 Back
BackAntimicrobial Drugs: Principles, Mechanisms, and Resistance
Study Guide - Smart Notes

Antimicrobial Drugs
Definition and Historical Impact
Antimicrobial drugs are therapeutic compounds designed to kill or inhibit the growth of microbes, thereby preventing or treating infectious diseases. Their development revolutionized modern medicine, drastically reducing mortality from infections that were once often fatal.
Antimicrobial drug: Any substance that kills or inhibits the growth of microorganisms, including bacteria, viruses, fungi, and parasites.
Historically, minor infections could be deadly; the introduction of purified antimicrobials in the 20th century changed this.

Discovery of Penicillin
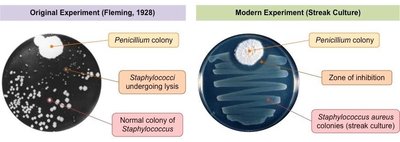
Alexander Fleming discovered penicillin in 1928 when he observed that a mold (genus Penicillium) inhibited the growth of Staphylococcus aureus on a culture plate. This led to the development and mass production of the first clinically useful antibiotic in the 1940s.- PENICILLIN
Zone of inhibition: Area around the mold where bacterial growth is prevented, indicating antimicrobial activity.
Spectrum of Activity and Empiric Therapy
1. Basic Criteria for drugs:
Effective against microbes
sufficiently non-toxic to the host
able to be purified in high amounts
2. Spectrum of Antimicrobial Activity
The spectrum of activity refers to the range of microbes an antimicrobial agent can affect.
Broad-spectrum: Effective against a wide variety of microbes; useful when the pathogen is unknown but can disrupt normal microbiota.
Narrow-spectrum: Targets a limited group of microbes; preferred when the pathogen is identified to minimize collateral damage. It can take days.
Limited-spectrum: Effective against a single organism or disease.
Empiric Therapy
Empiric therapy = of medical treatment with a broad-spectrum drug initiation while waiting for culture/ID results
–”Educated guess” based on clinical experience and observation in the absence of complete information
NO initial empiric choice is perfect
–Send for culture! Identify microbe! Ensure follow-up!
–Switch to a narrow-spectrum drug specific to the pathogen
Classification and Modification of Antimicrobial Drugs
Sources of Antimicrobials
Naturally occurring: Produced by microorganisms (e.g., soil bacteria, fungi).
Synthetic: Fully manufactured by chemical processes.
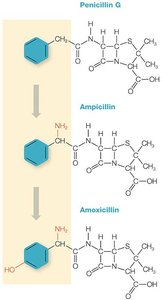
Semisynthetic: Chemically modified natural antibiotics to improve efficacy, spectrum, or pharmacokinetics.
Drug Modification and Generations
Antimicrobial compounds can be modified by chemical means (e.g., adding R groups
– Next-generation drugs result from each successive round of chemical modification (1st Gen, 2nd Gen, etc)
Drugs in later generations have expanded capabilities over their predecessors:
– Altered spectrum of activity
– Better penetration of different cell wall types
– Increased stability (longer half-life)
– Higher potency
– Circumvent resistance mechanisms
– Easier synthesis/purification/administration
– Lower toxicity
Example: Ampicillin and amoxicillin are semisynthetic derivatives of penicillin G, with broader activity due to added chemical groups.
Principles of Antimicrobial Drug Safety
Selective Toxicity and Therapeutic Index
Selective toxicity is achieved by exploiting vulnerable targets for the drug in the microbe that do not exist in the patient.
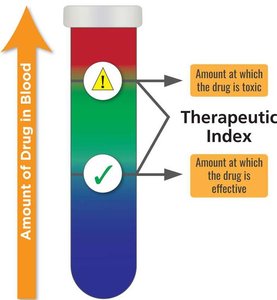
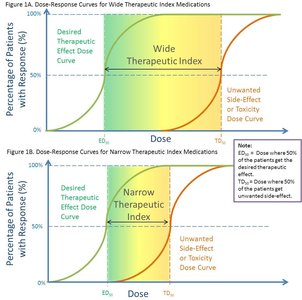
Selective toxicity is the ability of a drug to target microbial processes without harming the host. The therapeutic index (TI) quantifies drug safety:
Therapeutic Index (TI):
TI= ratio of the toxic dose (to the patient) to the theraputic dose (to eliminate the infection).
High TI = safer drug; low TI = higher risk of toxicity.
Drugs with a wide/high TI have a large margin of safety and less danger of producing toxic effects vs drugs with a narrow/low TI.
Toxicity Considerations
Antimicrobials can cause toxicity, especially to the liver (hepatotoxicity), kidneys (nephrotoxicity), and gut microbiome. They metabolize and eliminate drugs.
