medicine, designed to selectively target and kill or inhibit microorganisms while minimizing harm to the host. The concept of selective toxicity is central, meaning drugs are toxic to the target microbe but not to human cells. The development of these drugs began with Paul Ehrlich's "Magic Bullet" concept and the discovery of penicillin by Alexander Fleming, which revolutionized treatment of bacterial infections.
Selective toxicity: Drugs target microbial structures or processes not found in humans.
Antimicrobial: General term for drugs targeting microbes.
Antibiotic: Produced by microorganisms, often used to refer to antibacterial drugs.
Antiviral, Antifungal, Antiparasitic: Target viruses, fungi, and parasites respectively.
Threats: Effectiveness is threatened by antimicrobial resistance.
Magic Bullet: A molecule that kills microbes selectively.

How Antimicrobial Drugs Work
Mechanisms of Action and Selective Toxicity
Antimicrobial drugs act by targeting specific structures or processes in microbes. Selective toxicity is easier to achieve with prokaryotes due to their distinct cellular machinery. Drugs may be bactericidal (kill bacteria) or bacteriostatic (inhibit growth, allowing the immune system to clear infection).
Cell Wall Synthesis: Targets peptidoglycan unique to bacteria.
Cell Membrane: Targets unique lipids in bacterial membranes.
Nucleic Acid Synthesis: Targets bacterial replication and transcription enzymes.
Ribosome and Translation: Targets bacterial 70S ribosomes, distinct from human 80S ribosomes.
Metabolic Pathways: Targets pathways like folic acid synthesis, not present in humans.
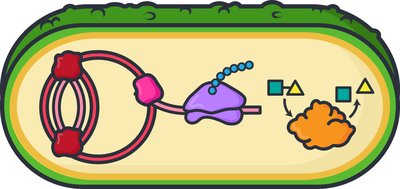
Spectrum of Antimicrobial Activity
Broad vs Narrow Spectrum Drugs
Antimicrobial drugs are classified by the range of microbes they affect. Broad-spectrum drugs target a wide variety, while narrow-spectrum drugs are specific to certain types. For bacteria, spectrum often depends on Gram-positive or Gram-negative classification.
Broad-spectrum: Effective against both Gram-positive and Gram-negative bacteria.
Narrow-spectrum: Effective against one group, often Gram-positive.
Gram-negative: Have an outer membrane; drugs must pass through porins.
Gram-positive: Single membrane; cell wall more accessible to drugs.
Superinfections
Consequences of Broad-Spectrum Antibiotic Use
Broad-spectrum antibiotics can disrupt normal microbiota, leading to superinfections—secondary infections by resistant microbes. Common superinfections include Candida yeast infections and Clostridium difficile (C-diff) diarrhea, often acquired in healthcare settings.
Superinfection: Secondary infection due to disruption of normal flora.
Candida: Causes yeast infections and thrush.
C. difficile: Causes severe diarrhea, often after antibiotic use.





























Drug Interactions: Synergism & Antagonism
Effects of Combining Antimicrobial Drugs
The effectiveness of antimicrobial drugs can be altered when used together. Synergism increases effectiveness, often by targeting different steps of a pathway or improving drug absorption. Antagonism decreases effectiveness, such as when bacteriostatic and bactericidal drugs interfere with each other.
Synergism: Two drugs enhance each other's effects.
Antagonism: Two drugs reduce each other's effects.

Therapeutic Window & Therapeutic Index
Drug Safety and Dosage
Drugs must be administered within a therapeutic window—a range where they are effective but not toxic. The therapeutic index is the ratio of toxic dose to effective dose; a higher index indicates greater safety.
Therapeutic window: Range of safe and effective dosages.
Therapeutic index:
Toxicity: Most likely affects liver (hepatotoxicity) or kidneys (nephrotoxicity).







Inhibitors of Cell Wall Synthesis: Beta-Lactam & Penicillin
Beta-Lactam Drugs
Beta-lactam antibiotics, including penicillins, cephalosporins, carbapenems, and monobactams, disrupt bacterial cell wall synthesis by inhibiting enzymes that cross-link peptidoglycan. This leads to cell lysis and death.
Beta-lactam ring: Core structure in these drugs.
Bactericidal: Cause cell lysis by disrupting cell wall integrity.
Peptidoglycan: Main component of bacterial cell wall.