Hepatotoxicity: Drug-induced liver injury; a common reason for discontinuing drugs.
Nephrotoxicity: Kidney damage; antimicrobials are a leading cause.
Gut microbiome toxicity: Disruption of normal intestinal flora.
Routes of Administration
Oral: Preferred for convenience and cost;
must be stable in stomach acid and absorbed in the intestines, but not irritating to GI mucosa.
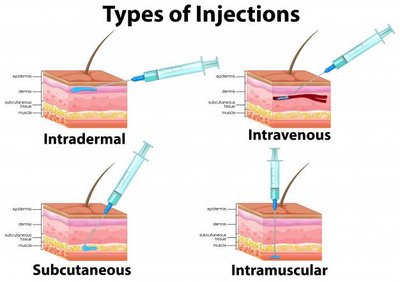
Parenteral: Non-oral routes (not involving the intestines)
(e.g., intravenous, intramuscular, subcutaneous, intradermal); used for rapid or targeted delivery.
Downside: needles are used, and needle phobia; injection discomfort; infection risk
Drug Half-Life and Interactions
Half-life: Time required for half the drug to be eliminated from the body; determines dosing frequency; time to reach steady state; duration of drug activity.
Short half-life = frequent administration
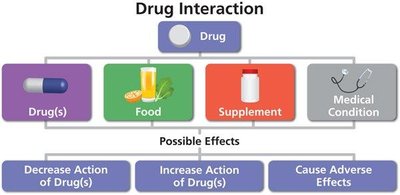
Drug interactions: Some antimicrobials interact with other drugs, foods, or supplements, affecting efficacy or safety.
Survey of Antimicrobial Drugs
Antibiotics vs. Antimicrobials
Antibiotics: Subset of antimicrobials that specifically target bacteria.
Antimicrobials: Broader term including agents against bacteria, viruses, fungi, and parasites.
Bacteriostatic vs. Bactericidal Drugs
Bacteriostatic: Inhibit bacterial growth; rely on the immune system to clear infection.
Bactericidal: Kill bacteria directly; useful in immunocompromised patients or severe infections.
CATEGORY | BACTERICIDAL | BACTERIOSTATIC |
|---|---|---|
Definition | Kills bacteria | Inhibits bacterial growth |
Minimum Concentration | MBC (minimum bactericidal concentration): lowest conc. needed to kill bacteria. | MIC (minimum inhibitory concentration): lowest conc. to inhibit growth |
# of bacterial cells | number decreases | # remains the same |
Viability | Bacteria die | Bacteria remain viable |
Activation of Immune System | No effect | Helps immune system deal with infection |
High dose effect | kills all bacterial cells | may act as a bactericidal at high dose |
Low dose effect | acts as bacteriostatic | not effective |
Reversibility | Irreversible | Reversible |
Mechanisms of Action
Antibacterial drugs exploit differences between prokaryotic and eukaryotic cells. Major targets include:
Bacteriostatic: inhibition of...
Metabolic pathways- folic acid synthesis (e.g., sulfa drugs, trimethoprim)
Protein synthesis (e.g., tetracyclines, macrolides)
Bactericidal: inhibition of...
Nucleic acid synthesis (e.g., quinolones, rifamycins)
Cell membrane integrity (e.g., polymyxins)
Cell wall synthesis (e.g., penicillins, cephalosporins)
Antiviral, Antifungal, and Antiparasitic Drugs
Antivirals: Target steps in viral replication or stimulate host immune responses; difficult to design due to reliance on host cell machinery.
Do not destroy their target pathogen
Goal – inhibit viral development to treat active infection
Antiviral drugs available today designed against HIV, herpes viruses, hepatitis viruses (B,C), influenza viruses, SARS-CoV2
Difficult to design safe and effective antiviral drugs
Viruses use host cells to replicate
Challenging to ID drug targets specific to the virus that won’t also harm the host organism's cells
Aim – target and disable essential virus-specific proteins shared among multiple viral strains and unlike host-cell components
Antiviral drugs are mainly effective against actively replicating viruses
Latent viruses are difficult to treat
Antiviral Drug Targets
1st category – target viral replication cycle: 1. Attachment 2. Penetration 3. Uncoating 4. Viral replication 5. Viral assembly 6. Viral release
2nd category – stimulate the host’s anti-viral immune responses (e.g., interferons)
Vaccination remains the primary and most effective way to reduce severe viral infection and disease (prevent vs treat)
Antifungals: Target fungal-specific structures (e.g., ergosterol in membranes, cell wall synthesis); toxicity is a concern due to eukaryotic similarities.
Antiparasitics: Target unique features of protozoa or helminths; often limited by toxicity and complex life cycles.
Antifungal Drug Targets
Mycoses – range from common benign infections like 'jock itch’ and ’athlete’s foot’ to serious, life-threatening infections such as cryptococcal meningitis
Most antifungal drugs target fungal-specific compounds not in host cells
A. Interfere with nucleic acid synthesis ▪ Leads to failure of fungal cell replication
B. Interfere with cell wall synthesis resulting in lysis ▪ Targets synthesis of essential fungal CW components (ex. glucans, chitin)
C. Interfere with cell membrane stability and structure causing death ▪ Targets essential CM sterol ergosterol (not cholesterol)
Antiparasitic Drug Targets
intracellular components– Function to ‘stun’ (paralyze, detach) or ‘kill’
Assessing Sensitivity to Antimicrobial Drugs
Antibiotic Susceptibility Testing (AST)
Patient specimens are sent for culture and A ntibiotic S T esting (AST) usceptibility
– Samples assessed include blood, urine, cerebrospinal fluid, sputum, wound exudates, stool, and other body fluids/discharge
AST is performed by clinical microbiology laboratories
– Determine organism causing the specific infection and which antimicrobials will inhibit the growth of the infecting microbe
– Identify and report drug resistance in pathogens
AST methods include drug diffusion and minimum inhibitory concentration (MIC) assays
– Large healthcare facilities also utilize commercial molecular systems to characterize bacterial resistance
Agar Diffusion Tests: Kirby Bauer vs Etest Agar Assays
Kirby- Bauer (Disk diffusion) | E-test (epsilometer test) |
|---|---|
|
|
|
|
|
|
Broth Dilution Tests
Differentiate between static (inhibit) vs cidal (kill) drug concentrations
1. Minimum Inhibit ory Concentration (MIC) assays
• Determine the lowest concentration of an antimicrobial agent that prevents visible growth
Antibiotic is serially diluted in growth media and a standardized amount of bacterial culture is added to each tube
Following incubation, MIC is determined
–Level of growth is measured by assessing turbidity or colorimetric indicators
–MIC = Lowest concentration of drug at which growth is completely inhibited (absence of visible turbidity/color)
2. Minimum Bactericidal Concentration (MBC) assays
• Determine the lowest concentration of an antimicrobial agent required to kill the organism .
• Defined as a ≥99.9% reduction of the initial inoculum within a fixed period of time (usually 24 hrs)
Plate the dilution where the MIC was observed along with at least 2 adjacent samples onto agar media that does not contain antibiotics
Only live cells will be able to grow
MBC = minimum concentration of drug that kills at least 99.9% of bacteria present in the assay
Antimicrobial Drug Resistance and Stewardship
Types o1f Resistance
Intrinsic resistance: Natural, due to inherent structural or functional traits (e.g., lack of target, impermeable cell wall).
Natural resistance to antimicrobial drugs based on inherent microbial structure/function
Makes certain pathogens/infections harder to treat/eliminate
Acquired resistance: Due to genetic mutations or horizontal gene transfer (conjugation, transformation, transduction).
3 main types of acquired resistance to evade antimicrobial drugs:
(1) Alter drug’s target
(2) Inactivate drug
(3) Reduce drug concentrations inside the cell
A. Block drug entry = decrease cell permeability
B. Pump the drug out = efflux pumps
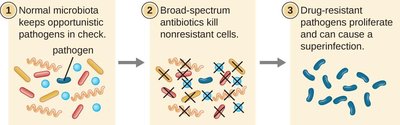
Superbugs Cause Superinfections
Superbugs: Microbes resistant to multiple antimicrobials that are commonly used
Superinfection: result when superbugs emerge from the initial infection resistant to the treatment and become the primary cause of a second infection
Long-term use of broad-spectrum antimicrobials sets the stage for superinfections
Factors Accelerating Resistance
Noncompliance with dosing
Misuse in healthcare and agriculture
Unregulated use in developing countries
Antimicrobial Stewardship
Stewardship programs promote appropriate use of antimicrobials to improve outcomes, reduce resistance, and limit the spread of multidrug-resistant organisms. Key strategies include:
Using narrow-spectrum drugs when possible
Educating healthcare workers and patients
Following proper dosing and infection control practices
Summary Table: Key Concepts
Concept | Explanation |
|---|---|
Antimicrobial drug | Compound that kills or inhibits microbes |
Spectrum of activity | Range of microbes affected (broad, narrow, limited) |
Therapeutic index | Ratio of toxic to therapeutic dose; higher is safer |
Bacteriostatic | Inhibits growth |
Bactericidal | Kills bacteria |
Intrinsic resistance | Natural, structural resistance |
Acquired resistance | Genetic mutation or gene acquisition |
Superinfection | Secondary infection by resistant microbes |
Stewardship | Appropriate use to limit resistance |