Natural and Semisynthetic Penicillins
Natural penicillins are produced by Penicillium mold and are narrow-spectrum. Resistance is common due to beta-lactamase enzymes. Semisynthetic penicillins are chemically modified for broader spectrum and beta-lactamase resistance.
Penicillin G: Destroyed by stomach acid; used intravenously.
Penicillin V: Stable in stomach acid; can be taken orally.
Semisynthetic penicillins: Ampicillin, amoxicillin, methicillin, oxacillin.
Beta-lactamase inhibitors: Clavulanate often combined with amoxicillin.
Other Beta-Lactam Antibiotics
Cephalosporins, carbapenems, and monobactams are structurally distinct from penicillins but share the beta-lactam ring. They vary in spectrum and are often reserved for resistant infections.
Cephalosporins: Broad spectrum; multiple generations.
Carbapenems: Very broad spectrum; reserved for severe, resistant infections.
Monobactams: Single ring structure; narrow spectrum.
Inhibitors of Cell Wall Synthesis: Polypeptide Antibiotics & Isoniazid
Polypeptide Antibiotics and Antimycobacterial Agents
Some antibiotics target cell wall synthesis without a beta-lactam ring. Polypeptide antibiotics like bacitracin and vancomycin are bactericidal. Isoniazid blocks mycolic acid synthesis, effective against mycobacteria (e.g., tuberculosis).
Bacitracin: Available in topical ointments.
Vancomycin: Effective against Gram-positive bacteria.
Isoniazid: Narrow spectrum; treats tuberculosis.
Inhibitors of Protein Synthesis
Targeting Bacterial Ribosomes
Drugs that inhibit protein synthesis target the bacterial 70S ribosome, which is structurally different from the human 80S ribosome. Most are broad-spectrum.
Tetracyclines: Bind to 30S subunit, block tRNA binding.
Macrolides: Bind to 50S subunit, block exit of polypeptide chain.
Chloramphenicol: Bind to 50S subunit, block peptide bond formation.
Aminoglycosides: Bind to 30S subunit, cause mistranslation.
Disruptors of Cell Membranes
Targeting Bacterial Membrane Lipids
Some drugs disrupt bacterial cell membranes, leading to cell death. Lipopeptides and polymyxins are examples, with polymyxin B used topically and polymyxin E (colistin) reserved for severe Gram-negative infections due to toxicity.
Lipopeptides: Bind and disrupt cell membranes.
Polymyxin B: Topical use for Gram-negative infections.
Polymyxin E (colistin): Systemic use; nephrotoxic.
Inhibitors of Nucleic Acid Synthesis
Quinolones and Rifamycins
These drugs target bacterial enzymes involved in DNA replication and transcription. Quinolones inhibit topoisomerase and DNA gyrase, while rifamycins inhibit RNA polymerase.
Quinolones: Inhibit DNA replication enzymes.
Fluoroquinolones: Include a fluorine atom; e.g., ciprofloxacin.
Rifamycins: Inhibit RNA synthesis; e.g., rifampin for tuberculosis.
Competitive Inhibitors of Metabolic Pathways
Sulfa Drugs and Folic Acid Synthesis
Bacterial cells synthesize folic acid, which is essential for nucleotide production. Sulfonamides (sulfa drugs) and trimethoprim block different steps in this pathway, often used together for synergistic effect.
Sulfonamides: Competitive inhibitors; structurally similar to PABA.
Trimethoprim: Inhibits a later step in folic acid synthesis.
Combination therapy: Reduces resistance and increases effectiveness.
Antifungal Drugs
Mechanisms and Targets
Fungi are eukaryotes, making selective toxicity challenging. Antifungal drugs target ergosterol in cell membranes, β-glucan in cell walls, microtubule assembly, or nucleic acid synthesis.
Cell membrane disruption: Target ergosterol (e.g., azoles, polyenes).
Cell wall disruption: Inhibit β-glucan formation (e.g., echinocandins).
Cell division disruption: Inhibit microtubule assembly (e.g., griseofulvin).
Nucleic acid synthesis: Flucytosine interferes with DNA/RNA synthesis.
Antiviral Drugs
Mechanisms and Targets
Viruses replicate inside host cells, making selective toxicity difficult. Antivirals target stages of the viral lifecycle: attachment, replication, assembly, and exit.
Attachment/Entry inhibitors: Block viral binding to cells.
Replication inhibitors: Nucleic acid analogs inhibit viral genome replication.
Assembly/Exit inhibitors: Protease inhibitors prevent protein maturation; neuraminidase inhibitors block viral release.
Tests to Guide Antimicrobial Use
Disk Diffusion and Broth Dilution Methods
Laboratory tests determine the effectiveness of antimicrobials against specific bacteria. The Kirby-Bauer test uses paper disks on agar to measure zones of inhibition. The broth dilution test determines minimal inhibitory concentration (MIC) and minimal bactericidal concentration (MBC).
MIC: Lowest concentration inhibiting growth.
MBC: Lowest concentration killing bacteria.
Antimicrobial Resistance
Mechanisms and Spread
Antibiotic resistance arises through genetic mutations and horizontal gene transfer. Resistance mechanisms include decreased influx, rapid efflux, enzymatic inactivation, and alteration of target sites. Resistance genes can spread vertically (reproduction) or horizontally (transformation, conjugation, transduction).
Decreased influx: Changes to porins prevent drug entry.
Rapid efflux: Proteins pump drugs out of the cell.
Enzymatic inactivation: Enzymes like beta-lactamase destroy drugs.
Alteration of target site: Mutations change drug binding sites.
Practices Leading to Resistance Spread
Overuse and misuse of antibiotics in healthcare and agriculture select for resistant strains. Horizontal gene transfer can spread resistance genes between bacteria, including from animals to humans.
Overuse: Prescribing antibiotics when not needed.
Agriculture: Use in animals can transfer resistance genes to human pathogens.
Drug Class | Target | Spectrum | Example |
|---|---|---|---|
Beta-lactams | Cell wall synthesis | Varies | Penicillin, cephalosporin |
Polypeptides | Cell wall synthesis | Narrow | Bacitracin, vancomycin |
Protein synthesis inhibitors | Ribosome | Broad | Tetracycline, macrolide |
Membrane disruptors | Cell membrane | Narrow | Polymyxin B, colistin |
Nucleic acid inhibitors | DNA/RNA synthesis | Broad | Quinolone, rifamycin |
Metabolic inhibitors | Folic acid synthesis | Broad | Sulfa drugs, trimethoprim |
Antifungals | Ergosterol, cell wall | Narrow | Azole, echinocandin |
Antivirals | Viral lifecycle | Narrow | Acyclovir, oseltamivir |
